Hafez RA et al. found that Egyptian patients' serum periostin is significantly related to asthma and positively associated with IL13 level supporting its utility as a diagnostic biomarker in a 2022 study [1]. Accumulating evidence has demonstrated that periostin is involved in the pathogenesis of asthma and other allergic disorders. Periostin is a pleiotropic molecule with multiple functions. As an extracellular matrix protein, periostin is secreted mainly by stromal cells and plays an essential role in tissue microenvironment remodeling and tumor metastasis during tumor progression. Herein will mainly introduce the functions of periostin in asthma and cancer.
1. What Is Periostin?
In 1993, Takeshita et al. cloned a functional bone adhesion molecule that can regulate the differentiation and adhesion of osteoblasts from the cDNA library of the mouse osteoblast cell line MC3r13-E1 and named it osteoblast-specific factor-2 (OSF-2) [2]. Later, Horiuchi et al. discovered this protein in the periosteum and periodontal ligament in 1999 and renamed it periostin (POSTN) [3].
POSTN is a secreted matricellular protein expressed in collagen-rich fibrous connective tissues, including skin, bone, heart valves, fascia of skeletal muscles, periodontal ligament, embryonic periosteum, placenta, adrenal glands, lung, stomach, colon, thyroid, prostate, ovary, breast, and testis. It is produced by epithelial cells, fibroblasts, and endothelial cells.
Generally, POSTN is expressed in adult healthy tissues at low levels, but it is upregulated when rapidly induced by various cytokines, growth factors, and mechanical stress during the pathological process.
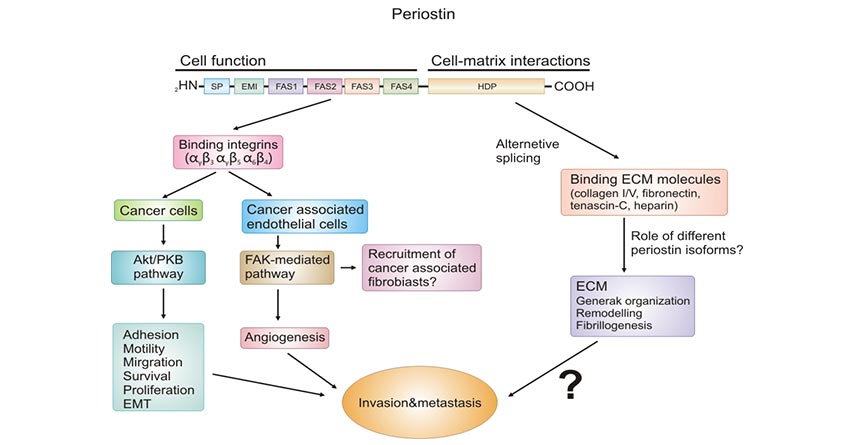
Structurally, POSN contains a typical signal peptide sequence (necessary for secretion) followed by a cysteine-rich EMI domain, a tandem of four repeated and conserved FAS domains (FAS1-4), and a C-terminal variable hydrophilic domain. The N-terminal EMI domain is important in multimerization in non-reducing conditions. POSTN binds to integrins and activates the Akt/PKB and the FAK-mediated signaling pathways via FAS domains, resulting in enhanced invasion and metastasis. C-terminal domains have four isoforms due to alternative splicing during the transcription process and regulate the composition and interaction of extracellular matrix by binding to extracellular matrix (ECM) proteins, such as collagen I/IV, fibronectin, tenascin C, and acid mucopolysaccharides [4].
Figure 1. The structure of POSTN and roles of each region
This picture is cited from: https://link.springer.com/article/10.1007/s00428-011-1151-5
2. What Are Functions of Periostin?
Through interaction with different integrins, such as αVβ3, αVβ5, and α6β4, POSTN mediates the cell-matrix interactions, cell adhesion, migration, survival, proliferation, epithelial-to-mesenchymal transition (EMT), collagen formation, fibrosis, and regeneration of cardiac myocytes after damage [5-7]. POSTN also participates in odontogenesis and osteogenesis [3].
In the skin, POSTN participates in chronic inflammation, fibrosis, and wound healing.
In blood vessels, POSTN is involved in vascular lesions and valve degeneration.
In kidneys, POSTN takes part in interstitial fibrosis and inflammation.
In the lungs, POSTN induces eosinophilic infiltration, bronchial hyperrectivity, and fibrosis.
In bones, POSTN is involved in bone formation, bone cortical response to mechanical loading and parathyroid hormone, and fracture healing.
In cancer cells, POSTN mainly contributes to creating a microenvironment that supports primary tumor and metastasis.
POSTN overexpression has been observed in different diseases characterized by inflammation, fibrosis, and tumorigenesis.
