[1] Deborah M. Brown, Anna T. Lampe, Aspen M. Workman. (2016). The Differentiation and Protective Function of Cytolytic CD4 T Cells in Influenza Infection.
[2] Tomas Lukes, Daniela Glatzova, Zuzana Kvicalova, Florian Levet, Ales Benda, Tomas Brdicka, Theo Lasser, Marek Cebecauer. (2017). Quantifying protein densities on cell membranes using super-resolution optical fluctuation imaging.
[3] Mikaël Simard, Zhuoyan Shen, Konstantin Bräutigam, Rasha Abu-Eid, Maria A. Hawkins, Charles-Antoine Collins-Fekete. (2024). Immunocto: a massive immune cell database auto-generated for histopathology.
[4] Xin Shu, Sameera Sansre, Di Jin, Xiangxiang Zeng, Kai-Yu Tong, Rishikesh Pandey, Renjie Zhou. (2020). Artificial Intelligence Enabled Reagent-free Imaging Hematology Analyzer.
[5] D. Rider, C. Havenith, R. De Ridder, J. Schuurman, C. Favre, J. Cooper, S. Walker, O. Baadsgaard, S. Marschner, Jan G J vandeWinkel, J. Cambier, P. Parren, D. Alexander. (2007). A human CD4 monoclonal antibody for the treatment of T-cell lymphoma combines inhibition of T-cell signaling by a dual mechanism with potent Fc-dependent effector activity.
[6] J. T. Ulrich-Lewis, K. Draves, Kelsey Roe, M. O'Connor, E. Clark, D. Fuller. (2022). STING Is Required in Conventional Dendritic Cells for DNA Vaccine Induction of Type I T Helper Cell- Dependent Antibody Responses.
[7] Constance Renault, N. Veyrenche, F. Mennechet, A. Bedin, J. Routy, P. Van de Perre, J. Reynes, E. Tuaillon. (2022). Th17 CD4+ T-Cell as a Preferential Target for HIV Reservoirs.
[8] Rong Wang, Keliang Huang. (2020). CCL11 increases the proportion of CD4+CD25+Foxp3+ Treg cells and the production of IL-2 and TGF-β by CD4+ T cells via the STAT5 signaling pathway.
[9] Jiaxu Qin, Xiangwei Zheng, Yancheng He, Yang Hong, Shuang Liang, X. Fang. (2022). The regulation of T helper cell polarization by the diterpenoid fraction of Rhododendron molle based on the JAK/STAT signaling pathway.
[10] Fanping Wang, Yonghui Yang, Zhixin Li, Yan Wang, Zhenchao Zhang, Wei Zhang, Yonghui Mu, Jing-ke Yang, Lili Yu, Mingyong Wang. (2022). Mannan-Binding Lectin Regulates the Th17/Treg Axis Through JAK/STAT and TGF-β/SMAD Signaling Against Candida albicans Infection.
[11] R. Deng, C. Hurtz, Qingxiao Song, Chanyu Yue, G. Xiao, Hua Yu, Xiwei Wu, M. Muschen, S. Forman, P. Martin, D. Zeng. (2017). Extrafollicular CD4+ T-B interactions are sufficient for inducing autoimmune-like chronic graft-versus-host disease.
[12] Laura M. Fahey, Elizabeth B. Wilson, Heidi J. Elsaesser, Chris Fistonich, D. McGavern, D. Brooks. (2011). Viral persistence redirects CD4 T cell differentiation toward T follicular helper cells.
[13] A. Dejean, Emeline Joulia, T. Walzer. (2018). The role of Eomes in human CD4 T cell differentiation: A question of context.
[14] Yuki Sato, Abhinav Jain, Shozo Ohtsuki, Hirohisa Okuyama, Ines Sturmlechner, Yoshinori Takashima, Kevin-Phu C. Le, Melanie C. Bois, Gerald J. Berry, K. Warrington, J. Goronzy, C. Weyand. (2023). Stem-like CD4+ T cells in perivascular tertiary lymphoid structures sustain autoimmune vasculitis.
[15] S. Keck, M. Schmaler, S. Ganter, Lena Wyss, Susanne G. Oberle, E. Huseby, D. Zehn, C. King. (2014). Antigen affinity and antigen dose exert distinct influences on CD4 T-cell differentiation.
