BioLineRx Ltd presently has completed a successful pre-New Drug Application (NDA) meeting regarding a chemokine receptor CXCR4 antagonist (Motixafortide), from a report on January 18, 2022. In particular, the biopharmaceutical company further announced that Motixafortide NDA submission will be on track for H1 2022 (January to June). It is expected to be the second CXCR4 antagonist FDA approved for cancer therapy after Plerixafor.
In the human body, the interaction of the chemokine receptor members and their ligands constitute a complex chemokine regulatory network with important implications for autoimmune or cancer diseases. As a representative member of GPCR, CXCR4 receptor plays a significant role in tumor-related processes, including migration, invasion, and metastasis of cells. By now, CXCR4 antagonists suggested great potential in clinical trials, which will open up exciting possibilities for future cancer research. The findings summarized in this article will be of interest to CXCR4 researchers.
1. What does CXCR4 Stand For?
The C X C chemokine receptor 4 (CXCR4) belongs to G protein-coupled receptor (GPCR) family with a conserved 7 transmembrane (7TM) protein structure, consisting of 352 amino acids, located on chromosome 2q21. At present, GPCRs superfamily provides many desirable therapeutic targets in cancer and other diseases (click here for "The Collection of Progress of Anti-GPCR Antibody Drugs") [1-2]. Where is CXCR4 found? CXCR4 is expressed by most cells, including hematopoietic and endothelial cells (ECs), neurons and stem cells (embryonic and adult).
Recent studies have revealed that the metastasis of tumor cells to specific organs is the result of chemokines and their receptors function in chemotaxis of metastatic cells. What is the function of CXCR4? Among the chemokine receptors, CXCR4 has been discovered strongly expressed in multiple types of cancers and take effects on angiogenesis, cell metastasis, growth or survival [3]. In addition, CXCR4 is also involved in HIV virus infection [4]. These biological functions supports that CXCR4 could be potentially an ideal target for tumors, hematopoietic stem cell mobilization, HIV and other diseases.
2. What is the CXCR4 Ligand?
The chemokine CXCL12 (also known as SDF-1, stromal cell-derived factor 1) is the only ligand for CXCR4, a member of the CXC chemokine family with a molecular weight of approximately 8 kD. CXCL12 contains two isoforms, α and β, and is secreted by bone marrow stromal cells and other related mesothelial and epithelial cells [5]. The CXCR4/CXCL12 axis is involved in many biological processes, including (i) multiple mechanisms of tumor metastasis, proliferation, adhesion, and vascularization; (ii) mediating mesenchymal cell chemotaxis and organ-specific homing in injured tissue, expanding paracrine secretion, and promoting vascularization; (iii) regulating the movement of mature B cells, plasma cells, neutrophils, monocytes, T cells, and dendritic cells, which is closely related to the immune function of the body [3, 6]. All in all, CXCR4/ CXCL12 plays key roles in many physiological and pathological processes, including embryogenesis, wound healing processes, angiogenesis, in the development and metastasis of tumors and during HIV-1 infection.
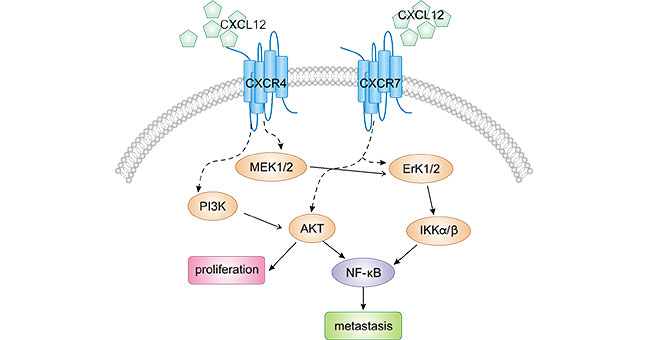
3. How's the Mechanism of CXCR4 in Tumors?
CXCR4 specifically binds to its ligand CXCL12 to activate a series of downstream signaling pathways, which in turn regulate biological behaviors such as cell survival, proliferation, migration and adhesion in tumors. As presented in Figure 1, CXCR4-CXCL12 is involved in activating a variety of pro-oncogenic pathways that promote cell proliferation, inhibit apoptosis, and promote metastasis, including signaling pathways RAS-MAPK-MEK1/2 [7], ERK1/2 [8], PI3K-AKT [9] and NF-kB [10].
