CD33 (Siglec-3) is an important member of the Siglec family, primarily expressed on the surface of myeloid cells such as monocytes, macrophages, and dendritic cells. As an inhibitory immune receptor, CD33 mediates signal transduction through its intracellular immunoreceptor tyrosine-based inhibitory motif (ITIM), playing a crucial role in regulating immune homeostasis and inflammatory responses. Recent studies have shown that CD33 not only serves as a significant targeting antigen in acute myeloid leukemia (AML) but also plays key roles in Alzheimer's disease (AD), chronic viral infections, and tumor immune microenvironment regulation. Particularly in the nervous system, CD33 influences β-amyloid clearance by regulating microglial phagocytic function, thereby participating in the pathogenesis of neurodegenerative diseases. With the advancement of immunotherapy and genomic research, CD33 has gradually become an important molecule in immune regulation research and disease-targeted therapy. This review summarizes the molecular characteristics, major signaling pathways, and roles of CD33 in various diseases, aiming to provide reference for mechanistic research and targeted therapy development for diseases such as acute myeloid leukemia and Alzheimer's disease.
1. Research Background of CD33 in Immune Regulation: From Siglec Family to Disease Target
CD33 (Sialic acid-binding immunoglobulin-like lectin-3, Siglec-3) belongs to the Siglec family, which recognizes sialic acid-modified glycan structures and plays an important negative regulatory role in the immune system [1],[2]. CD33 is mainly expressed in myeloid cells, including monocytes, macrophages, dendritic cells, and myeloid progenitor cells, and is of great significance in regulating innate immune responses and maintaining immune homeostasis.
The intracellular domain of CD33 contains a typical immunoreceptor tyrosine-based inhibitory motif (ITIM). When CD33 binds to its ligand, this domain is phosphorylated and recruits SH2 domain-containing phosphatases such as SHP-1 and SHP-2, thereby inhibiting the activation of various cellular signaling pathways [3]. This inhibitory signaling mechanism makes CD33 an important regulatory factor in myeloid cell immune responses.
In recent years, with advancements in immunology, neurobiology, and tumor research, the role of CD33 in various diseases has gradually gained attention. Studies have found that approximately 80%--90% of leukemia cells in acute myeloid leukemia (AML) express CD33, making this molecule an important therapeutic target [4]. Simultaneously, genome-wide association studies have shown that CD33 gene polymorphisms are significantly associated with Alzheimer's disease risk, with its mechanism closely related to microglial function regulation [5]. Additionally, CD33 participates in regulating the immunosuppressive function of myeloid cells in chronic viral infections and tumor immune microenvironments. Therefore, systematic study of CD33 signaling mechanisms and their disease correlations is of great significance for understanding immune regulatory networks and developing new therapeutic strategies.
2. Molecular Biological Characteristics of CD33: Structure, Function, and Splice Variants
CD33 is a type I transmembrane glycoprotein whose structure consists mainly of an extracellular domain, transmembrane domain, and intracellular domain. The extracellular domain contains two immunoglobulin-like domains: the distal IgV domain and the proximal membrane IgC2 domain. The IgV domain recognizes sialylated glycan ligands and is the key structural element for CD33 ligand recognition. The transmembrane domain anchors CD33 to the cell membrane and maintains its spatial conformational stability.
The intracellular domain of CD33 contains a conserved ITIM sequence. When CD33 binds to its ligand and becomes activated, tyrosine residues in this sequence are phosphorylated, subsequently recruiting SHP-1, SHP-2, and other phosphatases, thereby inhibiting the activation of multiple cell signaling pathways [3]. This inhibitory signaling mechanism is of great importance in the immune system, preventing excessive inflammatory responses and maintaining immune balance.
At the post-transcriptional regulatory level, the CD33 gene also exhibits multiple splice variants. The most common are full-length CD33-FL and CD33-Δ2, which lacks exon 2. Since exon 2 encodes the IgV domain, its deletion prevents CD33 from effectively binding sialic acid ligands, thereby altering receptor function. Studies have shown that genetic variants associated with Alzheimer's disease protection promote the expression of CD33-Δ2, thereby reducing functional CD33 levels and enhancing microglial phagocytic capacity [5].
