In analyzing the structure and function of recombinant proteins, the most important thing is to get pure recombinant proteins. Therefore, various methods have been explored to isolate high-purity recombinant proteins. Among these purification approaches, labeling recombinant proteins with affinity tagging is an indispensable tool to promote subsequent protein purification and detection. In addition, it may also have a favorable effect on the physicochemical properties of the target protein and even increase the production of recombinant proteins. At present, the most widely used affinity tag in high-throughput protein purification is His-tag.
This article will introduce the definition, advantages as a purification tag, purification mechanism of His-tag, as well as its applications.
1. What Is A His-tag?
The His-tag, also called polyhistidine tag, is one of the simplest and smallest protein tags. It is composed of 6 to 10 consecutive histidine residues in a string and can be placed on the C- or N-terminus of the target protein. It was originally developed for immobilized metal ion affinity chromatography (IMAC)-a rapid and efficient method used for protein purification in 1987 [1][2].
2. Why Is His-tag Popular as A Purification Tag?
Compared with other affinity tags, such as GST, Flag, and Strep, His-tag has distinct characteristics that makes it become the preferred tag for protein purification, as follows:
Small size and neutral charge at physiological pH values
His-Tag is very small, with a molecular weight of about 0.84 KD. His has an isoelectric point of 7.6 and almost no charge in the commonly used purification buffer system (pH 8.0). Therefore, His-tag has little effect on the structure of fusion protein after purification, which is helpful for downstream assays.
Do not change the characteristics of the target protein
His-tag has almost no effect on the characteristics of the target protein itself, and will not change the solubility and biological functions of the target protein. And His-tag can be incorporated at both the N- and C-terminus of a protein. Following affinity chromatography, proteins can be eluted under mild conditions, ensuring that the biological properties of the target proteins are unchanged.
Strong binding to metal ion-immobilized resin
His-tag can chelate with various metal ions, such as Ca2+, Mg2+, Ni2+, and Cu2+, forming stable chelates that are beneficial to protein purification. His-tags' high affinity for immobilized metal ions allows the recombinant proteins to be purified using affinity column chromatography. IMAC is the most commonly used purification method for the His-tagged fusion protein.
IMAC is simple in operation and is mild in purification conditions, and has little effect on the target protein. His-tag enables affinity purification using cheap and widely available nickel or cobalt resins, reducing experimental costs.
Facilitate protein expression
The N-terminal His-tag is compatible with the transcriptional and translational mechanism of bacteria, which is conducive to protein expression.
Preparation for His-tag antibody
The immunogenicity of the His-tag is relatively low, enabling the preparation for His-tag antibodies by directly immunizing the animal with the His-tag.
Construction of double affinity tag
A double affinity tag constructed by His-tag with other affinity tags can be applied to a variety of expression systems.
No removal of the His-tag after crystallization
It is not necessary to remove the His-tag after protein purification, and it will not affect the function of the protein.
The His-tag fusion protein can be purified in the presence of nonionic surfactants as well as under denaturing conditions. The former is usually used to purify highly hydrophobic target proteins, while the latter is usually used to purify proteins expressed as inclusion bodies.
Choosing to purify the target protein under native or denaturing conditions is dependent on protein location and solubility, the accessibility of the His-tag, and the desired downstream application.
Although His-tag has many advantages, it is not flawless. The His-tag has low specificity, and the purity of the His-tagged recombinant proteins is relatively low. And microbial hosts naturally express metal-binding proteins that may co-purify with the protein of interest. Some findings have found that His-tag possesses catalytic properties, which are indeed not negligible.
3. How to Add A His-tag to the Target Protein?
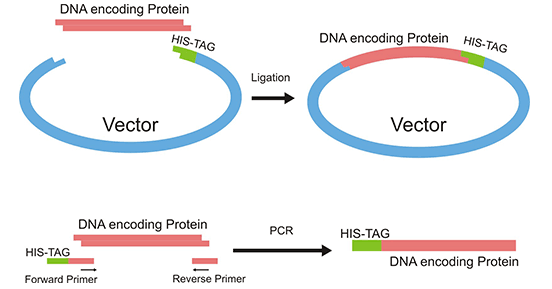
The His-tag is introduced by inserting the DNA fragment encoding a protein of interest in a vector that has the tag ready to fuse at the C-terminus. The forming recombinant plasmid is subsequently transferred or transformed into an expression system.
