Today, progress is widely being made in targeted cancer therapy. Most current drug targets are G-protein coupled receptors, nuclear receptors, ion channels or enzymes. Nevertheless, up to 80% of proteins remain “undruggable” meaning that small molecule drugs are unable to target given proteins to produce desirable effects. Substantial and significant efforts have been made among scientists to improve the small molecule drugs.
Now, a "revolutionary" targeting strategy has emerged - PROTAC, Proteolysis Targeting Chimera. PROTAC technology can degrade target proteins regardless of their function, making it possible to degrade non-druggable proteins, expanding the range of proteins being targeted for drug discovery. Looking further back, in March 2019, PROTAC entered clinical trials for the first time. Amazingly, within a few years, over 200 targeted protein degraders are currently in the different clinical-trial stages, in which are mainly PROTAC molecules. To learn how PROTAC can advance drug discovery research, we herein provide a detail summary on PROTAC technology and its perspectives in future.
1. Background of PROTAC
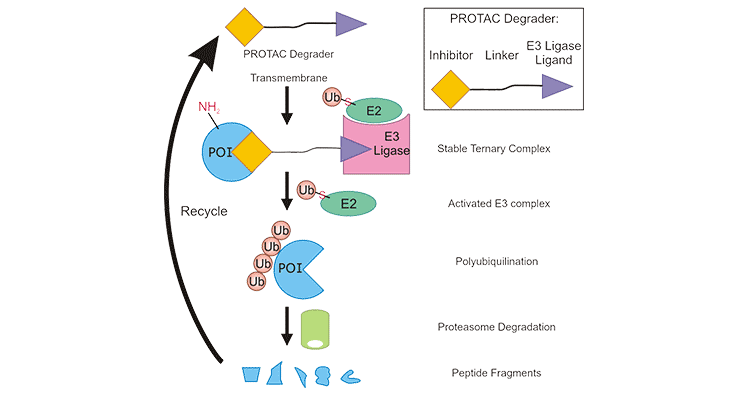
Proteolysis targeting chimeras (PROTAC) is a novel technology for targeted protein degradation. In 2001, Professors Craig Crews and Raymond Deshaies described the PROTAC idea and reported the first PROTAC molecule PROTAC-1, which was designed to target the methionyl aminopeptidase-2 (METAP2), as a pioneering new technology [1]. In 2008, Crews' team used Nutlin recruitment MDM2 to initiate a degradation cascade for targeting the androgen receptor (AR). This small molecule recruits the AR to the vicinity of MDM2, ubiquitinating it and subsequently degrading it by the proteasome complex [2]. This chimeric molecule, capable of targeting the protein for degradation, is named PROTAC (Proteolysis Targeting Chimera) (Figure 1) [3].
Nowadays, PROTAC has gained the attention of many cancer drug developers. Different PROTAC molecules have been developed for targeted protein reduction as an alternative approach to cancer therapy. PROTAC can inactivate cancer cells by inducing the target protein into the protein degradation system without necessarily binding to the active site of the target protein. At the same time, it can overcome the drug resistance problems of traditional small molecules. To date, many companies are involved in PROTAC R&D, which is favored by research institutes, pharmaceutical companies, and investment institutions.
Figure 1. PROTAC: a novel targeted protein degradation technology [3]
2. What is PROTAC Technology?
2.1 The "Ubiquitin-Proteasome System" (UPS)
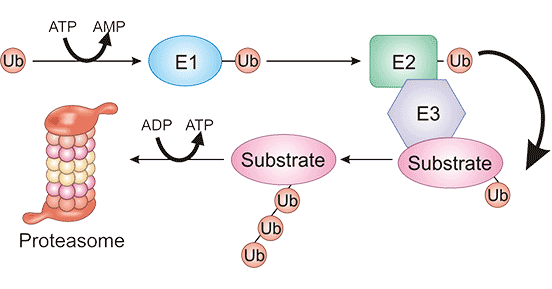
The Ubiquitin Proteasome System (UPS), or ubiquitin-mediated protein degradation system, is the pathway taken by PROTAC to degrade proteins [4]. The UPS is also the main pathway in the degradation of more than 80% of intracellular proteins, which is to degrade proteins that are damaged or no longer needed in the cell. In general, abnormalities in degradation often lead to diseases such as tumors [5, 6]. Which is to say, ubiquitin molecules act on a series of enzymes to sort intracellular proteins, from which target protein molecules are selected and specifically modified to form target protein polyubiquitin chains. Then the polyubiquitin chains direct the degradation of substrates at the proteasome. In the UPS process, the related enzymes mainly include E1 activating enzymes, E2 conjugating enzymes, and E3 ubiquitin ligases.