CD33 is primarily expressed in myeloid cells, with expression levels dynamically changing during hematopoietic differentiation. Early myeloid progenitor cells show higher expression levels, while mature granulocytes exhibit relatively lower expression. This expression pattern reflects the important role of CD33 in myeloid cell differentiation and immune regulation.
3. CD33-Related Signaling Pathways: ITIM-Mediated Immune Regulatory Mechanisms
3.1 CD33's ITIM-Mediated Inhibitory Signaling Pathway
The most classic signal transduction mechanism of CD33 is through its intracellular ITIM domain-mediated inhibitory signaling. When CD33 recognizes sialylated ligands on the cell surface, its intracellular ITIM sequence is phosphorylated by Src family kinases. Subsequently, SH2 domain-containing protein phosphatases SHP-1 or SHP-2 are recruited to this domain and inhibit the activation of various downstream signaling molecules through dephosphorylation [3]. This mechanism reduces the activation level of immune cells, thereby avoiding excessive inflammatory responses.
In myeloid cells, this signaling pathway can inhibit multiple inflammation-related signaling molecules, such as Src kinases, MAPK pathways, and NF-κB signaling pathways. Through this mechanism, CD33 can regulate the inflammatory response levels of macrophages and dendritic cells, thereby maintaining immune system homeostasis.
3.2 CD33 and Microglial Signal Regulation: Key Mechanisms in Alzheimer's Disease
In the central nervous system, CD33 is mainly expressed in microglia. Microglia are the primary immune cells in the brain, responsible for clearing pathogens and abnormal proteins. Studies have shown that CD33 primarily plays an inhibitory regulatory role in microglia. When CD33 expression increases, the phagocytic activity of microglia significantly decreases, thereby reducing their ability to clear β-amyloid protein.
In this process, there is a functional antagonistic relationship between CD33 and the TREM2 signaling pathway. TREM2 promotes microglial activation and enhances phagocytic function, while CD33 reduces cellular activity by inhibiting signal transduction. When CD33 expression increases or signaling is enhanced, microglial phagocytic function is inhibited, thereby promoting β-amyloid deposition and accelerating the progression of neurodegenerative lesions.
3.3 CD33's Signaling Role in the Tumor Immune Microenvironment: Functional Regulation of Myeloid-Derived Suppressor Cells
Recent studies have shown that CD33 also plays an important role in the tumor immune microenvironment. Tumor tissues are often enriched with large numbers of myeloid-derived suppressor cells (MDSCs), which promote tumor immune escape by inhibiting T cell activity. MDSCs typically express high levels of CD33, and their signaling pathways can enhance immunosuppressive function and inhibit anti-tumor immune responses.
Activation of CD33 signaling can regulate the expression of various immune regulatory factors, such as inhibitory cytokines and immune checkpoint molecules, thereby further strengthening the tumor immunosuppressive environment. Therefore, CD33 is considered an important molecule in regulating the tumor immune microenvironment.
4. CD33-Related Diseases: From Hematological Tumors to Neurodegenerative Diseases
4.1 Acute Myeloid Leukemia (AML): CD33 as an Important Therapeutic Target
Acute myeloid leukemia is one of the most extensively studied diseases involving CD33. AML is a malignant hematological tumor originating from myeloid progenitor cells, characterized by the proliferation of abnormal leukemia cells in the bone marrow and peripheral blood. Studies have shown that the vast majority of AML patients express CD33 molecules on the surface of their leukemia cells [4]. This feature makes CD33 an important molecular marker for AML diagnosis, classification, and targeted therapy.
In the pathogenesis of AML, CD33 not only exists as a surface antigen but may also participate in regulating the proliferation and differentiation of leukemia cells. Through CD33-targeted antibodies or immunotherapeutic approaches, cytotoxic drugs can be selectively delivered to leukemia cells, thereby improving treatment efficacy. However, since normal myeloid cells also express CD33, this may lead to side effects such as bone marrow suppression.
4.2 Alzheimer's Disease (AD): CD33 Gene Polymorphisms and Disease Risk
Alzheimer's disease is a common neurodegenerative disease whose main pathological features include β-amyloid deposition and neuronal degeneration. Recent genomic studies have shown that CD33 is an important risk gene associated with late-onset Alzheimer's disease [5].
