The HeLa cell line, the first immortalized human cell line, was established in 1951 [1]. In 1975, hybridoma technology enabled the generation of stable hybridoma cell lines producing monoclonal antibodies [2]. Recombinant DNA technology in the 1970s and 1980s allowed the introduction of genes into cells [3]. In the 1980s, transposons and retroviruses integrated exogenous genes into host cell genomes, establishing stable transgenic cell lines. In the 1990s and 2000s, improved gene delivery techniques enhanced efficiency [4-7]. In the 2010s, genome editing technologies like CRISPR-Cas9 enabled precise genetic modifications [8][9]. Continuous progress and innovation led to the establishment of stable cell lines.
Stable cell lines play a crucial role in various fields of biotechnology and biomedical research. This essay explores the definition, main characteristics, construction methods, and applications of stable cell lines, shedding light on their significance in modern research.
1. What Is A Stable Cell Line?
A stable cell line refers to a population of cells that have been genetically modified to express a specific gene or genetic construct in a stable and heritable manner. This stable expression is due to the integration of the foreign DNA into the cell's chromosomes, which allows for consistent production of the desired protein or function.
2. Main Characteristics of Stable Cell Lines
Stable cell lines possess several critical characteristics distinguishing them from primary or transiently transfected cells.
2.1 Genetic Stability
Stable cell lines offer a distinct advantage over transiently transfected cells as they ensure sustained and stable expression of the introduced gene. Unlike transient transfections where gene expression is temporary and diminishes over time, stable cell lines achieve stability by integrating the foreign gene into the host cell's genome, enabling consistent and continuous production. This stability is crucial for studying the effects of the introduced gene or mutation over extended periods and ensures reproducibility in downstream experiments.
2.2 Phenotypic Stability
Stable cell lines also exhibit phenotypic stability, meaning that they retain the desired phenotype associated with the transgene expression. This stability allows researchers to investigate the functional consequences of the introduced genetic modification and analyze the cellular responses under various experimental conditions consistently.
2.3 Reproducibility
Reproducibility is a hallmark of stable cell lines. By maintaining genetic and phenotypic stability, stable cell lines enable researchers to replicate experiments over time and across different laboratories. This consistency is critical in validating experimental findings, confirming observations, and establishing reliable protocols for subsequent research.
2.4 Long-term Culture
Unlike primary cells, which have a limited lifespan due to replicative senescence, stable cell lines can be maintained in culture for prolonged periods, ranging from months to years. This long-term culture capability is advantageous for long-duration experiments, continuous protein production, and facilitating the availability of research materials.
3. Construction of Stable Cell Lines
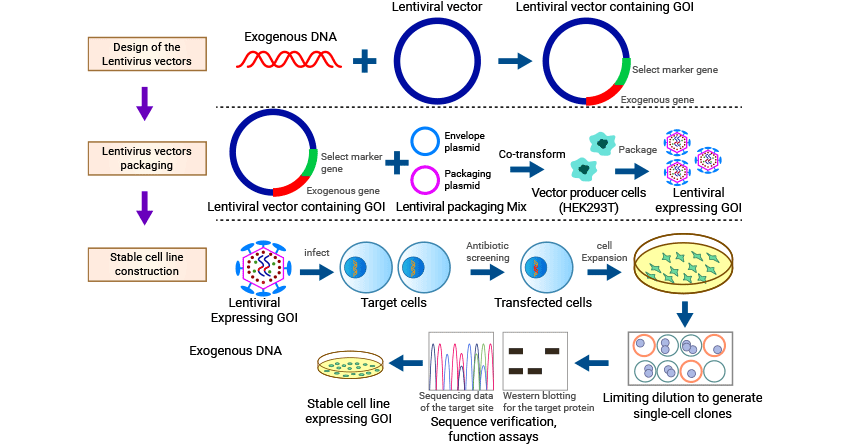
Here mainly introduces the construction of stable cell line through lentivirus.
3.1 Lentiviral Vector Construction
The lentiviral vector is constructed to carry the gene of interest (GOI) and other necessary genetic elements. The lentiviral vector typically contains a promoter (e.g., CMV) to drive the expression of the GOI, a selectable marker gene (e.g., antibiotic resistance gene) to select and identify transduced cells, and viral elements required for packaging and integration into the host genome. The GOI is cloned into the vector using molecular biology techniques such as restriction enzyme digestion, ligation, and transformation.
3.2 Lentiviral Packaging
Lentiviral packaging involves the production of infectious lentivirus particles using a packaging cell line like the HEK293T cell line. The lentiviral vector construct is co-transfected into the packaging cell line along with the helper plasmids (envelope plasmid e.g. pMD2.G and packaging plasmid e.g. pCMV-dR8.2 dvpr), leading to the generation of lentivirus particles that contain the vector construct.
After transfection, the cell culture supernatant was collected and filtered. The collected viral supernatant should be concentrated and purified to improve the titer and purity of the virus, facilitating the development of downstream experiments.
The fluorescence counting method and quantitative PCR method are used to detect the virus sequences integrated into host genomic DNA. p24 protein ELISA kit and qRT-PCR kit are used to detect the genome copy number in viral samples.
3.3 Stable Cell Line Establishment
● Transduction of Target Cells
The lentivirus particles are typically added to the target cells along with a transduction enhancer (e.g., polybrene) to increase transduction efficiency. Consider optimizing the multiplicity of infection (MOI) to achieve an appropriate transduction efficiency.