Specifically, the ubiquitin-proteasome system degrades proteins in a four-stage pathway (Figure 2): (i) E1, ubiquitin activation, under the energy supply of adenosine-triphosphate (ATP), the C-glycine residue of ubiquitin (Ub) forms a high energy thioester bond with the cysteine residue of E1; (ii) E1 connects the activated Ub to E2 through the intermediate product of Ub adenylation to form the E2-Ub sulfhydryl ester; (iii) E3, a key element in UPS pathway, which is needed to identify specific substrates; (iv) polyubiquitin (polyUb) chain is recognized and degraded by proteasome [7].
Figure 2. The pathway of protein degradation via the "ubiquitin-proteasome system" [7]
2.2 The Characteristics and Mechanism of PROTACs
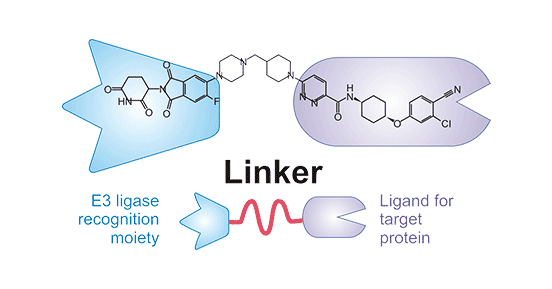
The existing PROTACs consist of three parts: a ligand for recruiting a target protein of interest (POI), a ligand for an E3 ubiquitin ligase, and an appropriate linker (Figure 3) [7]. PROTAC is a bifunctional molecule that binds to both target proteins and E3. Thus, PROTAC could target proteins that would otherwise not bind E3 to be ubiquitinated, recognized, and degraded by the proteasome. Unlike traditional small molecules targeting the active site of protein kinases, the target protein of PROTAC can be a protein with no obvious active site, which greatly broaden the strategies of PROTAC molecule and allow researchers to use existing E3 ligases to develop newly target proteins.
Figure 3. The Structure of PROTACs [7]
2.3 "Target Protein-PROTAC-E3 Ubiquitin Ligase" Ternary Complex
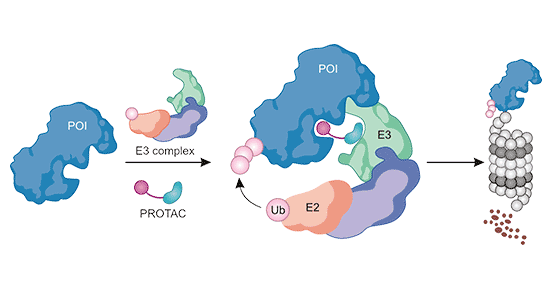
The formation of the "target protein-PROTAC-E3 ubiquitin ligase" ternary complex is the core of PROTAC-induced degradation of target proteins. The ternary complex is a necessary step in the polyubiquitination modification of target proteins by PROTAC. PROTAC can only function when it binds to both the target protein and E3 ligase to form a ternary complex (Figure 4) [8].
To date, more than 600 human genomes encoded E3 ligases have been identified. Several E3 ubiquitin ligases applied to the design of PROTAC molecules have been revealed, including CRBN, apoptotic protein inhibitor VHL, apoptosis protein inhibitory proteins (IAPs), MDM2 and DCAF16 etc. However, most of the clinical PROTAC pipeline disclosed E3 degrading enzymes are CRBN. Other studies suggest that KLHL41 expressed in skeletal muscle, RNF182 and TRIM9 expressed in the nervous system, and CDC20, CIAO1, WD82 expressed in tumors may be used for PROTAC molecular design, which will further enrich the properties of PROTAC [9-10].
At the same time, lots of target proteins are involved in the design of PROTAC including nuclear receptors (ER, AR, RAR), protein kinases (AKT, RIPK2, and CDK9, TBK1, BTK, TBK1, BCR-ABL, CDK2/4/6/9, ALK, CK2, PI3K, ERK1/2), protein transcriptional regulatory proteins (BRD4, Sirt2, TRIM24, HDAC6), regulatory proteins (ERRα, FKBP12, and TACC3), neurodegenerative-associated proteins (Huntingtin, Tau, α-synuclein), extracellular metabolic enzymes (MetAP-2, DHODH), epigenetic-related proteins (PCAF/GCN5), and more than 30 proteins and their mutants [9-12, 26].