In the nervous system, CD33 is mainly expressed in microglia. Research has found that CD33 can inhibit the phagocytic capacity of microglia for β-amyloid protein, thereby affecting its clearance efficiency. When CD33 expression levels increase, microglial phagocytic function decreases, β-amyloid deposition increases, thereby promoting the development of neurodegenerative lesions.
Furthermore, CD33 gene splicing variants are also closely related to disease risk. Some genetic variants can promote the expression of the CD33-Δ2 form, thereby reducing functional CD33 production and enhancing microglial clearance capacity, which is considered an important mechanism for its protective effect against Alzheimer's disease.
4.3 Chronic Viral Infections: CD33's Role in Immune Regulation
In chronic viral infectious diseases such as hepatitis B, CD33 also participates in immune regulatory processes. Research has shown that CD33 can inhibit the antigen presentation function of dendritic cells and reduce T cell activation levels, thereby promoting viral persistence. Blocking CD33 signaling can, to some extent, restore immune cell activity and enhance antiviral immune responses.
4.4 Tumor Immune Regulation: Synergistic Action of CD33 and Myeloid-Derived Suppressor Cells
In various solid tumors, CD33 also participates in the formation of the tumor immune microenvironment. Tumor tissues are often enriched with myeloid-derived suppressor cells, which promote tumor growth by secreting immunosuppressive factors and inhibiting T cell function. Since these cells typically express high levels of CD33, this molecule is considered an important target for regulating tumor immunosuppression.
5. Latest Research Advances in CD33-Targeted Drugs: Multi-Platform Therapeutic Strategies from ADC to CAR-T
