[1] Mattera, Vanesa, et al. "Remyelinating effect driven by transferrin‐loaded extracellular vesicles." Glia 72.2 (2024): 338-361.
[2] Koneru, Tejaswi, et al. "Transferrin: biology and use in receptor-targeted nanotherapy of gliomas." ACS omega 6.13 (2021): 8727-8733.
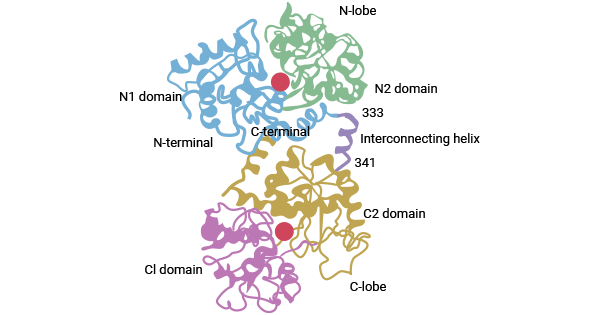
[3] Chasteen, N. Dennis. "Human serotransferrin: structure and function." Coordination Chemistry Reviews 22.1-2 (1977): 1-36.
[4] Kimawaha, Phongsaran, et al. "Serum α2, 6-sialylated glycoform of serotransferrin as a glycobiomarker for diagnosis and prediction of clinical severity in cholangiocarcinoma." Clinica Chimica Acta 536 (2022): 142-154.
[5] Barik, Sushanta Kumar, et al. "Identification and differential expression of serotransferrin and apolipoprotein AI in the plasma of HIV-1 patients treated with first-line antiretroviral therapy." BMC infectious diseases 20 (2020): 1-8.
[6] Lai, Ren, et al. "Transferrin is upregulated by microbes and acts as a negative regulator of immunity to induce intestinal immunotolerance." Research (2024).
[7] Zakin, Mario M. "Regulation of transferrin gene expression." The FASEB journal 6.14 (1992): 3253-3258.
[8] Lok, C. N., and T. T. Loh. "Regulation of transferrin function and expression: review and update." Neurosignals 7.3 (1998): 157-178.
[9] Ogun, Aminat S., and Adebayo Adeyinka. "Biochemistry, transferrin." StatPearls [Internet]. StatPearls Publishing, 2022.
[10] Silva, André MN, et al. "Human transferrin: An inorganic biochemistry perspective." Coordination Chemistry Reviews 449 (2021): 214186.
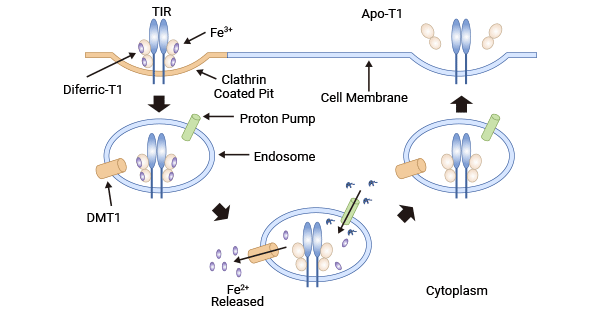
[11] Daniels TR, Delgado T, Helguera G, Penichet ML. The transferrin receptor part II: targeted delivery of therapeutic agents into cancer cells. Clin Immunol. 2006 Nov;121(2):159-76.
[12] Candelaria, Pierre V., et al. "Antibodies targeting the transferrin receptor 1 (TfR1) as direct anti-cancer agents." Frontiers in immunology 12 (2021): 607692.
[13] Li, Jiaojiao, and Wei Zhang. "From iron chelation to overload as a therapeutic strategy to induce ferroptosis in hematologic malignancies." Hematology 27.1 (2022): 1163-1170.
[14] Zhao, Yan, Zineng Huang, and Hongling Peng. "Molecular mechanisms of ferroptosis and its roles in hematologic malignancies." Frontiers in oncology 11 (2021): 743006.