Figure 4. "Target protein-PROTAC-E3 ubiquitin ligase" ternary complex formation process [8]
3. Applications of PROTACs in Cancer Therapeutic Research
PROTAC has overturned the concept of traditional small molecule inhibitors by using the ubiquitin proteasome pathway to degrade target proteins. PROTAC may exert highly effective at small doses and reduces drug resistance. Besides, PROTAC can act on "non-druggable" proteins. Currently, PROTAC technology has been successfully implemented in clinical studies for prostate cancer, breast cancer, non-small cell lung cancer and other diseases. Many targets have been successfully degraded in clinical trials e.g., AR, ER, EGFR, KRAS, ABL [13-27].
3.1 PROTAC in Prostate Cancer
Prostate cancer is a very common cancer in men. The androgen receptor (AR) is the key indicator for the diagnosis of this disease. AR antagonists such as Enzalutamide have been used with significant efficacy in the treatment of prostate cancer patients, but resistance often occurs. PROTAC against AR has been repeatedly shown to be superior to Enzalutamide, particularly in cases with mutant-driven conversion of antiandrogen from AR antagonist to AR agonist [13]. Currently, several PROTACs for AR are in clinical trials for the therapy of drug-resistant prostate cancer.
3.2 PROTAC in Breast Cancer
About 70% of breast cancer patients present with estrogen receptor (ER) positive, making ER an important target in breast cancer drug research. However, most primary patients acquire resistance to estrogen antibody-based drugs (e.g., Fulvestrant). In contrast, PROTAC using ER induces effective degradation and can improve its pharmacological properties [14].
In addition to ER, there are a number of targets associated with breast cancer, such as CDK4/6, HER2, and PARP1, and TACC3, BET family proteins (BRD2, BRD3, BRD4 and BRDT), etc. Researchers have designed and synthesized the corresponding PROTAC molecules, which have degradation effects on target proteins at the cellular level [15-17]. These studies suggest that PROTAC-targeted protein degradation is expected to provide a new strategy for the treatment of breast cancer.
3.3 PROTAC in Non-Small Cell Lung Cancer
EGFR, a receptor tyrosine kinase (RTK), is closely related to the regulation of apoptosis, proliferation, metabolism, and survival. EGFR is mutated in 10%-15% of NSCLC patients, and EGFR inhibitors are a hot spot for drug development. However, many patients develop drug resistance, which affects the subsequent treatment effect. Currently, many research teams are also conducting research on targeted degradation of EGFR using PROTAC technology [18]. In addition, as a new target for the treatment of NSCLC, KRAS plays an important role in cell survival and cycle progression. In 2020, Crews' team reported the first PROTAC molecule that degrades KRAS G12C, an important breakthrough in the field of PROTAC technology targeting non-druggable proteins. Moreover, about 4% of NSCLC is accompanied by ALK fusion genes, ALK targeting PROTAC is expected to find a new direction for patients with ALK-resistant mutations [19, 20].
3.4 PROTAC in Chronic Myeloid Leukemia
The advent of BCR-ABL kinase inhibitors has transformed the lives of patients with chronic granulocytic leukemia (CML); however, kinase inhibition does not provide complete cure in most patients. The development of PROTAC, which induces BCR-ABL degradation, has been reported to exhibit significant selectivity and significantly inhibit the growth of BCR-ABL+leukemic cells, thereby inhibiting the BCR-ABL-mediated signaling pathway [21]. Now, more novel PROTAC target protein degraders for CML are in the clinical stage.
3.5 ROTAC in Other Diseases
In addition to targeting degradation of key proteins in tumors, another challenging is aggregation-prone proteins. α-Synuclein/SNCA and tau proteins are closely associated with the pathogenesis and dysfunction of neurodegenerative diseases. Degradation of these proteins by direct recruitment of PROTAC to E3 ligases is a viable approach to treat this class of diseases as well as protein aggregation diseases. Indeed, several studies of PROTAC have been explored to treat Amosheimer's disease by inducing tau degradation [22-24].
