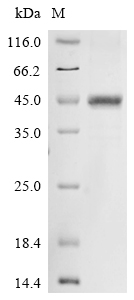
The formation of the recombinant plasmid by co-incorporating the gene encoding the human Norrin (NDP) (25-133aa) with the N-terminal 10xHis-GST-tag gene and C-terminal Myc-tag gene into a plasmid vector initiates the generation of the recombinant human NDP protein. Transforming the recombinant plasmid into E.coli cells and then selecting the positive E.coli cells based on their ability to survive in the presence of a specific antibiotic. The positive E.coli cells are cultured under conditions that stimulate the expression of the gene of interest. After expression, affinity purification is employed to isolate and purify the recombinant human NDP protein from the cell lysate. Denaturing SDS-PAGE is utilized to resolve the resulting recombinant human NDP protein, revealing a purity level exceeding 85%.
NDP is a multifunctional protein involved in various cellular processes. Initially considered a housekeeping enzyme essential for DNA and RNA synthesis, recent studies have revealed its involvement in cellular regulatory functions such as growth control, developmental control, and tumor metastasis suppression [1]. NDP kinase is known for its catalytic activity of nucleotide phosphotransfer, but it also carries out other functions beyond its catalytic activity, indicating its multifunctionality [2]. Furthermore, it has been reported that NDP kinase may have multiple regulatory functions besides the phosphotransferase activity and behaves as a tumor metastasis suppressor [3]. Additionally, NDP kinase has been found to interact with vimentin intermediate filaments and energy metabolism enzymes in a tissue-specific manner, suggesting its tissue-specific roles [4]. Moreover, NDP kinase has been implicated in transmembrane signaling, indicating its involvement in diverse cellular processes [5]. Studies have also suggested that NDP kinases are multifunctional proteins and some of their functions operate via the interaction with G-proteins [6]. Furthermore, NDP kinase has been identified as a tumor metastasis suppressor, a morphological regulator, a transcription factor, and a differentiation inhibitor, indicating its involvement in different levels of cellular regulation [7]. Additionally, it has been reported that NDP kinases can participate in phosphorelay networks that regulate gene expression and metabolism [8]. These findings collectively demonstrate the multifunctional nature of NDP kinase and its involvement in various cellular processes beyond its traditional role in nucleotide metabolism.
References:
[1] Q. Lü, H. Park, L. Egger, & M. Inouye, Nucleoside-diphosphate kinase-mediated signal transduction via histidyl-aspartyl phosphorelay systems in escherichia coli, Journal of Biological Chemistry, vol. 271, no. 51, p. 32886-32893, 1996. https://doi.org/10.1074/jbc.271.51.32886
[2] S. Mesnildrey, F. Agou, A. Karlsson, D. Bonne, & M. Véron, Coupling between catalysis and oligomeric structure in nucleoside diphosphate kinase, Journal of Biological Chemistry, vol. 273, no. 8, p. 4436-4442, 1998. https://doi.org/10.1074/jbc.273.8.4436
[3] N. Ishikawa, H. Taniguchi-Seto, Y. Munakata, Y. Takagi, N. Shimada, & N. Kimura, Multiple transcripts for rat nucleoside diphosphate kinase α isoform are structurally categorized into two groups that exhibit cell-specific expression and distinct translation potential, Journal of Biological Chemistry, vol. 272, no. 6, p. 3289-3295, 1997. https://doi.org/10.1074/jbc.272.6.3289
[4] A. Otero, Copurification of vimentin, energy metabolism enzymes, and a mer5 homolog with nucleoside diphosphate kinase, Journal of Biological Chemistry, vol. 272, no. 23, p. 14690-14694, 1997. https://doi.org/10.1074/jbc.272.23.14690
[5] L. Xu, J. Murphy, & A. Otero, Participation of nucleoside-diphosphate kinase in muscarinic k+ channel activation does not involve gtp formation, Journal of Biological Chemistry, vol. 271, no. 35, p. 21120-21125, 1996. https://doi.org/10.1074/jbc.271.35.21120
[6] N. Orlov, T. Orlova, Y. Reshetnyak, E. Burstein, & N. Kimura, Interaction of recombinant rat nucleoside diphosphate kinase α with bleached bovine retinal rod outer segment membranes: a possible mode of ph and salt effects, Iubmb Life, vol. 41, no. 1, p. 189-198, 1997. https://doi.org/10.1080/15216549700201191
[7] N. Orlov, T. Orlova, K. Nomura, N. Hanai, & N. Kimura, Transducin‐mediated, isoform‐specific interaction of recombinant rat nucleoside diphosphate kinases with bleached bovine retinal rod outer segment membranes, Febs Letters, vol. 389, no. 2, p. 186-190, 1996. https://doi.org/10.1016/0014-5793(96)00575-3
[8] M. Levit, B. Abramczyk, J. Stock, & E. Postel, Interactions between escherichia colinucleoside-diphosphate kinase and dna, Journal of Biological Chemistry, vol. 277, no. 7, p. 5163-5167, 2002. https://doi.org/10.1074/jbc.m111170200"