Express the tagged protein in bacterial, mammalian, or insect cells depending on the promoter employed. Alternatively, protein expression can be carried out using cell-free expression systems. The result is the expression of a recombinant protein with a 6*His or poly-His-tag fused to its C-terminus.
Alternatively, the His-tag is added using primers containing the tag, after a PCR reaction the tag gets fused to the N-terminus of the gene of interest.
Figure 1. Addition of His-tag to a protein
This picture is cited from Wikipedia
4. How Does His-tagged Protein Purification Work?
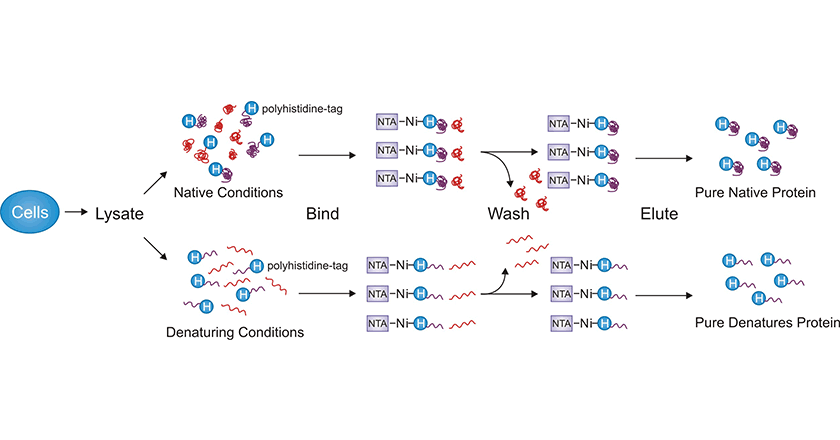
IMAC is frequently used to purify His-tagged proteins. IMAC is based on the interaction of histidine residues with divalent metal ions immobilized on resins. This purification method is quick, inexpensive, and straightforward, which has led to widespread adoption.
In the IMAC technique, transition metal ions like Ca2+, Mg2+, Ni2+, and Co2+ are immobilized on a resin matrix using a chelating agent such as nitrilotriacetic acid (NTA). Ni2+ is the most commonly used metal ion.
The histidine residue has an imidazole group, which can chelate with the transition metal ions. Therefore, proteins with His-tag can be selectively attached to the IMAC media at a neutral to slightly basic pH (pH 7.5-8), while other impurity proteins cannot bind or can only bind weakly [4]. Non-specifically bound proteins are washed away with an appropriate washing buffer, which should already contain a low molarity of imidazole.
It is worth noting that the binding affinity between those two interaction partners is proportional to the length of the His-tag. The binding affinity of 10*His-tags is ten times higher than that of 6*His-tags [4][5]. However, it is suggested to reduce the number of histidine residues as a first test to prevent potential protein function disruptions [6].
The His-tagged proteins bound to the media can be eluted by lowering the pH to 4–6, stripping the metal from the polymeric support with high concentrations of ethylenediaminetetraacetic acid (EDTA) or, most commonly, by competition with imidazole (100-250 mM).
Figure 2. Purification of His-tagged protein through IMAC
It often does not require removal before the use of the recombinant protein. While the tag can be removed by introducing a protease cleavage site, there are many examples of proteins that have been crystallized with intact His-tags and little or no impact on the structure of the tagged protein [3].
Additionally, the affinity of the His-tag for divalent metal cations is not dependent on a specific protein conformation, enabling the IMAC to be performed under denaturing conditions (e.g. 6 mol/l guanidine hydrochloride or 8 mol/l urea). This is useful when the target protein can not be expressed in a soluble form and refolding is being pursued.
The His-tag works well to rapidly purify proteins expressed in E. coli, but for proteins expressed in mammalian or insect cells, the greater number of histidine residues means there is higher background binding. Therefore, to increase protein purity, the metal resins that bind His-tagged proteins need to be stringently washed. In addition, denaturing agents like 8M urea or 6M guanidine HCl can be used with His-tagged membrane or insoluble proteins to improve purification.