3. How Does Periostin Help Cancer Cells Spread?
Ilaria Malanchi et al. found that stromal POSTN is crucial for metastatic colonization by regulating the interactions between breast cancer stem cells and their metastatic niche [8]. Infiltrating tumor cells need to induce stromal POSTN expression in the secondary target organ (lung) to initiate colonization. POSTN has been shown to play an important role in the formation of myeloid-derived suppressor cells (MDSCs)-mediated immunosuppressive microenvironment for tumor metastasis.
Knockdown of POSTN significantly inhibits the recruitment of MDSCs to the pre-tumor microenvironment and subsequent formation of the immunosuppressive micro-environment, thereby suppressing lung metastasis of breast cancer. In other words, tumor stem cells cannot start transferring to the lung without POSTN. POSTN is required to allow cancer stem cell maintenance, and blocking its function can prevent cancer metastasis. Therefore, POSTN is considered the "helper" of breast cancer transferring to the lung.
Ample studies have shown that POSTN is highly expressed in diverse tumor types, including breast, colon, pancreatic, and ovarian cancers. POSTN upregulation has been associated with several mechanisms that drive carcinogeneses such as angiogenesis, cell survival, metastatic growth, cell motility, epithelial-mesenchymal transition (EMT), invasion, and adhesion.
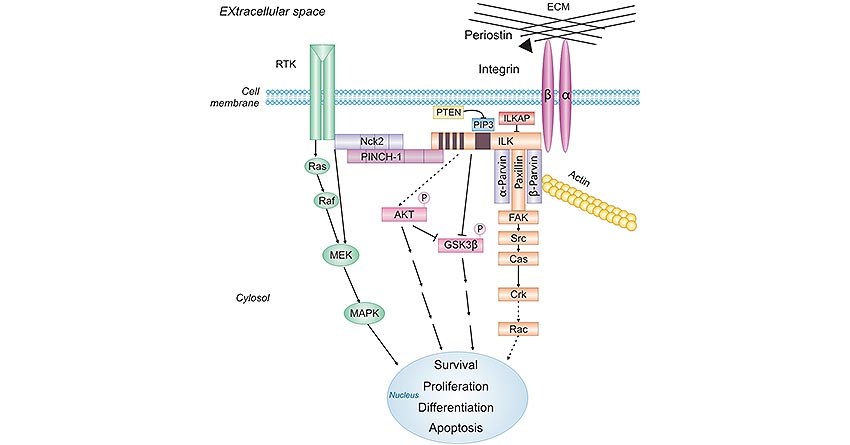
To implement these functions in cancer, POSTN binds to αvβ3, αvβ5, and α6β4 integrins and then initiates the crosstalk between those integrins and receptor tyrosine kinases (RTKs) such as EGFR at the plasma membrane, leading to the activation of the PI3K/Akt and FAK-mediated signaling pathways [9][10]. POSTN's role in ECM fibrillogenesis also might be conducive to its pro-tumorigenic effect.
Figure 2. POSTN-activated pathways during cancer progression
This picture is cited from: https://link.springer.com/article/10.1007/s00428-011-1151-5
However, a few studies reported that POSTN was downregulated in bladder and lung cancer [11][12].
4. What Is the Role of Periostin in Asthma?
The relationship between POSTN and allergy disorders was initially described in 2006 [13]. Additionally, less than ten years have passed since it was revealed in 2011 that serum POSTN can predict the efficacy of the neutralizing anti-IL-13 antibody-lebrikizumab in a phase 2 trial for patients with asthma, demonstrating for the first time the usefulness of POSTN as a biomarker for allergic diseases [14].
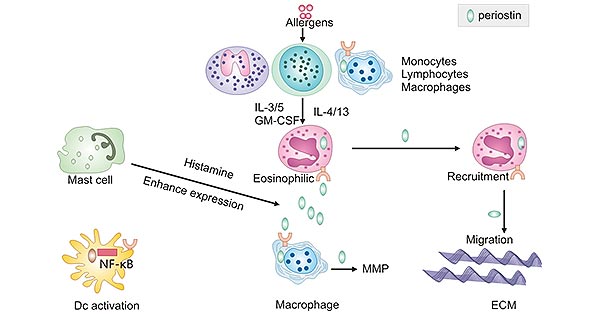
POSTN is involved in the pathogenic process of eosinophilic asthma. Allergens such as house dust mites, cat dander, and pollen activate and induce Th2 cells to produce IL-4 and IL-13, thereby stimulating eosinophils to generate POSTN. POSTN acts on eosinophils via αV integrin to stimulate the adhesion of purified human blood eosinophils, while also enhancing their recruitment to an asthmatic airway. This cascade results in a thickening airway wall, hyperplasia and hypertrophy of airway smooth muscle cells, mucus hypersecretion, fibroblast infiltration and proliferation, and bronchoconstriction, ultimately leading to asthma.
