The SARS-CoV-2 spike glycoprotein represents one of the most intensively studied viral proteins in recent history, serving as the primary mediator of host cell entry and the principal target for neutralizing antibodies and vaccine development. The receptor-binding domain within the S1 subunit directly engages human ACE2, making it a critical focus for understanding viral infectivity, immune evasion, and therapeutic intervention strategies.
This recombinant monoclonal antibody against the SARS-CoV-2 spike protein offers researchers the reproducibility essential for longitudinal studies and diagnostic development. As a chimeric construct combining mouse variable regions with human kappa and IgG1 constant domains, it provides consistent performance across experiments while the recombinant production method ensures sequence-defined specificity that eliminates the lot-to-lot variability inherent in traditional hybridoma-derived antibodies.
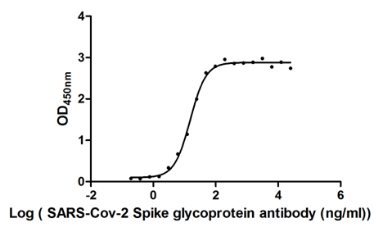
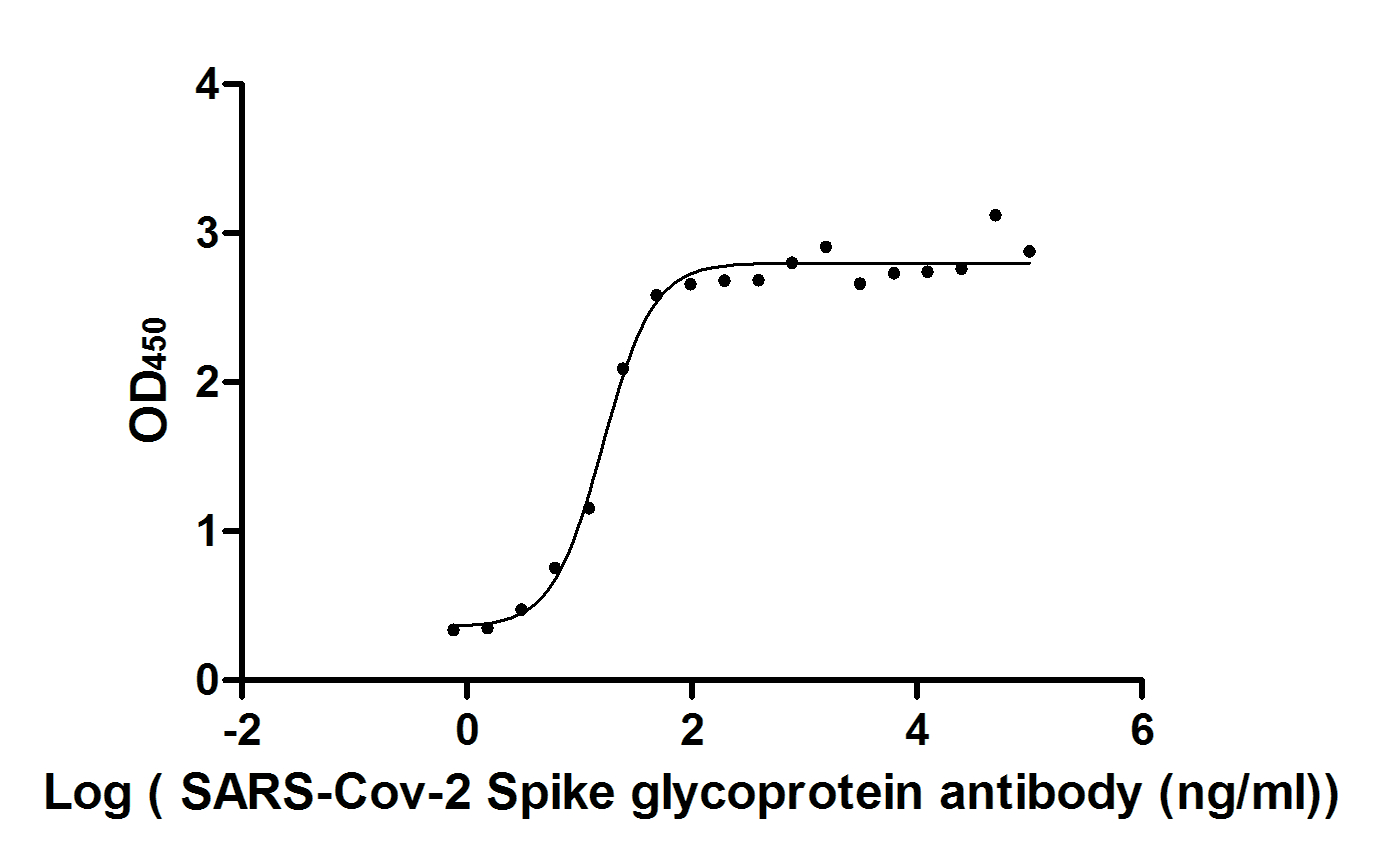
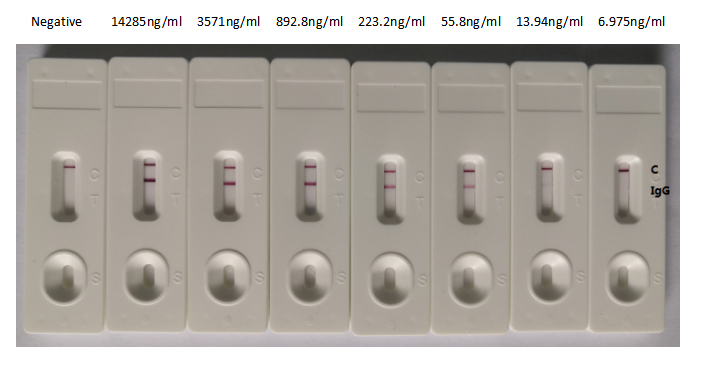
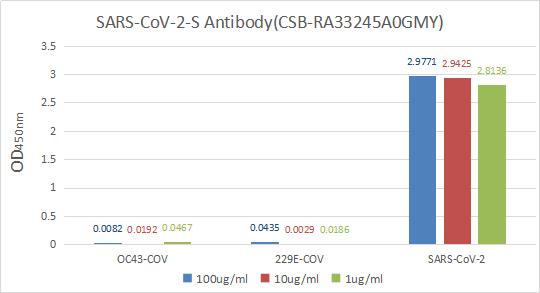
Functional validation demonstrates exceptional binding characteristics, with EC50 values of 15.29 ng/ml and 16.49 ng/ml against the S1-RBD in ELISA formats. Importantly, specificity testing confirms no cross-reactivity with seasonal coronavirus strains HCoV-OC43 and HCoV-229E, ensuring reliable discrimination of SARS-CoV-2 in mixed-pathogen research contexts. For rapid detection applications, colloidal gold immunochromatography assay validation shows clean background signal with detection limits reaching 13.94 ng/ml, demonstrating sensitivity suitable for point-of-care diagnostic development.
The antibody performs effectively in both ELISA at dilutions from 1:10,000 to 1:50,000 and gold immunochromatography assay formats at 1:500 to 1:25,000, providing flexibility for researchers developing quantitative assays or lateral flow platforms. This versatility makes it particularly valuable for COVID-19 surveillance research, antigen detection system development, and studies investigating spike protein biology and immune recognition.