5. His-tag Applications
His-tags have different applications in addition to their metal--coordinating properties used for protein purification.
|
Applications
|
Description
|
|
Preparation of His-specific antibody
|
His-tag is applied to prepare His-tag-specific antibodies that are useful for the detection of His-tagged proteins.
|
|
Detection of Protein-protein interaction
|
6*His tags can interact with immobilized metal cations to provide for the capture of a target protein complex of interest thus purify and identify it [7].
|
|
Separating one from two polyhistidine tags
|
Proteins with different numbers of polyhistidine tags elute from nickel-affinity resin in various ways. Specific protein assemblies, such as designated heteromultimers, may be isolated from a mixture using this step-by-step elution method. This method was applied to isolate monovalent streptavidin.
|
|
Fluorescent tags
|
Hexahistadine CyDye tags produce dyes that bind to the polyhistidine tag using Nickel covalent coordination to EDTA groups linked to fluorophores. This method has been demonstrated to work well for monitoring protein trafficking and migration. Recent research has also shown that his method may be useful for measuring distance using fluorescent resonance energy transfer [8].
|
|
Fluorohistidine tags
|
A polyfluorohistidine tag has been used in in vitro translation systems. In this approach, 4-fluorohistidine is employed in place of histidine in an extended genetic code. The histidine-tRNA ligase's relaxed substrate specificity allows for the incorporation of the fluorinated analog, which decreases the tag's total pKa. By changing the pH of the wash buffers, this enables the selective enrichment of polyfluorohistidine tagged peptides in the presence of complicated mixtures of classical polyhistidine tags [9].
|
|
Catalytic properties
|
His-tags also have catalytic proteties. Lise Schoonen et al. found that His-tags are able to ester hydrolysis when attached to the green fluorescent protein (GFP) and the cowpea chlorotic mottle virus (CCMV) capsid protein [10].
|
Since His-tag is available, the qualitative and quantitative expression of recombinant protein can be detected by WB, ELISA, and other methods. CUSABIO can provide several His-tag antibodies for relevant research.
References
[1] Hochuli E, Dobeli H, Schacher A. New metal chelate adsorbent selective for proteins and peptides containing neighbouring histidine residues [J]. J Chromatogr. 1987;411:177–184.
[2] Hochuli E. Large-scale chromatography of recombinant proteins [J]. J Chromatogr. 1988;444:293–302.
[3] Mike Carson, David H Johnson, et al. His-tag impact on structure [J]. Acta Crystallogr D Biol Crystallogr. 2007 Mar;63(Pt 3):295-301.
[4] Guignet, E.G., Hovius, R., Vogel, H. Reversible site-selective labeling of membrane proteins in live cells [J]. Nature Biotechnology, 22 (4), pp. 440-444 (2004).
[5] Fessenden, JD., Förster resonance energy transfer measurements of ryanodine receptor type 1 structure using a novel site-specific labeling method [J]. PLoS One, 4 (10), p. e7338 (2009).
[6] Bornhorst, J. A. & Falke, J. J. Purification of proteins using polyhistidine affinity tags [J]. Methods in enzymology, 326, 245–254 (2000).
[7] Mary C Puckett. Hexahistidine (6xHis) fusion-based assays for protein-protein interactions [J]. Methods Mol Biol. 2015;1278:365-70.
[8] Zhao, Chunxia, Hellman, Lance M. et al. (2010). Hexahistidine-tag-specific optical probes for analyses of proteins and their interactions [J]. Analytical Biochemistry. 399 (2): 237–45.
[9] Ring, Christine M.; Iqbal, Emil S.; et al. (2017-05-31). Genetic incorporation of 4-fluorohistidine into peptides enables selective affinity purification [J]. Organic & Biomolecular Chemistry. 15 (21): 4536–4539.
[10] Schoonen, L., van Esterik, K.S., Zhang, C. et al. Alternative application of an affinity purification tag: hexahistidines in ester hydrolysis [J]. Sci Rep 7, 14772 (2017).
CUSABIO team. Get An Overview of His-Tag. https://www.cusabio.com/c-21091.html



Comments
Leave a Comment