[1] Parthasarathy Sampathkumar, Heekyung Jung, Hui Chen, Zhengjian Zhang, Nicholas Suen, Yiran Yang, Zhong Huang, Tom Lopez, Robert Benisch, Sung‐Jin Lee, Jay Ye, Wen‐Chen Yeh, Yang Li.(2024). Targeted protein degradation systems to enhance Wnt signaling.
[2] Michael Tanowitz, Lisa Hettrick, Alexey S. Revenko, Garth A. Kinberger, Thazha P. Prakash, Punit P. Seth.(2017). Asialoglycoprotein receptor 1 mediates productive uptake of N-acetylgalactosamine-conjugated and unconjugated phosphorothioate antisense oligonucleotides into liver hepatocytes.
[3] Michael Tanowitz, Lisa Hettrick, Alexey S. Revenko, Garth A. Kinberger, Thazha P. Prakash, Punit P. Seth.(2017). Asialoglycoprotein receptor 1 mediates productive uptake of N-acetylgalactosamine-conjugated and unconjugated phosphorothioate antisense oligonucleotides into liver hepatocytes.
[4] Harry Aldworth, Nigel M. Hooper.(2024). Post-translational regulation of the low-density lipoprotein receptor provides new targets for cholesterol regulation.
[5] Yingying Xu, Jiawang Tao, Xiaorui Yu, Yuhang Wu, Yan Chen, Kai You, Jiaye Zhang, Anteneh Getachew, Tingcai Pan, Yuanqi Zhuang, Fang Yuan, Fan Yang, Xian-Hua Lin, Yinxiong Li.(2021). Hypomorphic ASGR1 modulates lipid homeostasis via INSIG1-mediated SREBP signaling suppression.
[6] Xiaorui Yu, Jiawang Tao, Yuhang Wu, Yan Chen, Penghui Li, Fan Yang, Miaoxiu Tang, Abdul Sammad, Tao Yu, Yingying Xu, Yinxiong Li.(2024). Deficiency of ASGR1 Alleviates Diet-Induced Systemic Insulin Resistance via Improved Hepatic Insulin Sensitivity.
[7] Paul Nioi, Ásgeir Sigurðsson, Guðmar Þorleifsson, Hannes Helgason, Arna B. Agustsdottir, Gudmundur L. Norddahl, Anna Helgadóttir, Audur Magnusdottir, Áslaug Jónasdóttir, Sólveig Grétarsdóttir, Ingileif Jónsdóttir, Valgerður Steinthórsdóttir, Þórunn Rafnar, Dorine W. Swinkels, Tessel E. Galesloot, Niels Grarup, Torben Jørgensen, Henrik Vestergaard, Torben Hansen, Torsten Lauritzen, Allan Linneberg, Nele Friedrich, Nikolaj T. Krarup, Mogens Fenger, Ulrik Abildgaard, Peter Riis Hansen, Anders Galløe, Peter S. Braund, Christopher P. Nelson, Alistair S. Hall, Michael Williams, André M. van Rij, Gregory T. Jones, Riyaz Patel, Allan I. Levey, Salim S. Hayek, Svati H. Shah, Muredach P. Reilly, Guðmundur I. Eyjólfsson, Ólöf Sigurðardóttir, Ísleifur Ólafsson, Lambertus A. Kiemeney, Arshed A. Quyyumi, Daniel J. Rader, William E. Kraus, Mark M. Iles, Oluf Pedersen, Guðmundur Þorgeirsson, Gísli Másson, Hilma Hólm, Daníel F. Guðbjartsson, Patrick Sulem, Unnur Þorsteinsdóttir, Kári Stéfansson.(2016). VariantASGR1Associated with a Reduced Risk of Coronary Artery Disease.
[8] Monika Svecla, Lorenzo Da Dalt, Annalisa Moregola, Jasmine Nour, Andrea Baragetti, Patrizia Uboldi, Elena Donetti, Lorenzo Arnaboldi, Giangiacomo Beretta, Fabrizia Bonacina, Giuseppe Danilo Norata.(2024). ASGR1 deficiency diverts lipids toward adipose tissue but results in liver damage during obesity.