Currently, the development of CD33-targeted drugs has entered a stage of diversified, platform-based rapid development. This target has been proven to have core value in the treatment of acute myeloid leukemia (AML) and its related subtypes (such as refractory/relapsed acute myeloid leukemia, myelodysplastic syndrome, etc.). From a technological perspective, this field is no longer limited to traditional antibody-drug conjugates (ADCs) but has comprehensively deployed new-generation treatment modalities including CAR-T, CAR-NK, bispecific antibodies, DARPins, immunostimulatory antibody conjugates (ISACs), and targeted degradation antibodies. The research and development strategy has also evolved from single-target to dual-target and even multi-target (such as CD33 combined with CD123, CLL-1, etc.) synergistic attacks, aiming to improve treatment efficacy and overcome drug resistance caused by tumor heterogeneity. Although some drugs have been approved for marketing, the majority of research pipelines are still concentrated in Phase 1 and Phase 2 clinical stages. This indicates that CD33, as a classic target, is currently undergoing a wave of clinical re-development driven by technological innovation, and is expected to further improve the clinical prognosis of patients with hematological malignancies through more precise and efficient immunotherapies and genetically modified cell therapies in the future. Some research pipelines are listed below:
| Drug |
Target |
Drug Type |
Investigated Indications |
Investigational Institutions |
Highest Development Phase |
| Gemtuzumab | CD33 x DNA | ADC | CD33-positive acute myeloid leukemia | Acute myeloid leukemia | Acute promyelocytic leukemia etc. | Pfizer Inc. | Pfizer Japan, Inc. | Wyeth Pharmaceuticals LLC | Pfizer Europe MA EEIG | The Children's Mercy Hospital | Pfizer Canada ULC | Approved |
| BL-M11D1 | CD33 x Top I | ADC | Acute myeloid leukemia | Refractory acute myeloid leukemia | Recurrent acute myeloid leukemia | Myelodysplastic syndromes | Sichuan Baili Pharmaceutical Co., Ltd. | Systimmune, Inc. | Phase 2/3 |
| VCAR-33 (National Cancer Institute) | CD33 | Autologous CAR-T | Refractory acute myeloid leukemia | Recurrent acute myeloid leukemia | Center for International Blood & Marrow Transplant Research | Phase 1/2 |