IL-5, IL-3, or GM-CSF can also stimulate the adhesion of purified human blood eosinophils, thereby leading to a vicious cycle. Macrophages produce POSTN, which enhances the secretion of matrix metalloproteinases (MMPs). Furthermore, in the presence of histamine, expression of POSTN by other cells is also enhanced.
Figure 3. The involvement of POSTN in the pathogenic process of eosinophilic asthma
This picture is cited from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4437675/
POSTN has also been shown to be involved in many aspects of asthma as well, including eosinophil recruitment [15], airway remodeling, establishment of a Th2 phenotype, and contributes to the enhanced expression of inflammatory mediators [16][17].
Additionally, POSTN is also a useful biomarker for atopic dermatitis, interstitial pneumonia, systemic sclerosis, and proliferative diabetic retinopathy.
CUSABIO provides some POSTN reagents for research use.
References
[1] Hafez RA, Hassan ME, et al. Association of Interleukin 13 rs20541 Gene Polymorphism and Serum Periostin with Asthma and Allergic Conjunctivitis Among Egyptian Patients [J]. 26 July 2022 Journal of Asthma and Allergy, Volume 2022:15, Pages: 971—982.
[2]Takeshita S, Kikuno R, Tezuka K, et al. Ostcoblast-specific factor 2: cloning of a putative bone adhesion protein with homology with the insect protein fasciclin I [J]. Bio Chem J,1993,294:271-278.
[3] Horiuch K, Amizuka N, Takeshita S, et al. Identification and characterization of a novel protein, with restricted expression to periosteum and periodontal ligament and increased expression by transforming growth factor β [J]. J Bone Miner Res, 1999,14:1239-1249.
[4] RA Norris, et al. Periostin regulates collagen fibrillogenesis and the biomechanical properties of connective tissues [J]. J Cell Biochem 101, 695–711 (2007).
[5] GW Dorn, Periostin and myocardial repair, regeneration, and recovery [J]. N Engl J Med 357, 1552–1554 (2007).
[6] JT Butcher, RA Norris, et al. Periostin promotes atrioventricular mesenchyme matrix invasion and remodeling mediated by integrin signaling through Rho/PI 3-kinase [J]. Dev Biol 302, 256–266 (2007).
[7] L Gillan, et al. Periostin secreted by epithelial ovarian carcinoma is a ligand for alpha(V)beta(3) and alpha(V)beta(5) integrins and promotes cell motility [J]. Cancer Res 62, 5358–5364 (2002).
[8] I. Malanchi, A. Santamaria-Martínez, et al. Interactions between cancer stem cells and their niche govern metastatic colonization [J]. Nature, 481 (2011), pp. 85-89.
[9] Bao S, Ouyang G, et al. Periostin potently promotes metastatic growth of colon cancer by augmenting cell survival via the Akt/PKB pathway [J]. Cancer Cell. 2004;5(4):329–39.
[10] Gillan L, Matei D, et al. Periostin secreted by epithelial ovarian carcinoma is a ligand for alpha(V)beta(3) and alpha(V)beta(5) integrins and promotes cell motility [J]. Cancer Res. 2002;62:5358–5364.
[11] Kim CJ, Yoshioka N, et al. (2005) Periostin is down-regulated in high grade human bladder cancers and suppresses in vitro cell invasiveness and in vivo metastasis of cancer cells [J]. Int J Cancer 117:51–58.
[12] Yoshioka N, Fuji S, et al. (2002) Suppression of anchorage-independent growth of human cancer cell lines by the TRIF52/periostin/OSF-2 gene [J]. Exp Cell Res 279:91–99.
[13] Takayama GO, Arima K, et al. Periostin: a novel component of subepithelial fibrosis of bronchial asthma downstream of IL-4 and IL-13 signals [J]. J Allergy Clin Immunol. 2006; 118: 98- 104.
[14] Corren J, Lemanske RF, et al. Lebrikizumab treatment in adults with asthma [J]. N Engl J Med. 2011; 365: 1088- 1098.
[15] Uchida M, Shiraishi H, et al. Periostin, a matricellular protein, plays a role in the induction of chemokines in pulmonary fibrosis [J]. Am J Respir Cell Mol Biol. 2012;46:677–86.
[16] Bentley JK, Chen Q, et al. Periostin is required for maximal airways inflammation and hyperresponsiveness in mice [J]. J Allergy Clin Immunol. 2014;134:1433–42.
[17] Yamaguchi Y. Periostin in skin tissue and skin-related diseases [J]. Allergol Int. 2014;63:161–70.
CUSABIO team. Periostin, the Accomplice of Cancer and Asthma. https://www.cusabio.com/c-19803.html



Comments
Leave a Comment