[16] M. Martínez-Sánchez, L. Huerta, E. Álvarez-Buylla, Carlos Villarreal Luján. (2018). Role of Cytokine Combinations on CD4+ T Cell Differentiation, Partial Polarization, and Plasticity: Continuous Network Modeling Approach.
[17] E. Zorn, E. Nelson, M. Mohseni, F. Porcheray, Haesook T. Kim, Despina Litsa, R. Bellucci, E. Raderschall, C. Canning, R. Soiffer, D. Frank, J. Ritz. (2006). IL-2 regulates FOXP3 expression in human CD4+CD25+ regulatory T cells through a STAT-dependent mechanism and induces the expansion of these cells in vivo.
[18] M. Kroenke, D. Eto, M. Locci, Michael Cho, T. Davidson, E. Haddad, S. Crotty. (2012). Bcl6 and Maf Cooperate To Instruct Human Follicular Helper CD4 T Cell Differentiation.
[19] Megan A. Luckey, M. Kimura, A. Waickman, L. Feigenbaum, A. Singer, Jung-Hyun Park. (2014). The transcription factor ThPOK suppresses Runx3 and imposes CD4+ lineage fate by inducing the SOCS suppressors of cytokine signaling.
[20] Qingtian Li, Jia Zou, Mingjun Wang, Xilai Ding, I. Chepelev, Xikun Zhou, Wei Zhao, Gang Wei, J. Cui, K. Zhao, Helen Y Wang, Rong-Fu Wang. (2014). Critical role of histone demethylase Jmjd3 in the regulation of CD4+ T-cell differentiation.
[21] Y. Shu, Qinghua Hu, H. Long, Christopher Chang, Q. Lu, R. Xiao. (2017). Epigenetic Variability of CD4+CD25+ Tregs Contributes to the Pathogenesis of Autoimmune Diseases.
[22] Sarah A. LaMere, Ryan C. Thompson, Xiangzhi Meng, H. Komori, Adam M. Mark, D. Salomon. (2017). H3K27 Methylation Dynamics during CD4 T Cell Activation: Regulation of JAK/STAT and IL12RB2 Expression by JMJD3.
[23] M. Martínez-Sánchez, L. Mendoza, C. Villarreal, E. Álvarez-Buylla. (2015). A Minimal Regulatory Network of Extrinsic and Intrinsic Factors Recovers Observed Patterns of CD4+ T Cell Differentiation and Plasticity.
[24] B. L. Puniya, R. G. Todd, Akram Mohammed, Deborah M. Brown, Matteo Barberis, T. Helikar. (2018). A Mechanistic Computational Model Reveals That Plasticity of CD4+ T Cell Differentiation Is a Function of Cytokine Composition and Dosage.
[25] V. Proserpio, A. Piccolo, Liora Haim-Vilmovsky, G. Kar, T. Lönnberg, Valentine Svensson, J. Pramanik, K. Natarajan, Weichao Zhai, Xiuwei Zhang, G. Donati, M. Kayikci, J. Kotar, A. McKenzie, Ruddy Montandon, O. Billker, S. Woodhouse, P. Cicuta, M. Nicodemi, S. Teichmann. (2016). Single-cell analysis of CD4+ T-cell differentiation reveals three major cell states and progressive acceleration of proliferation.
[26] S. Tullius, H. Biefer, Suyan Li, A. Trachtenberg, K. Edtinger, M. Quante, Felix Krenzien, H. Uehara, Xiaoyong Yang, H. Kissick, W. Kuo, I. Ghiran, M. A. de la Fuente, M. Arredouani, Virginia Camacho, J. Tigges, Vasilis Toxavidis, Rachid El Fatimy, Brian D. Smith, A. Vasudevan, A. Elkhal. (2014). NAD+ protects against EAE by regulating CD4+ T-cell differentiation.
[27] Nizar Chetoui, M. Boisvert, S. Gendron, F. Aoudjit. (2010). Interleukin‐7 promotes the survival of human CD4+ effector/memory T cells by up‐regulating Bcl‐2 proteins and activating the JAK/STAT signalling pathway.