| CD123/CD33 CART (Essen Biotech) | CD123 x CD33 | CAR-T | Adult acute myeloid leukemia | Refractory acute myeloid leukemia | Essen BioTech, LLC. | Phase 1/2 |
| CD33/CD123/CLL-1 CAR-T Cells (Hebei Taihe Chunyu Biotechnology) | CD123 x CD33 x CLL-1 | CAR-T | - | Hebei Taihe Chunyu Biotechnology Co., Ltd. | Phase 1/2 |
| Lintuzumab Ac-225 | CD33 | Antibody-radionuclide conjugate | Therapeutic radiopharmaceutical | Adult acute myeloid leukemia | CD33-positive acute myeloid leukemia | Refractory acute myeloid leukemia etc. | National Cancer Institute | Actinium Pharmaceuticals, Inc. | Phase 1/2 |
| Anti-CLL1-CD33-NKG2D Bicephali CAR-T (Yake) | CD33 x CLL-1 x NKG2D | CAR-T | Refractory acute myeloid leukemia | Shanghai Yake Biotechnology Co., Ltd. | Phase 1/2 |
| Universal donor derived CD33 CAR-NK (Nationwide Children's Hospital) | CD33 | CAR-NK | - | Nationwide Children's Hospital | Phase 1/2 |
| MP-0533 | CD123 x CD3 x CD33 x CD70 | DARPin | Myelodysplastic syndromes | Refractory acute myeloid leukemia | Molecular Partners AG | Phase 1/2 |
| CLL1+CD33 CAR-T (Zhejiang University) | CD33 x CLL-1 | CAR-T | Acute myeloid leukemia | Zhejiang University | Phase 1 |
| CD33 CAR-NK (Xinqiao Hospital) | CD33 | CAR-NK | Refractory acute myeloid leukemia | Recurrent acute myeloid leukemia | Xinqiao Hospital | Phase 1 |
| ORM-6151 | CD33 x GSPT1 | Degraduct-ADC (Antibody degrader conjugate) | Myelodysplastic syndromes | Refractory acute myeloid leukemia | Acute myeloid leukemia etc. | Bristol Myers Squibb Co. | Orum Therapeutics, Inc. | Therapex Co., Ltd. | Phase 1 |
| CD33 CAR-NK cell (Hangzhou Qihan Biotechnology) | CD33 | CAR-NK | Refractory acute myeloid leukemia | Hangzhou Qihan Biotechnology Co., Ltd. | Phase 1 |
| JNJ-89853413 | CD3 x CD33 x TCRs | Bispecific T-cell engager | Refractory acute myeloid leukemia | Myelodysplastic syndromes | Recurrent acute myeloid leukemia | Hematologic malignancies | Janssen Research & Development LLC | LAVA Therapeutics NV | Phase 1 |