For example, CXCR4-CXCL12 promotes the growth, infiltration, and metastasis of gastric cancer cells via PI3K-AKT. In details, the highly expressed CXCL12-CXCR4 activates PI3Kγ. by binding to phosphatidylinositol 3-kinaseγ (phos-photidylionsitol 3-kinasseγ (PI3Kγ), which in turn activates downstream effectors of PI3Kγ, such as nuclear factor κB (NF-κB), protein kinase B (PKB), extracellular regulated protein kinase 1/2 (ERK1/2), and mitogen-activated protein kinases (MAPK) [11, 12].
Figure1: CXCR4/CXCL12 participates multiple signaling pathways in tumor cells
4. What's the Function of CXCR4 in Tumors and Other Diseases?
CXCR4, as the most commonly expressed chemokine receptor in tumor cells, which has been reported in many tumors, including breast cancer [13] ovarian cancer [14] lung cancer [15] colorectal cancer [16] glioma [17] pancreatic cancer [18] prostate cancer [19] multiple myeloma (MM) [20] acute myeloid leukemia (AML) [21] chronic lymphocytic leukemia (CLL) [22] non-Hodgkin's lymphoma (NHL) [23]etc.
In hematological tumors, CXCR4 has undisputed clinical relevance. The evidence is overwhelming. The first CXCR4 antagonist, Plerixafor was approved in 2008, which is available in China. Currently, Plerixafor can be used in combination with G-CSF for hematopoietic stem cell mobilization in NHL and MM patients undergoing autologous hematopoietic stem cell transplantation (ASCT). CXCR4 inhibitors are also being investigated. For example, a phase Ib/II trial (NCT01359657) of ulocuplumab (BMS-936564), an anti-CXCR4 antibody, identified that blocking CXCR4-CXCL12 in combination with lenalidomide and dexamethasone has a high response rate in patients with relapsed or refractory myeloma; a humanized CXCR4 antibody PF-06747143 exhibited strong antitumor effects in a variety of hematologic tumor models, including NHL, AML, and MM.
In solid tumors, CXCR4 inhibitors also indicated an important anticancer potential. Several clinical trials supports the benefit of CXCR4 inhibitors in patients with glioma. Besides, a phase I/II trial (NCT01977677) suggested that Plerixafor inhibited CXCL4-mediated angiogenesis and enhanced the effect of radiation therapy. Beyond this, a CXCR4 peptide antagonist (LY2510924) showed antitumor potent in various solid tumors and preclinical models of metastatic breast cancer.
It is not just in tumors that CXCR4 play roles, also in several immune diseases (e.g., systemic lupus erythematosus [24] rheumatic-related diseases [25]etc.), the genetic disease WHIM syndrome [26] and the virus disease HIV [27] Furthermore, it has been reported that CXCR4 mediates axonal regeneration of damaged optic nerves during embryonic development, which may provide a new strategy for vision therapy [28] In a nutshell, CXCR4 provides a perspective on strategies that can lead to future drug discoveries for a variety of diseases, especially in tumors.
5. How's the Clinical Prospects of CXCR4?
Presently, studies on CXCR4 antagonist drugs are ongoing or relatively well understood. In addition to the only marked Plerixafor for hematologic malignancies, more CXCR4 inhibitors are emerging as potential therapeutic agents (As listed below). For instance, Motixafortide is in clinical phase III for breast cancer; Mavorixafor is also in clinical phase III, acts on autologous transplant patients with multiple myeloma. Noticeably, Mavorixafor is scheduled to submit an NDA first half of 2022 and is very likely to become the second CXCR4 drug worldwide in near future. Nevertheless, CXCR4 consider as the potent drug targets of GPCR, it is forecasted to provide new breakthroughs in many diseases, particularly in cancer.