● Selection of Transduced Cells
Determine the appropriate concentration of a selective agent (e.g., antibiotic) that kills untransduced cells but allows transduced cells to survive. After transduction, the target cells are cultured in the medium containing the selective agent. Positive selection markers include puromycin, hygromycin, G418/Geneticin, and neomycin. This allows for the survival and expansion of only the transduced cells containing the lentivirus vector integrated into their genome.
● Screening and Isolation of Single Stable Cell Line Cloning
After several days of selection, screen the infected cells for stable GOI expression. Verify expression at the mRNA level using qPCR or protein level using Western blotting or immunocytochemistry. Isolate individual clones by limiting dilution or using other methods such as FACS or manual colony picking.
● Expansion and Characterization of Single Stable Cell Line Cloning
Expand the isolated cell clones in culture to establish stable cell lines and then cryopreserve them. Confirm the stable expression of GOI in the clones through additional analyses, such as immunofluorescence, flow cytometry, or functional assays specific to the GOI. Validate the stable cell lines by conducting appropriate functional assays or downstream experiments to ensure they meet your experimental requirements.
Figure 1. Construction of Stable Cell Lines based on lentivirus method
4. Applications of Stable Cell Lines
Stable cell lines find wide-ranging applications in various research fields, including biotechnology, pharmacology, and gene therapy. Some of the key applications are:
4.1 Recombinant Protein Production
Stable cell lines are extensively used for the production of recombinant proteins, including therapeutic proteins, antibodies, and industrial enzymes. The integration of the transgene encoding the protein of interest allows for continuous protein production, enabling large-scale manufacturing and reducing batch-to-batch variability.
4.2 Drug Discovery and Screening
Stable cell lines play a vital role in drug discovery and screening assays. By introducing specific drug targets or disease-related genes into stable cell lines, researchers can evaluate the efficacy and toxicity of potential drug candidates. These cell-based assays facilitate the identification of lead compounds, evaluation of drug-drug interactions, and elucidation of signaling pathways involved in disease progression.
4.3 Functional Genomics
Stable cell lines are valuable tools for studying gene function and regulation. By manipulating the expression of specific genes or introducing gene knockouts, researchers can investigate the role of individual genes in various biological processes. Stable cell lines provide a controlled system for functional genomics studies, enabling the characterization of gene function and the identification of potential drug targets.
4.4 Gene Therapy and Biomanufacturing
Stable cell lines play a crucial role in gene therapy, where genetically modified cells are used to deliver therapeutic genes to patients. These cell lines can be engineered to express therapeutic proteins, such as cytokines or growth factors, for applications in regenerative medicine or cancer immunotherapy. Additionally, stable cell lines are employed in biomanufacturing to produce viral vectors used in gene therapy or vaccines.
5. Challenges and Future Perspectives
Despite their numerous advantages, stable cell line generation and maintenance present challenges. These include off-target effects, variability in transgene expression, and the potential for genetic instability over prolonged culture periods. Future research should focus on improving the efficiency of stable cell line generation methods, optimizing transgene expression, and developing strategies to ensure long-term stability and consistent productivity.
In summary, stable cell lines have revolutionized the field of biotechnology and biomedical research, enabling the study of gene function, recombinant protein production, drug discovery, and gene therapy. Their genetic and phenotypic stability, reproducibility, and long-term culture capabilities make them indispensable tools in various research applications. Advancements in stable cell line construction and characterization techniques will continue to enhance their potential for scientific discovery and therapeutic development, fostering progress in biomedicine and improving human health.
CUSABIO provides several stable cell lines of popular drug targets to fuel drug development. If you are looking for a stable cell line for your experiment, you can go to this page: https://www.cusabio.com/catalog-71-1.html.
References
[1] Scherer, W. F., Syverton, J. T., & Gey, G. O. (1953). STUDIES ON THE PROPAGATION IN VITRO OF POLIOMYELITIS VIRUSES : IV. VIRAL MULTIPLICATION IN A STABLE STRAIN OF HUMAN MALIGNANT EPITHELIAL CELLS (STRAIN HELA) DERIVED FROM AN EPIDERMOID CARCINOMA OF THE CERVIX. The Journal of Experimental Medicine, 97(5), 695-710.
[2] Köhler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
[3] Wright, S. (1986). Recombinant DNA Technology and Its Social Transformation, 1972-1982. Osiris. Volume 2, Number 1.
[4] Sung, Y., Kim, S. Recent advances in the development of gene delivery systems. Biomater Res 23, 8 (2019).
[5] Jones CH, Chen CK, Ravikrishnan A, Rane S, Pfeifer BA (November 2013). "Overcoming nonviral gene delivery barriers: perspective and future". Molecular Pharmaceutics. 10 (11): 4082–98.
[6] Kamimura K, Suda T, Zhang G, Liu D (October 2011). "Advances in Gene Delivery Systems". Pharmaceutical Medicine. 25 (5): 293–306.
[7] Mali S (January 2013). "Delivery systems for gene therapy". Indian Journal of Human Genetics. 19 (1): 3–8.
[8] Gaj, T., Sirk, S. J., & Liu, J. (2016). Genome-Editing Technologies: Principles and Applications. Cold Spring Harbor Perspectives in Biology, 8(12).
[9] Khalil, A.M. The genome editing revolution: review. J Genet Eng Biotechnol 18, 68 (2020).
CUSABIO team. The Power and Promise of Stable Cell Lines. https://www.cusabio.com/c-21128.html



Comments
Leave a Comment