| Anti-CD33/anti-FcgammaRI bispecific antibody (University of California San Diego) | CD33 x CD64 | Bispecific antibody | Acute myeloid leukemia | University of California San Diego | Phase 1 |
| LB1910 | CD33 x CLL-1 | CAR-T | Acute myeloid leukemia | Legend Biotech (Nanjing) Co., Ltd. | Phase 1 |
| CD33/CLL1 dual CAR-NK cells (Hangzhou Qihan Biotech) | CD33 x CLL-1 | CAR-NK | Refractory acute myeloid leukemia | Hangzhou Qihan Biotechnology Co., Ltd. | Hangzhou Qihan Jiyin Biotechnology Co., Ltd. | Phase 1 |
| Anti-CD33 CAR-NK cell therapy (Sichuan Kelun-Biotech Biopharmaceutical) | CD33 | CAR-NK | Refractory acute myeloid leukemia | Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. | Phase 1 |
| BECAR33 | CD33 | Universal CAR-T | Recurrent acute myeloid leukemia | Great Ormond Street Hospital for Children NHS Foundation Trust | Phase 1 |
| CD33 CAR-T (Zhejiang University) | CD33 | CAR-T | Recurrent acute myeloid leukemia | Zhejiang University | Phase 1 |
| PRGN-3006 | CD33 | Autologous CAR-T | Acute myeloid leukemia | CD33-positive acute myeloid leukemia | Myelodysplastic syndromes | Precigen, Inc. | Precigen ActoBio, Inc. | Phase 1 |
| CART33 (University of Pennsylvania) | CD33 | Universal CAR-T | Acute myeloid leukemia | University of Pennsylvania | Phase 1 |
| SC-DARIC33 | CD33 | Autologous CAR-T | Recurrent acute myeloid leukemia | Acute myeloid leukemia | bluebird bio, Inc. | Seattle Children's Hospital | Regeneron Pharmaceuticals, Inc. | Phase 1 |
| TTX-564 | CD3 x CD33 | Bispecific T-cell engager | Myelodysplastic syndromes | Solid tumors | Trueline Therapeutics, Inc. | Phase 1 |
| CD33-NKE | CD33 | NK cell engager | Relapsed multiple myeloma | Sino-American Shanghai Squibb Pharmaceuticals Ltd. (Note: likely meant Bristol Myers Squibb (China) entity) | Phase 1 |
| Anti-CD33 CAR T-cells (City of Hope Medical Center) | CD33 | Universal CAR-T | CD33-positive acute myeloid leukemia | Refractory acute myeloid leukemia | City of Hope National Medical Center | Phase 1 |