|
Drug name
|
R&D status
|
Company
|
Mechanism
|
Indications
|
|
Plerixafor (Plerixafor)
|
2008, FDA approved for marketing
2018, NMPA approved for marketing
|
Genzan USA; Genzyme Europe Bv; Magenta Therapeutics Inc; Sanofi
|
CXCR4 antagonists
|
Multiple myeloma; non-Hodgkin's lymphoma; hematopoietic stem cell transplantation; lymphoma; myelodysplastic syndrome acute lymphoblastic leukemia acute myeloid leukemia; Hodgkin's disease chronic lymphocytic leukemia; chronic myeloid leukemia; brain arrest; neuroblastoma; Ewing's sarcoma: organ transplant rejection; gastric disorders
|
|
Mavorixafor
|
Clinical Phase III
|
Shanghai Hechi Biomedical Technology Co Ltd:Abbisko Therapeutics CoLtd;Mayne Pharma Inc;X4 Pharmaceuticals
|
CXCR4 antagonists
|
WHIM syndrome; triple negative breast tumor: renal cell carcinoma; neutropenia; melanoma with Walden's macroglobulinemia; HIV infection
|
|
Balixafortide
|
Clinical Phase III
|
Polyphor Ltd
|
CXCR4 antagonists
|
Breast de; multiple myeloma; myeloproliferative disorders; myelodysplastic syndrome; acute lymphoblastic leukemia; acute myeloid leukemia; chronic lymphocytic leukemia; chronic follower cell leukemia
|
|
NRP-2945
|
Clinical Phase II
|
Neuren Pharmaceuticals
|
CXCR4 regulator
|
Epilepsy
|
|
[68Ga]Pentixafor
|
Clinical Phase II
|
PentixaPharm GmbH; The University Of lowa
|
CXCR4 antagonists
|
Central nervous system tumors; neuroendocrine tumors: diagnostic agents
|
|
PTX-9908
|
Clinical Phase II
|
Beijing Meikangstead Pharmaceutical Technology Co., Ltd; Pertinax Therapeutics; Taizong Biotechnology Co.
|
CXCR4 regulator
|
Liver cancer; hepatocellular carcinoma
|
|
MSX-122
|
Clinical Phase II
|
Altiris Therapeutics:Que Oncology
|
CXCR4 antagonists
|
Vasodystrophy; hot flushes: entity
|
|
Ulocuplumab
|
Clinical Phase II
|
Bristol-Myers Squibb
|
CXCR4 antagonists
|
Small cell lung cancer; pancreatic cancer; multiple myeloma; acute myeloid leukemia: solid tumors of leukemia; lymphocytic leukemia; follicular center lymphatic lodging: chronic lymphocytic leukemia
|
|
Motixafortide
|
Clinical Phase II
|
Biolinerx Ltd; F. Hoffmann-La Roche Ag
|
CXCR4 antagonists
|
Non-small cell lung cancer; esophageal cancer: pancreatic cancer; abnormal bone growth syndrome: aplastic anemia; acute cell-like leukemia; multiple bone follower tumors
|
|
JVS-100
|
Clinical Phase II
|
Cleveland Clinic;Juventas Therapeutics;Sironrx Therapeutics
|
Leukocyte stimulator; CXCR4 stimulator; gene transfer; apoptosis inhibitor; angiogenesis inducer
|
Ischemia; peripheral arterial disease; heart failure
|
|
LY-2510924
|
Clinical Phase II
|
Eli Lilly and Company
|
CXCR4 antagonists
|
Renal cell carcinoma small cell lung cancer; solid tumors
|
|
AD-214
|
Clinical Phase I
|
AdaltaLtd;Addpharma Inc
|
CXCR4 antagonists
|
Interstitial lung disease; gastroesophageal reflux disease
|
|
CXCR4 modified anti-BCMA CAR T celltherapy (SichuanUniversity)
|
Clinical Phase I
|
Sichuan University
|
Anti-BCMA (Anti-BCMA)
|
Multiple myeloma
|
|
Anti-CD4CART-celltherapy (University of Pennsylvania)
|
Clinical Phase I
|
University of Pennsylvania
|
CD4 modulator
|
HIV infection
|
|
GMI-1359
|
Clinical Phase I
|
Glycomimetics Inc
|
CXCR4 antagonists
|
Breast cancer; metastatic breast cancer
|
|
MLB-1707
|
Clinical Applications
|
Mainstream Source Biotechnology (Shanghai) Co.
|
CXCR4 antiagent
|
Triple negative breast tumor
|
|
MLB-010
|
Preclinical
|
Mainstream Source Biotechnology (Shanghai) Co.
|
CXCR4 antagonists
|
Tumors
|
|
MLB-014
|
Preclinical
|
Mainstream Source Biotechnology (Shanghai) Co.
|
CXCR4 antagonists
|
Leukemia
|
|
MLB-1807
|
Preclinical
|
Mainstream Source Biotechnology (Shanghai) Co.