4. The Clinical Perspectives of PROTACs
PROTAC technology is an emerging protein degradation technology. PROTAC boasts overwhelming superiority over small molecule inhibitors, such as targeting non-druggable targets and overcoming drug resistance. As researchers learn more about the UPS, PROTAC (eg., more identified protein target ligands and E3 ligase ligands), this will further accelerate the use of PROTAC in drug discovery. As of now, PROTAC has been intensively studied in the treatment of tumors, viral infections, and neurodegenerative diseases. A host of pharmaceutical companies puts more strategies on PROTACs to have successful clinical trials, including Roche, Biogen, GSK, Sanofi and others. It is believed that PROTAC molecular drugs will be a promising technology for cancer drug discovery (Table 1).
|
Drugs
|
Targets
|
Mechanism
|
Drug Type
|
Indications
|
Institutes
|
R&D status
|
|
Bavdegalutamide
|
AR
|
AR degradation enhancer
|
PROTACs
|
Prostate Cancer
|
Arvinas, Inc.
|
Clinical Phase 2
|
|
ARV-471
|
ESR1
|
ERα antagonists
|
PROTACs
|
Breast Cancer
|
Pfizer Inc.; Arvinas, Inc.; Pfizer Investment Co.
|
Clinical Phase 2
|
|
CFT 8634
|
BRD9
|
BRD9 inhibitor
|
PROTACs
|
Solid tumor; synovial sarcoma
|
C4 Therapeutics, Inc.
|
Clinical Phase 1/2
|
|
KT-474
|
IRAK1
|
IRAK1 antagonist
|
PROTACs
|
Atopic dermatitis; rheumatoid arthritis; septic sweat glanditis; multiple sclerosis
|
Kymera Therapeutics, Inc.; Sanofi
|
Clinical Phase 1
|
|
NX-2127
|
BTK | IKZF3
|
BTK inhibitor; IKZF3 inhibitor
|
PROTACs
|
B-cell chronic lymphocytic leukemia; diffuse large B-cell lymphoma; follicular lymphoma; set cell lymphoma; marginal zone B-cell lymphoma; non-Hodgkin's lymphoma; macroglobulinemia
|
Nurix Therapeutics, Inc.
|
Clinical Phase 1
|
|
KT-413
|
IRAK4
|
IRAK4 inhibitor
|
PROTACs
|
B-cell lymphoma; diffuse large B-cell lymphoma; lymphoma
|
Kymera Therapeutics, Inc.
|
Clinical Phase 1
|
|
DT-2216
|
Bcl/X
|
Bcl/X inhibitors
|
PROTACs
|
Hematologic tumors; solid tumors
|
Dialectic Therapeutics, Inc.
|
Clinical Phase 1
|
|
HSK-29116
|
BTK
|
BTK inhibitor
|
PROTACs
|
B-cell lymphoma
|
Haseko Pharmaceutical Group Co.
|
Clinical Phase 1
|
|
NX 5948
|
BTK
|
BTK inhibitor
|
PROTACs; small molecule chemotherapeutics
|
B-cell chronic lymphocytic leukemia; diffuse large B-cell lymphoma; follicular lymphoma; set cell lymphoma; marginal zone B-cell lymphoma; primary central nervous system lymphoma; macroglobulinemia; graft-versus-host disease; rheumatoid arthritis
|
Nurix Therapeutics, Inc.
|
Clinical Phase 1
|
|
LNK-01002
|
Ras
|
Ras inhibitor
|
PROTACs
|
Acute myelogenous leukemia; primary myelofibrosis; hematologic neoplasms
|
Linke Pharmaceutical (Hangzhou) Co.
|
Clinical Phase 1
|
|
BGB 16673
|
BTK
|
Protein degradation; BTK inhibitor
|
PROTACs
|
Non-Hodgkin's lymphoma; B-cell chronic lymphocytic leukemia; B-cell lymphoma
|
Baekje Shenzhou (Suzhou) Biotechnology Co.
|
Clinical Phase 1
|
|
GT-20029
|
AR
|
AR degradation enhancer
|
PROTACs
|
Acne vulgaris; hair loss
|
Suzhou Development Pharmaceutical Co.
|
Clinical Phase 1
|
|
FHD 609
|
BET
|
BET Inhibitors
|
PROTACs
|
Synovial sarcoma
|
Foghorn Therapeutics, Inc.
|
Clinical Phase 1
|
|
AC 0682
|
ESR1
|
ERα antagonists
|
PROTACs
|
ER-positive breast cancer; breast cancer
|
Ice Island Stone Biotechnology (Shanghai) Co.