[15] R Nogueira-Librelotto, Daniele, et al. "Transferrin-conjugated nanocarriers as active-targeted drug delivery platforms for cancer therapy." Current pharmaceutical design 23.3 (2017): 454-466.
[16] Li, XueMing, et al. "Targeted delivery of doxorubicin using stealth liposomes modified with transferrin." International journal of pharmaceutics 373.1-2 (2009): 116-123.
[17] Sen, Kacoli, and Mahitosh Mandal. "Second generation liposomal cancer therapeutics: transition from laboratory to clinic." International journal of pharmaceutics 448.1 (2013): 28-43.
[18] Huang, Shan-Zhou, et al. "Targeting TF-AKT/ERK-EGFR pathway suppresses the growth of hepatocellular carcinoma." Frontiers in oncology 9 (2019): 150.
[19] Li, Lihong, et al. "Targeted delivery of doxorubicin using transferrin-conjugated carbon dots for cancer therapy." ACS Applied Bio Materials 4.9 (2021): 7280-7289.
[20] Kölbl, Alexandra C., et al. "The role of TF-and Tn-antigens in breast cancer metastasis." (2016).
[21] Zhou, Jun, et al. "A transferrin-conjugated hollow nanoplatform for redox-controlled and targeted chemotherapy of tumor with reduced inflammatory reactions." Theranostics 8.2 (2018): 518.
[22] Mahani, Mohamad, et al. "Doxorubicin delivery to breast cancer cells with transferrin-targeted carbon quantum dots: An in vitro and in silico study." Journal of Drug Delivery Science and Technology 62 (2021): 102342.
[23] Ye, Yun, Su-Liang Li, and Sheng-Yu Wang. "Construction and analysis of mRNA, miRNA, lncRNA, and TF regulatory networks reveal the key genes associated with prostate cancer." PloS one 13.8 (2018): e0198055.
[24] Al Robaian, Majed, et al. "Therapeutic efficacy of intravenously administered transferrin-conjugated dendriplexes on prostate carcinomas." Nanomedicine 9.4 (2014): 421-434.
[25] Fernandes, Mariza Aires, et al. "Transferrin-functionalized liposomes for docetaxel delivery to prostate cancer cells." Colloids and Surfaces A: Physicochemical and Engineering Aspects 611 (2021): 125806.
[26] Dufès, Christine, Majed Al Robaian, and Sukrut Somani. "Transferrin and the transferrin receptor for the targeted delivery of therapeutic agents to the brain and cancer cells." Therapeutic delivery 4.5 (2013): 629-640.
[27] Wu, Yihe, et al. "Blocking transferrin receptor inhibits the growth of lung adenocarcinoma cells in vitro." Thoracic Cancer 9.2 (2018): 253-261.
[28] Deshpande, Pranali, et al. "Transferrin and octaarginine modified dual-functional liposomes with improved cancer cell targeting and enhanced intracellular delivery for the treatment of ovarian cancer." Drug delivery 25.1 (2018): 517-532.
[29] Metzgeroth, Georgia, et al. "The soluble transferrin receptor reflects tumor load in chronic lymphocytic leukemia." (2007): 1313-1318.
[30] Kobayashi, Eiji, et al. "Biomarkers for screening, diagnosis, and monitoring of ovarian cancer." Cancer Epidemiology, Biomarkers & Prevention 21.11 (2012): 1902-1912.
[31] Ge, Pingyun, et al. "Transferrin receptors/magnetic resonance dual-targeted nanoplatform for precise chemo-photodynamic synergistic cancer therapy." Nanomedicine: Nanotechnology, Biology and Medicine 39 (2022): 102467.
[32] Liu, Xiaoli, et al. "Therapeutic applications of multifunctional nanozymes." Nanoscale 11.44 (2019): 21046-21060.
[33] Boshuizen, M., et al. "Therapeutic use of transferrin to modulate anemia and conditions of iron toxicity." Blood Reviews 31.6 (2017): 400-405.
[34] Bruhn, Kevin W., and Brad Spellberg. "Transferrin-mediated iron sequestration as a novel therapy for bacterial and fungal infections." Current opinion in microbiology 27 (2015): 57-61.