|
CXCR4 antagonists
|
Tumors
|
|
X4P-002
|
Preclinical
|
X4 Pharmaceuticals
|
CXCR4 antagonists
|
Glioblastoma
|
|
GP-01CR11
|
Preclinical
|
GPCR Therapeutics USA Inc
|
CXCR4 antagonists
|
Tumors
|
|
GP-01CR21
|
Preclinical
|
GPCR Therapeutics USA Inc
|
CXCR4 antagonists
|
Tumors
|
Data from pharmacodia
To fully serve pharmaceutical companies in their clinical studies of CXCR4 in tumors, hematopoietic stem cell mobilization, HIV, autoimmune diseases, etc., CUSABIO has launched CXCR4 active protein product (Code: CSB-MP006254HU(F1)) to help your research on CXCR4 mechanism or its potential clinical applications.
References
[1] Wescott, Melanie P., et al. "Signal transmission through the CXC chemokine receptor 4 (CXCR4) transmembrane helices." Proceedings of the National Academy of Sciences 113.35 (2016): 9928-9933.
[2] Van den Bosch, Thierry, et al. "Chemokine receptor CCR7 expression predicts poor outcome in uveal melanoma and relates to liver metastasis whereas expression of CXCR4 is not of clinical relevance." Investigative ophthalmology & visual science 54.12 (2013): 7354-7361.
[3] Chatterjee, Samit, Babak Behnam Azad, and Sridhar Nimmagadda. "The intricate role of CXCR4 in cancer. "Advances in cancer research 124 (2014): 31-82.
[4] Hernandez, Paolo A., et al. "Mutations in the chemokine receptor gene CXCR4 are associated with WHIM syndrome, a combined immunodeficiency disease." Nature genetics 34.1 (2003): 70-74.
[5] De Paepe, Boel, et al. "Localization of the α-chemokine SDF-1 and its receptor CXCR4 in idiopathic inflammatory myopathies. "Neuromuscular disorders 14.4 (2004): 265-273.
[6] Asri, Amir, et al. "Homing in hematopoietic stem cells: focus on regulatory role of CXCR7 on SDF1a/CXCR4 axis." Excli Journal 15 (2016): 134.
[7] Dushyanthen, Sathana, et al. "Agonist immunotherapy restores T cell function following MEK inhibition improving efficacy in breast cancer." nature communications 8.1 (2017): 1-18.
[8] Chetram, Mahandranauth A., Valerie Odero-Marah, and Cimona V. Hinton. "Loss of PTEN permits CXCR4-mediated tumorigenesis through ERK1/2 in prostate cancer cells." Molecular cancer research 9.1 (2011): 90-102.
[9] Zheng, Hao, et al. "Migration of endothelial progenitor cells mediated by stromal cell-derived factor-1α/CXCR4 via PI3K/Akt/eNOS signal transduction pathway." Journal of cardiovascular pharmacology 50.3 (2007): 274-280.
[10] Cheng, Y., et al. (2020). Positive Cross-Talk Between CXC Chemokine Receptor 4 (CXCR4) and Epidermal Growth Factor Receptor (EGFR) Promotes Gastric Cancer Metastasis via the Medical science monitor: international medical journal of experimental and clinical research, 26, e925019.