[9] Baocai Xie, Xiaochen Shi, Yán Li, Bo Xia, Jia Zhou, Minjie Du, Xiangyang Xing, Liang Bai, Enqi Liu, Fernando Álvarez, Long Jin, Shaoping Deng, Grant A. Mitchell, Dengke Pan, Mingzhou Li, Jiangwei Wu.(2021). Deficiency of ASGR1 in pigs recapitulates reduced risk factor for cardiovascular disease in humans.
[10] Homa Hamledari, Seyedeh Fatemeh Sajjadi, Asieh Alikhah, Mohammad Ali Boroumand, Mehrdad Behmanesh.(2019). ASGR1 but not FOXM1 expression decreases in the peripheral blood mononuclear cells of diabetic atherosclerotic patients.
[11] Xinyi Yang, Yuqi Zhu, Xiaying Zhao, Jun Liu, Jiangna Xun, Songhua Yuan, Jun Chen, Hanyu Pan, Jinlong Yang, Jing Wang, Zhimin Liang, Xiaoting Shen, Liang Yue, Qinru Lin, Huitong Liang, Min Li, Hongzhou Lu, Huanzhang Zhu.(2022). ASGR1 is a candidate receptor for SARS-CoV-2 that promotes infection of liver cells.
[12] Daniel P. Collins, Clifford J. Steer.(2021). Binding of the SARS-CoV-2 Spike Protein to the Asialoglycoprotein Receptor on Human Primary Hepatocytes and Immortalized Hepatocyte-Like Cells by Confocal Analysis.
[13] Li Zhang, Yabin Tian, Zhiheng Wen, Feng Zhang, Ying Qi, Weijin Huang, Heqiu Zhang, Youchun Wang.(2016). Asialoglycoprotein receptor facilitates infection of PLC/PRF/5 cells by HEV through interaction with ORF2.
[14] Xingxin Zhu, Guangyuan Song, Shiyu Zhang, Jun Chen, Xiaoyi Hu, Hai Zhu, Xing Jia, Zequn Li, Wenfeng Song, Jian Chen, Cheng Jin, Mengqiao Zhou, Yongchao Zhao, Haiyang Xie, Shusen Zheng, Penghong Song.(2022). Asialoglycoprotein Receptor 1 Functions as a Tumor Suppressor in Liver Cancer via Inhibition of STAT3.
[15] Amparo Roa Colomo, María Ángeles López Garrido, Pilar Molina-Vallejo, Ángela Rojas, Mercedes González Sanchez, Violeta Aranda-García, Javier Salmerón, Manuel Romero‐Gómez, Jordi Muntané, Javier Padillo–Ruiz, J.M. Álamo-Martínez, José A. Lorente, María José Serrano, M. Carmen Garrido-Navas.(2022). Hepatocellular carcinoma risk-stratification based on ASGR1 in circulating epithelial cells for cancer interception.