|
Clinical Phase 1
|
|
HP 518
|
AR
|
AR antagonists
|
PROTACs
|
Prostate Cancer
|
Haichuang Pharmaceutical Co.
|
Clinical Phase 1
|
|
AC 0176
|
AR
|
AR degradation enhancer
|
PROTACs
|
Destructive resistant prostate cancer; prostate cancer
|
Ice Island Stone Biotechnology (Shanghai) Co.
|
Clinical Phase 1
|
|
ARV-766
|
AR
|
AR degradation enhancer
|
PROTACs
|
Prostate cancer; prostate disease
|
Arvinas, Inc.
|
Clinical Phase 1
|
|
KT-333
|
STAT3
|
STAT3 inhibitor
|
PROTACs
|
Hematologic tumors; solid tumors
|
Kymera Therapeutics, Inc.
|
Clinical Phase 1
|
|
CG 001419
|
NTRK
|
Protein degradation; NTRK inhibitor
|
PROTACs
|
Solid tumors; tumors
|
Cullgen, Inc.; Shanghai Ruiyu Biotechnology Co.
|
Clinical Phase 1
|
|
CBP-8088
|
/
|
/
|
PROTACs
|
Tumors
|
Coherent Biopharma Suzhou Co. Ltd.
|
Clinical Applications
|
|
NW-8-153
|
/
|
Protein degradation
|
PROTACs
|
Tumors
|
Guangzhou Lu Peng Pharmaceutical Co.
|
Preclinical
|
|
HPB-001
|
/
|
Protein degradation
|
PROTACs
|
Tumors
|
Hangzhou Multi-Domain Biotechnology Co.
|
Preclinical
|
|
Homo-PROTAC pVHL30 degrader 1 (University of Dundee)
|
/
|
Protein degradation
|
PROTACs
|
/
|
University Of Dundee
|
Preclinical
|
|
E3 ubiquitin ligase based protein degradation (GSK/Kymera)
|
/
|
Protein degradation
|
PROTACs
|
Autoimmune diseases; tumors
|
Kymera Therapeutics, Inc.; GSK Plc
|
Preclinical
|
|
ASN-1764
|
/
|
Protein degradation
|
Molecular gels; PROTACs
|
Ovarian cancer; triple-negative breast cancer
|
Yahong Pharmaceutical Technology Co.
|
Preclinical
|
|
CBP-8028
|
/
|
/
|
PROTACs
|
/
|
Coherent Biopharma Suzhou Co. Ltd.
|
Preclinical
|
|
MTX-23
|
/
|
Protein degradation
|
PROTACs
|
Destructive resistant prostate cancer
|
Montelino Therapeutics, LLC
|
Preclinical
|
|
CT-03
|
/
|
/
|
PROTACs
|
Hematologic tumors; solid tumors
|
Captor Therapeutics SA
|
Preclinical
|
|
NW-7-251
|
/
|
Protein degradation
|
PROTACs
|
Hematologic Tumors
|
Guangzhou Lu Peng Pharmaceutical Co.
|
Preclinical
|
|
Alhena Project
|
/
|
/
|
PROTACs
|
Tumors
|
-
|
Preclinical
|
|
Oncology program (Arvinas)
|
/
|
/
|
PROTACs
|
Solid tumors
|
Arvinas, Inc.
|
Preclinical
|
|
ASN-1780
|
/
|
Protein degradation
|
PROTACs; molecular glues
|
Destructive resistant prostate cancer
|
Yahong Pharmaceutical Technology Co.
|
Preclinical
|
|
HPB-002
|
/
|
Protein degradation
|
PROTACs
|
Autoimmune diseases
|
Hangzhou Multi-Domain Biotechnology Co.
|
Preclinical
|
|
CBP-8008
|
/
|
/
|
PROTACs
|
/
|
Coherent Biopharma Suzhou Co. Ltd.
|
Preclinical
|
|
CBP-8018
|
/
|
/
|
PROTACs
|
/
|
Coherent Biopharma Suzhou Co. Ltd.
|
Preclinical
|
|
gw3965-peg5-vh032
|
/
|
Protein degradation
|
PROTACs
|
Inflammation
|
National Institutes of Health
|
Preclinical
|
|
PG21
|
GSK3B
|
GSK3B inhibitor
|
PROTACs
|
Neurological degenerative diseases
|
China University of Pharmaceutical Sciences
|
Preclinical
|
|
MYCN Degarders (Nurix Therapeutics)
|
MYCN
|
Protein degradation
|
PROTACs
|
Neuroblastoma
|
Nurix Therapeutics, Inc.