| SENTI-202 | CD33 x FLT3 | CAR-NK | Acute myeloid leukemia with FLT3/ITD mutation | Myelodysplastic syndromes | Recurrent acute myeloid leukemia | Refractory acute myeloid leukemia | Senti Sub I, Inc. | Phase 1 |
| CC-96191 | CD33 | CAR-NK | Acute myeloid leukemia | Dragonfly Therapeutics, Inc. | Phase 1 |
| RJK-RT2831 | CD123 x CD33 | Bispecific antibody | Refractory acute myeloid leukemia | Acute myeloid leukemia | Acute promyelocytic leukemia | Hematologic malignancies | Nanjing Rongjiekang Biotechnology Co., Ltd. | Phase 1 |
| OXS-C3550 | CD16a x CD33 x IL15R | Immune-stimulating antibody conjugate (ISAC) | Acute myeloid leukemia | Myelodysplastic syndromes | Myeloid neoplasms | Refractory acute myeloid leukemia | University of Minnesota | GT Biopharma, Inc. | Phase 1 |
| DXC-007 | CD33 x Tubulin | ADC | Refractory acute myeloid leukemia | Recurrent acute myeloid leukemia | Hangzhou Duoxi Biotechnology Co., Ltd. | Phase 1 |
| CD33 CAR-T Cells (Shenzhen Geno-Immune Medical) | CD33 | CAR-T | Refractory acute myeloid leukemia | Acute lymphoblastic leukemia | Refractory anemia with excess blasts | Refractory leukemia | Shenzhen Geno-Immune Medical Institute | Phase 1 |
| Anti-CD33 CAR-T (Beijing Gaobo Boren Hospital) | CD33 | Autologous CAR-T | Refractory acute myeloid leukemia | Beijing Gaobo Boren Hospital Co., Ltd. | Phase 1 |
| IBR-733 | CD33 x CLL-1 | CAR-NK | Acute myeloid leukemia | Inbioray (Hangzhou) Biomedicine Co., Ltd. | Phase 1 |
(Data as of March 16, 2026, sourced from synapse)
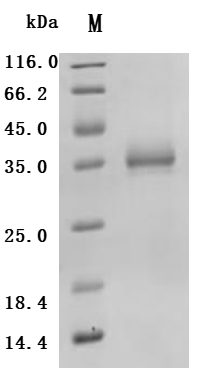
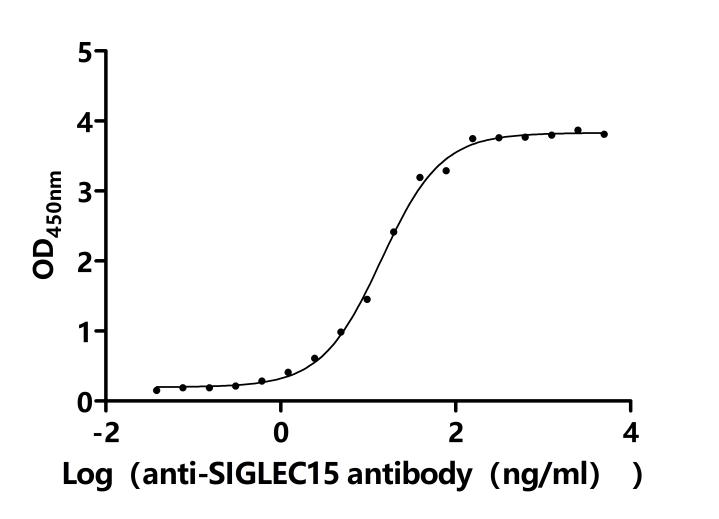
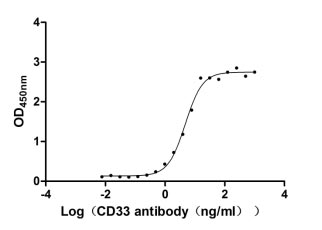
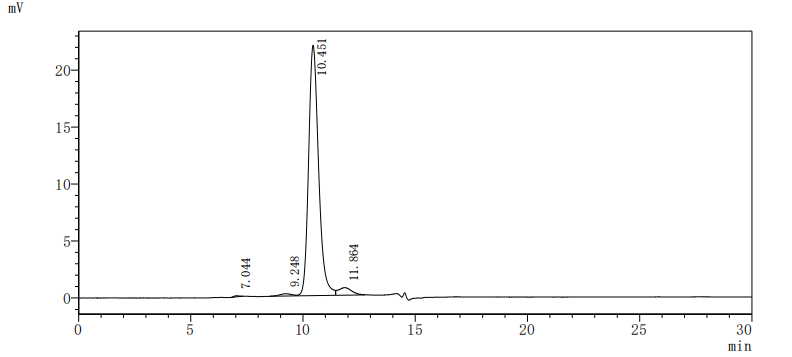
6. CD33-Related Research Tools: Recombinant Proteins, Antibodies, and ELISA Kits
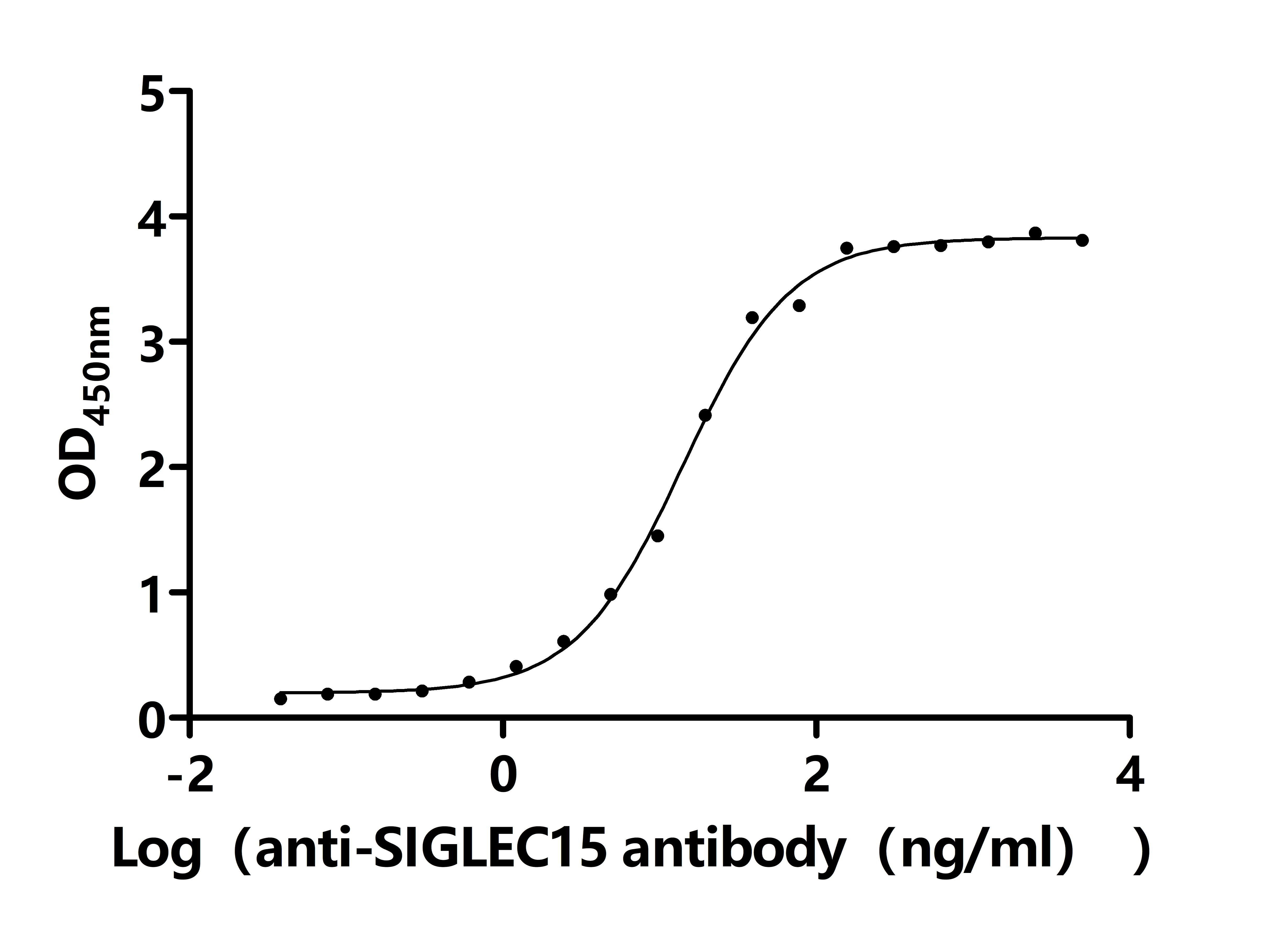
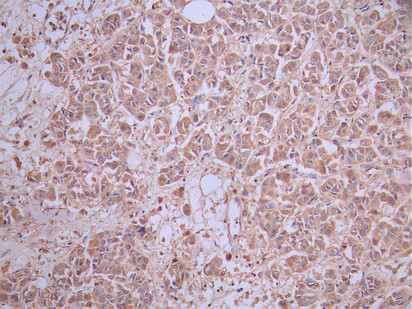
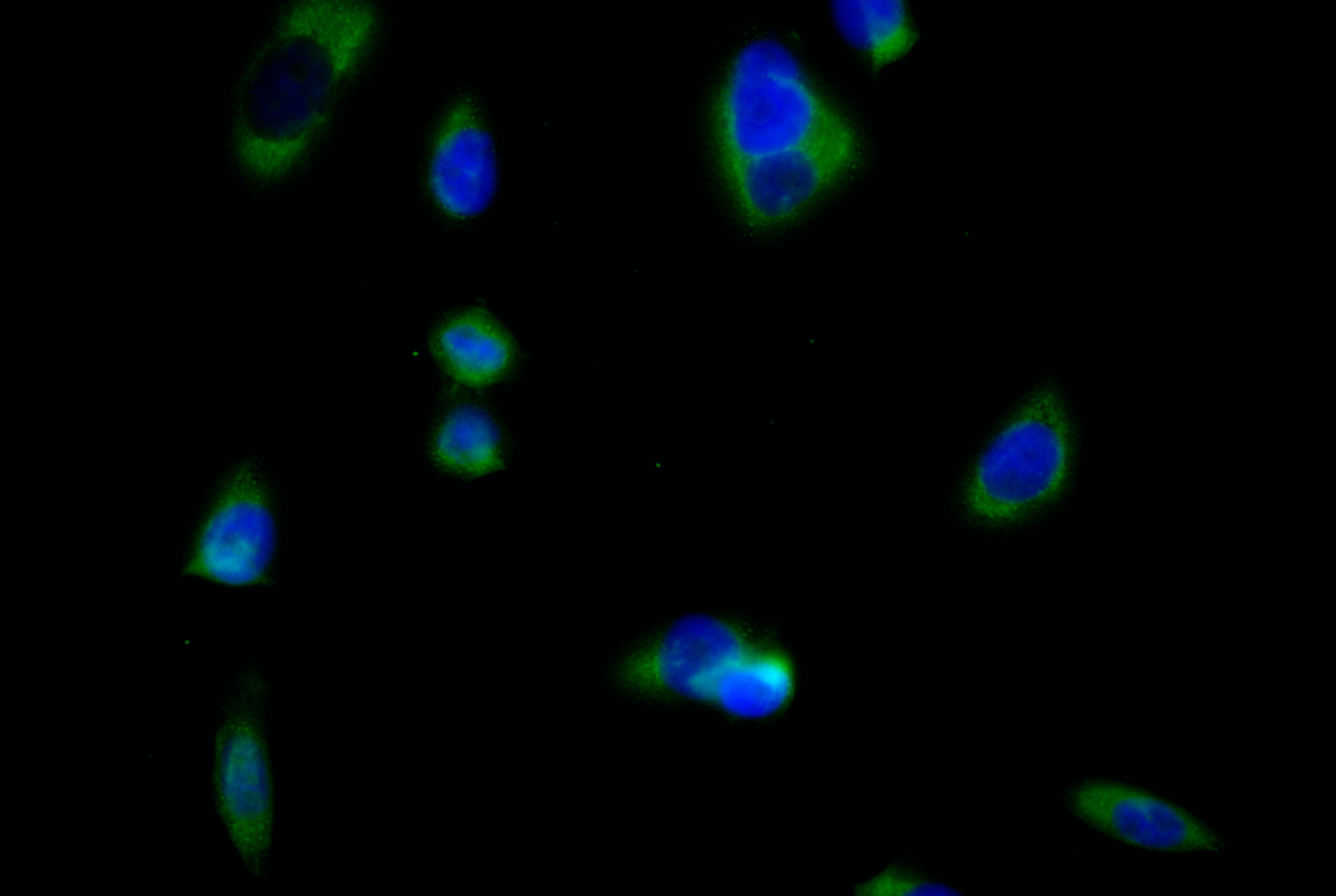
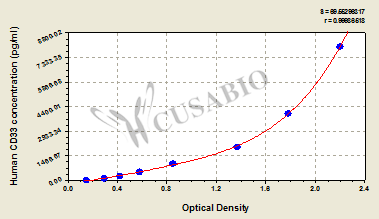