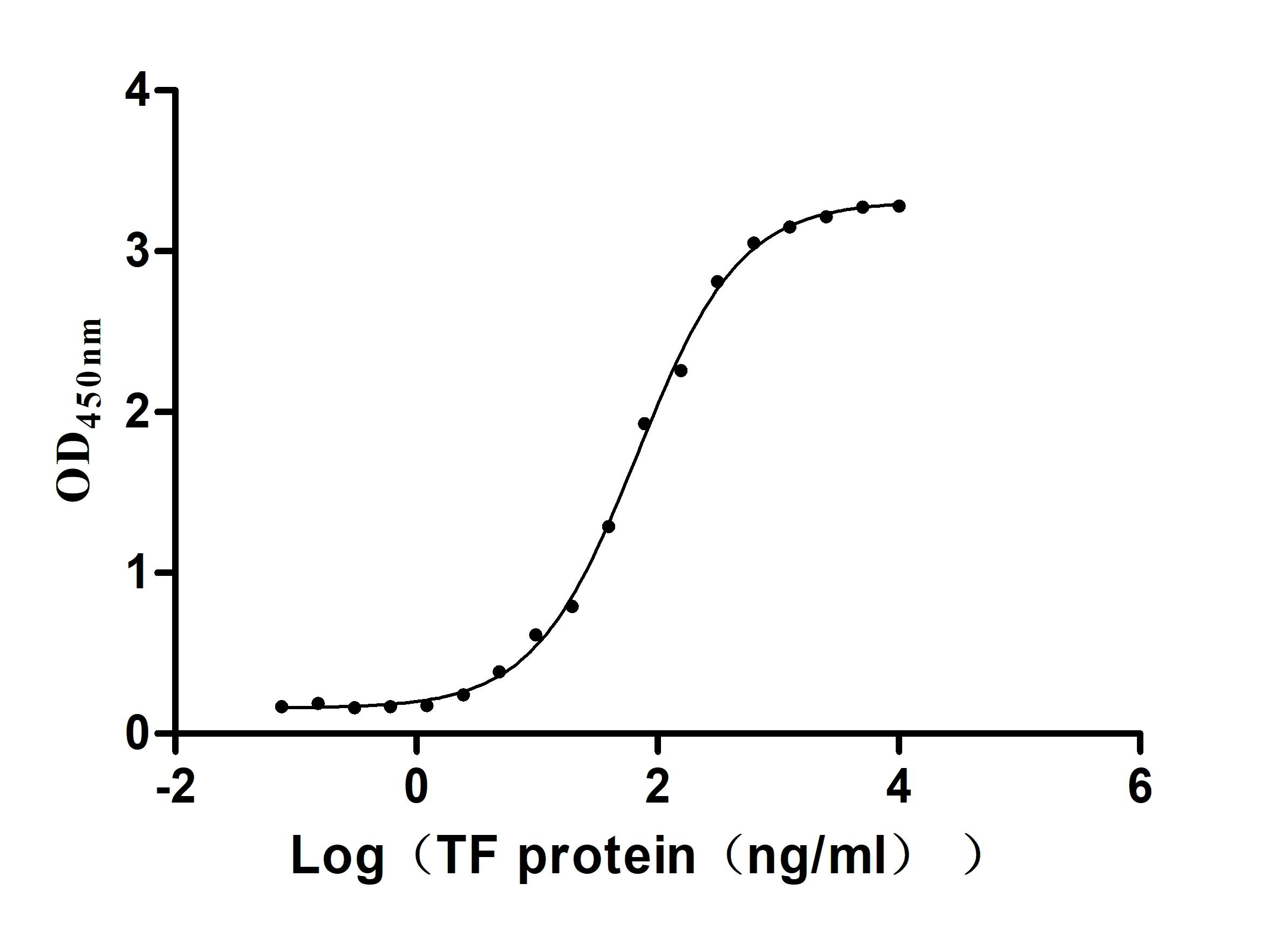


Comments
Leave a Comment