|
Preclinical
|
|
SGK3-PROTAC1
|
SGK3
|
Protein degradation; SGK3 inhibitor
|
PROTACs
|
Tumors
|
University Of Dundee
|
Preclinical
|
|
CP5V
|
CDC20
|
Protein degradation; CDC20 inhibitor
|
PROTACs
|
Tumors
|
Northwestern University
|
Preclinical
|
|
SMARCA2 inhibitors (Arvinas/Genentech)
|
SMARCA2
|
SMARCA2 inhibitor
|
PROTACs
|
Non-small cell lung cancer
|
Arvinas, Inc.; Genentech, Inc.
|
Preclinical
|
|
PROTAC CRABP-II Degrader-1 (Mercachem BV)
|
CRABP2
|
Protein degradation; CRABP2 inhibitor
|
PROTACs
|
Tumors
|
Mercachem BV
|
Preclinical
|
|
MD13
|
MIF
|
Protein degradation; MIF inhibitor
|
PROTACs
|
Lung Cancer
|
University of Groningen
|
Preclinical
|
|
dTRIM24 (Dana-Farber Cancer Institute)
|
TRIM24
|
Protein degradation
|
PROTACs
|
Tumors
|
Harvard Medical School; Dana-Farber Cancer Institute, Inc.
|
Preclinical
|
|
BI-3663
|
PTK2
|
FAK1 inhibitor
|
PROTACs
|
Tumors
|
Boehringer Ingelheim GmbH
|
Preclinical
|
|
SJFδ
|
MAPK13
|
Protein degradation; MAPK13 inhibitor
|
PROTACs
|
Tumors
|
Yale University; Arvinas, Inc.
|
Preclinical
|
|
ORM 5029
|
GSPT1
|
GSPT1 inhibitor
|
PROTACs
|
Breast Cancer
|
Orum Therapeutics, Inc.
|
Preclinical
|
|
VZ185
|
BRD7 | BRD9
|
BRD7 inhibitor; BRD9 inhibitor
|
PROTACs
|
Tumors
|
University Of Dundee
|
Preclinical
|
|
SIAIS164018
|
EGFR | ALK
|
EGFR antagonists; ALK inhibitors
|
PROTACs
|
Non-small cell lung cancer
|
Shanghai University of Science and Technology
|
Preclinical
|
|
VZ-185
|
BRD7 | BRD9
|
BRD7 inhibitor; BRD9 inhibitor
|
PROTACs
|
Tumors
|
Boehringer Ingelheim International GmbH
|
Preclinical
|
Partial data from PharmSnap
5. Popular Target Proteins and E3 Ubiquitin Ligases Involved in PROTAC Research
Targeting protein degradation with Proteolysis-Targeting Chimera (PROTAC) is the most popular technology in the current drug discovery. Here, CUSABIO summarizes popular target proteins and E3 ubiquitin ligases in PROTAC research to help you explore the functions of PROTAC for specific targets or its potential clinical value!
● Targeted protein products
● E3 ubiquitin ligase related protein products
References
[1] Sakamoto, Kathleen M., et al. "Protacs: chimeric molecules that target proteins to the Skp1-Cullin-F box complex for ubiquitination and degradation." Proceedings of the National Academy of Sciences 98.15 (2001): 8554-8559.
[2] Schneekloth, Ashley R., et al. "Targeted intracellular protein degradation induced by a small molecule: En route to chemical proteomics." Bioorganic & amp; medicinal chemistry letters 18.22 (2008): 5904-5908.
[3] Han, Xin, Wenyi Wei, and Yi Sun. "PROTAC degraders with ligands recruiting MDM2 E3 ubiquitin ligase: an updated perspective." Acta Materia Medica (2022).
[4] Cecchini, Carlotta, et al. "Exploring the ubiquitin-proteasome system (UPS) through PROTAC technology." Chimia 74.4 (2020): 274-274.
[5] Li, Xinyi, et al. "Therapeutic targeting of RNA-binding protein by RNA-PROTAC." Molecular Therapy 29.6 (2021): 1940-1942.
[6] Bondeson, Daniel P., et al. "Lessons in PROTAC design from selective degradation with a promiscuous warhead." cell chemical biology 25.1 (2018): 78-87.