[28] M. S. Alam, Y. Maekawa, A. Kitamura, K. Tanigaki, T. Yoshimoto, K. Kishihara, K. Yasutomo. (2010). Notch signaling drives IL-22 secretion in CD4+ T cells by stimulating the aryl hydrocarbon receptor.
[29] Shengpan Wu, Baojun Wang, Hong-zhao Li, Hanfeng Wang, Songliang Du, Xing Huang, Yang Fan, Yu Gao, L. Gu, Qingbo Huang, Jianjun Chen, Xu Zhang, Yan Huang, Xin Ma. (2024). Targeting STing elicits GSDMD-dependent pyroptosis and boosts anti-tumor immunity in renal cell carcinoma.
[30] Dongmei Li, Idalia Cruz, Yahui Feng, Maha Moussa, Jie Cheng, Digvijay Patil, Alexander Kroemer, Joseph A. Bellanti. (2025). Divergent Immunomodulatory Roles of Fungal DNA in Shaping Treg and Inflammatory Responses.
[31] C. Elsner, Aparna Ponnurangam, J. Kazmierski, T. Zillinger, Jenny Jansen, D. Todt, K. Döhner, Shuting Xu, A. Ducroux, Nils Kriedemann, Angelina Malassa, P. Larsen, G. Hartmann, W. Barchet, E. Steinmann, U. Kalinke, B. Sodeik, C. Goffinet. (2020). Absence of cGAS-mediated type I IFN responses in HIV-1--infected T cells.
[32] Tatyana Veremeyko, Amanda W. Y. Yung, M. Dukhinova, Inna S. Kuznetsova, I. Pomytkin, A. Lyundup, T. Strekalova, N. Barteneva, E. Ponomarev. (2018). Cyclic AMP Pathway Suppress Autoimmune Neuroinflammation by Inhibiting Functions of Encephalitogenic CD4 T Cells and Enhancing M2 Macrophage Polarization at the Site of Inflammation.

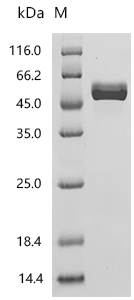
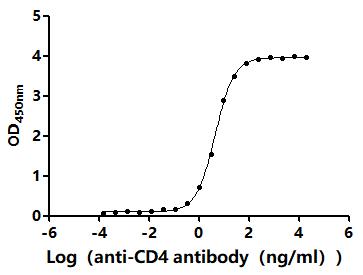
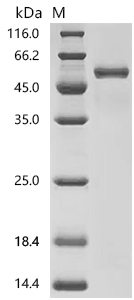
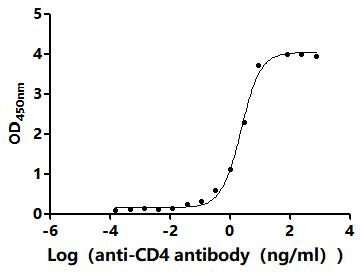
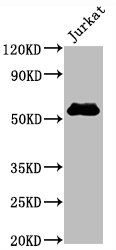
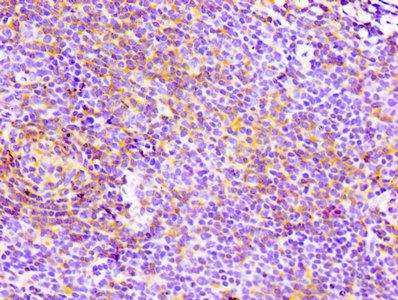
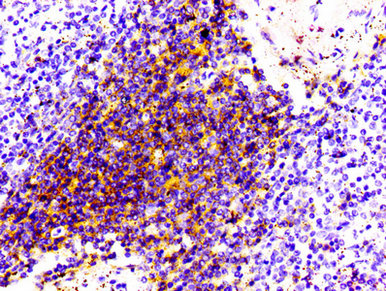
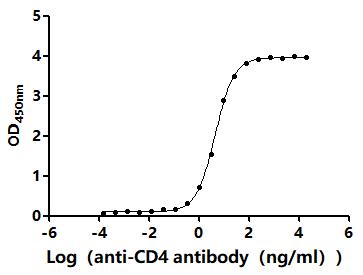


Comments
Leave a Comment