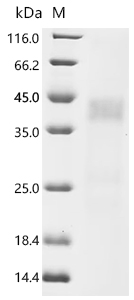
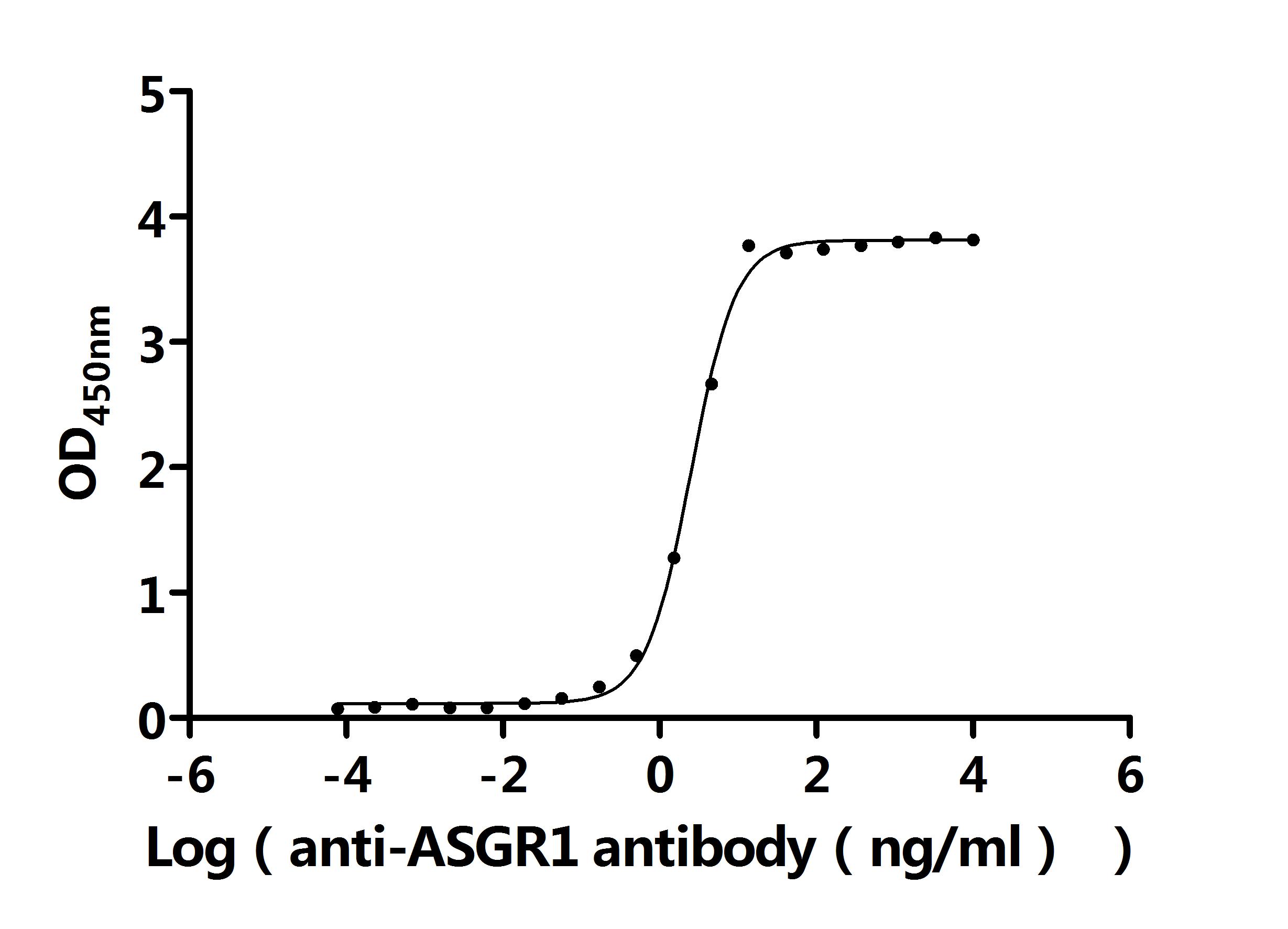

-D-SDS.jpg)
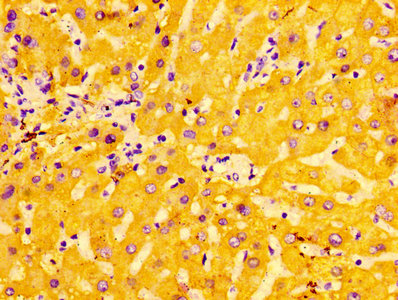
-D-AC1.jpg)
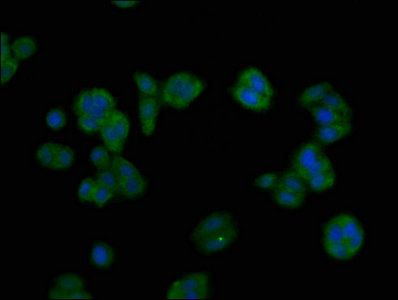
-D-AC2.jpg)


Comments
Leave a Comment