[11] Chen, Guang et al. "Inhibition of chemokine (CXC motif) ligand 12/chemokine (CXC motif) receptor 4 axis (CXCL12/CXCR4)-mediated cell migration by targeting mammalian target of rapamycin (mTOR) pathway in human gastric carcinoma cells." The Journal of biological chemistry vol. 287,15 (2012): 12132-41. doi:10.1074/jbc.M111.302299
[12] Qin, Yan et al. "Cancer-associated fibroblasts in gastric cancer affect malignant progression via the CXCL12-CXCR4 axis." Journal of Cancer vol. 12,10 3011-3023. 19 Mar. 2021, doi:10.7150/jca.49707
[13] Chen, Ivy X et al. "Blocking CXCR4 alleviates desmoplasia, increases T-lymphocyte infiltration, and improves immunotherapy in metastatic breast cancer." Proceedings of the National Academy of Sciences of the United States of America vol. 116,10 (2019): 4558-4566. doi:10.1073/pnas.1815515116
[14] Zeng, Yang et al. "Dual blockade of CXCL12-CXCR4 and PD-1-PD-L1 pathways prolongs survival of ovarian tumor-bearing mice by prevention of immunosuppression in the tumor microenvironment." FASEB journal : official publication of the Federation of American Societies for Experimental Biology vol. 33,5 (2019): 6596-6608. doi:10.1096/fj.201802067RR
[15] Wald, Ori. "CXCR4 Based Therapeutics for Non-Small Cell Lung Cancer (NSCLC)." Journal of clinical medicine vol. 7,10 303. 25 Sep. 2018, doi:10.3390/jcm7100303
[16] Wang, Dong et al. "Exosome-encapsulated miRNAs contribute to CXCL12/CXCR4-induced liver metastasis of colorectal cancer by enhancing M2 polarization of macrophages." Cancer letters vol. 474 (2020): 36-52. doi:10.1016/j.canlet.2020.01.005
[17] Jiang, Zheng et al. "Contribution of SDF-1α/CXCR4 signaling to brain development and glioma progression." Neuro-Signals vol. 21,3-4 (2013): 240-58. doi:10.1159/000339091
[18] Bockorny, Bruno et al. "BL-8040, a CXCR4 antagonist, in combination with pembrolizumab and chemotherapy for pancreatic cancer: the COMBAT trial." Nature medicine vol. 26,6 (2020): 878-885. doi:10.1038/s41591-020-0880-x
[19] Baci, Denisa et al. "Acetyl-L-Carnitine downregulates invasion (CXCR4/CXCL12, MMP-9) and angiogenesis (VEGF, CXCL8) pathways in prostate cancer cells: rationale for prevention and interception strategies." Journal of experimental & clinical cancer research : CR vol. 38,1 464. 12 Nov. 2019, doi:10.1186/s13046-019-1461-z
[20] Ullah, Tomalika Rahmat. "The role of CXCR4 in multiple myeloma: Cells' journey from bone marrow to beyond. "Journal of bone oncology vol. 17 100253. 16 Jul. 2019, doi:10.1016/j.jbo.2019.100253
[21] Cancilla, Daniel et al. "Targeting CXCR4 in AML and ALL." Frontiers in oncology vol. 10 1672. 4 Sep. 2020, doi:10.3389/ fonc.2020.01672
[22] El-Sherif, Wafaa T et al. "CXCR4 as a prognostic marker in Egyptian chronic lymphocytic leukemia patients." the Egyptian journal of immunology vol. 28,3 (2021): 114-126.
[23] Beider, Katia et al. "Targeting the CD20 and CXCR4 pathways in non-hodgkin lymphoma with rituximab and high-affinity CXCR4 antagonist BKT140." Clinical cancer research : an official journal of the American Association for Cancer Research vol. 19,13 (2013): 3495-507. doi: 10.1158/1078-0432.ccr-12-3015
[24] Ma, Kongyang et al. "TLR4+CXCR4+ plasma cells drive nephritis development in systemic lupus erythematosus." Annals of the rheumatic diseases vol. 77,10 (2018): 1498-1506. doi:10.1136/annrheumdis-2018-213615
[25] Peng, Liping et al. "Expression levels of CXCR4 and CXCL12 in patients with rheumatoid arthritis and its correlation with disease activity ." Experimental and therapeutic medicine vol. 20,3 (2020): 1925-1934. doi:10.3892/etm.2020.8950
[26] Kallikourdis, Marinos et al. "The CXCR4 mutations in WHIM syndrome impair the stability of the T-cell immunologic synapse." Blood vol. 122,5 (2013): 666-73. doi:10.1182/blood-2012-10-461830
[27] Zhang, Chaozai et al. "Discoveries and developments of CXCR4-targeted HIV-1 entry inhibitors." experimental biology and medicine (Maywood, N.J.) vol. 245,5 (2020): 477-485. doi:10.1177/1535370220901498
[28] Zanetti, Giulia et al. "A CXCR4 receptor agonist strongly stimulates axonal regeneration after damage." annals of clinical and translational neurology vol. 6,12 (2019): 2395-2402. doi:10.1002/acn3.50926
CUSABIO team. CXCR4: An Attractive Target of GPCR family Brings Promising New Drugs for Cancer Therapy!. https://www.cusabio.com/c-21062.html

-WB.jpg)
-AC1.jpg)


Comments
Leave a Comment