CD33 is an important member of the Siglec family and plays a key role in myeloid cell immune regulation. Its ITIM-mediated inhibitory signaling pathway can regulate inflammatory responses, cell phagocytic function, and immune homeostasis. Recent studies have shown that CD33 not only has important clinical value in acute myeloid leukemia but also plays crucial roles in Alzheimer's disease, chronic viral infections, and tumor immune regulation. Huamei Biotech provides CD33 recombinant proteins, antibodies, and ELISA kit products to assist you in related mechanism research and targeted drug development.
References
[1] E. B. D. Brinkman-van der Linden, T. Angata, S. Reynolds, L. Powell, S. Hedrick, A. Varki.(2003). CD33/Siglec-3 Binding Specificity, Expression Pattern, and Consequences of Gene Deletion in Mice.
[2] Gena S. Whitney, Shulin Wang, Han Chang, Ke-Yi Cheng, Pin Lu, Xia D. Zhou, Wen-Pin Yang, Murray McKinnon, Malinda Longphre.(2001). A new siglec family member, siglec-10, is expressed in cells of the immune system and has signaling properties similar to CD33.
[3] Yi-Jyun Lin, Pei-Shan Sung, Shie-Liang Hsieh.(2024). SIGLEC-3 (CD33) serves as an effective target in tumor progression.
[4] Mara Gilardi, Garima Kaushik, Brandon Walling, V. Sambandam, Paolo Schiavini, Stefano Cairo, Marianna Zipeto, Michael Ritchie.(2023). Correlation Revealed between Gemtuzumab-Ozogamicin Efficacy and CD33+ Expression in AML Primary Samples through a Novel AML in Vitro Model.
[5] Yoon-Chi Han, Jennifer Kahler, Nicole Piché-Nicholas, Wenyue Hu, S. Thibault, Fan Jiang, M. Leal, M. Katragadda, A. Maderna, R. Dushin, Nadira Prashad, M. Charati, T. Clark, L. N. Tumey, X. Tan, A. Giannakou, E. Rosfjord, H. Gerber, Lioudmila Tchistiakova, F. Loganzo, Christopher J. O'Donnell, P. Sapra.(2020). Development of Highly Optimized Antibody--Drug Conjugates against CD33 and CD123 for Acute Myeloid Leukemia.
CUSABIO team. CD33 (Siglec-3) Signaling Pathways and Disease Mechanisms: Roles in Acute Myeloid Leukemia, Alzheimer’s Disease and Immune Regulation. https://www.cusabio.com/c-21306.html




Comments
Leave a Comment