[7] Pagan, Julia, et al. "Role of the ubiquitin proteasome system in the heart." Circulation research 112.7 (2013): 1046-1058.
[8] Nalawansha, Dhanusha A., and Craig M. Crews. "PROTACs: an emerging therapeutic modality in precision medicine. "Cell chemical biology 27.8 (2020): 998 -1014.
[9] Gao, Hongying, Xiuyun Sun, and Yu Rao. "PROTAC technology: opportunities and challenges." acs medicinal chemistry letters 11.3 (2020): 237-240.
[10] Paiva, Stacey-Lynn, and Craig M. Crews. "Targeted protein degradation: elements of PROTAC design. "Current opinion in chemical biology 50 (2019): 111- 119.
[11] Kargbo, Robert B. "PROTAC degradation of IRAK4 for the treatment of cancer." ACS Medicinal Chemistry Letters 10.10 (2019): 1370-1371.
[12] Wang, Chao, et al. "PROTACs technology for targeting non-oncoproteins: Advances and perspectives." Bioorganic Chemistry 114 (2021): 105109.
[13] Petrylak, Daniel Peter, et al. "First-in-human phase I study of ARV-110, an androgen receptor (AR) PROTAC degrader in patients (pts) with metastatic castrate-resistant prostate cancer (mCRPC) following enzalutamide (ENZ) and/or abiraterone (ABI)." (2020): 3500-3500.
[14] Hu, Jiantao, et al. "Discovery of ERD-308 as a highly potent proteolysis targeting chimera (PROTAC) degrader of estrogen receptor (ER)." Journal of medicinal chemistry 62.3 (2019): 1420-1442.
[15] Noblejas-López, María del Mar, et al. "Antitumoral Activity of a CDK9 PROTAC Compound in HER2-Positive Breast Cancer." International Journal of Molecular Sciences 23.10 (2022): 5476.
[16] Yang, Tingting, et al. "A BRD4 PROTAC nanodrug for glioma therapy via the intervention of tumor cells proliferation, apoptosis and M2 macrophages polarization." Acta Pharmaceutica Sinica B (2022).
[17] Wang, Yang, et al. "Degradation of proteins by PROTACs and other strategies." Acta Pharmaceutica Sinica B 10.2 (2020): 207-238.
[18] Zhang, Hao, et al. "Discovery of potent epidermal growth factor receptor (EGFR) degraders by proteolysis targeting chimera (PROTAC)." European Journal of Medicinal Chemistry 189 (2020): 112061.
[19] Zhang, Z., et al. "28P Discovery of potent PROTAC degraders of KRASG12C based on a reversible non-covalent KRAS binder." Annals of Oncology 33 (2022): S14.
[20] Yan, Guoyi, et al. "Discovery of a PROTAC targeting ALK with in vivo activity." European Journal of Medicinal Chemistry 212 (2021): 113150.
[21] Pan, You-lu, et al. "The progress of small-molecules and degraders against BCR-ABL for the treatment of CML." European Journal of Medicinal Chemistry (2022): 114442.
[22] Kargbo, Robert B. "Treatment of Alzheimer's by PROTAC-Tau protein degradation." ACS Medicinal Chemistry Letters 10.5 (2019): 699-700.
[23] Wang, Weijin, et al. "A novel small-molecule PROTAC selectively promotes tau clearance to improve cognitive functions in Alzheimer-like models." Theranostics 11.11 (2021): 5279.
[24] Pradeepkiran, Jangampalli Adi, and P. Hemachandra Reddy. "Phosphorylated tau targeted small-molecule PROTACs for the treatment of Alzheimer's disease and tauopathies." Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1867.8 (2021): 166162.
[25] Zhou, Xin, et al. "PROTAC: A promising technology for cancer treatment." European journal of medicinal chemistry 203 (2020): 112539.
[26] Békés, Miklós, David R. Langley, and Craig M. Crews. "PROTAC targeted protein degraders: the past is prologue." Nature Reviews Drug Discovery 21.3 (2022): 181-200.
[27] Zou, Yutian, Danhui Ma, and Yinyin Wang. "The PROTAC technology in drug development." Cell biochemistry and function 37.1 (2019): 21-30.
CUSABIO team. Proteolysis Targeting Chimera (PROTAC): A "Revolutionary" Technology in Small Molecule Drug Discovery!. https://www.cusabio.com/c-21078.html



Comments
Leave a Comment