[1] Pezzutto A, Rabinovitch PS, et al. Role of the CD22 human B cell antigen
in B cell triggering by anti-immunoglobulin [J]. J Immunol. 1988, 140:1791–1795.
[2] Wilson GL, Fox CH, et al. cDNA cloning of the B cellmembrane protein
CD22: amediator of B-B cell interactions [J]. J ExpMed. 1991, 173:137–146.
[3] Thomas Dorner, Anthony Shock, et al. CD22 and Autoimmune Disease [J].
International Reviews of Immunology. 2012, 31:363–378.
[4] Tedder TF, Poe JC, et al. CD22: a multifunctional receptor that regulates
B lymphocyte survival and signal transduction [J]. Adv Immunol. 2005, 88:1-50.
[5] Daridon C, Blassfeld D, et al. Epratuzumab targeting of CD22 affects
adhesion molecule expression andmigration of B-cells in systemic lupus erythematosus
[J]. Arthritis ResTher. 2010, 12:R204.
[6] Wen T, Mingler MK, et al. The pan-B cell marker CD22 is expressed on
gastrointestinal eosinophils and negatively regulates tissue eosinophilia [J]. J
Immunol. 2012, 188:1075–1082.
[7] Engel P, Wagner N, et al. Identification of the ligand-binding domains of
CD22, a member of the immunoglobulin superfamily that uniquely binds a sialic
acid-dependent ligand [J]. J ExpMed. 1995, 181:1581–1586.
[8] Nath D, van derMerwe PA, et al. Theamino-terminal immunoglobulin like
domain of sialoadhesin contains the sialic acid binding site: comparison with CD22
[J]. J Biol Chem. 1995, 270:26184–26191.
[9] Wilson GL, Fox CH, et al. cDNA cloning of the B cell membrane protein
CD22: a mediator of B-B cell interactions [J]. J ExpMed. 1991, 173:137–146.
[10] Engel P, Nojima Y, et al. The same epitope on cD22 of B lymphocytes
mediates the adhesion of erythmcytes, T and B lymphocytes, neutrophils, and
monocytes [J]. J Immunol. 1993, 150(11):4719-4732.
[11] Ramya TN, Weerapana E, et al. In situ trans ligands of CD22 identi6ed by
glycan-protein photocross-linking-enabled proteomics [J].Mol Cell Pmteomics. 2010,
9(6):1339-1351.
[12] Smith KGC, Tarlinton DM, et al. Inhibition of the B cell by CD22: a
requirement for Lyn [J]. J ExpMed. 1998, 187:807-811.
[13] Xu Y, Harder KW, et al. Lyn tyrosine kinase: accentuating the positive
and the negative. Immunity [J]. 2005, 22:9–18.
[14] Doody GM, Justement LB, et al. A role in B cell activation for CD22 and
the protein tyrosine phosphatase SHP [J]. Science. 1995, 269:242–244.
[15] Gerlach J, Ghosh S, et al. B cell defects in SLP65/BLNK-deficient mice
can be partially corrected by the absence of CD22, an inhibitory coreceptor for BCR
signaling [J]. Eur J Immunol. 2003, 33:3418–3426.
[16] June Ereño-Orbea, Taylor Sicard, et al. Molecular basis of human CD22
function and therapeutic targeting [J]. Nature Communications. 2017, 8(764).
[17]Samardzie T, Marinkovic D, et al.Reduction of marginal zone B cells in
CD22-deficient mice [J]. Eur J Immunol. 2002, 32(2):561-567.
[18] Tooze RM, Doody GM, et al. Counterregulation by the coreceptom CDl9 and
CD22 of MAP kinase activation by membrane immunoglobulin [J]. Immunity. 1997,
7(1):59-67.
[19] O Keefe TL, Williams GT, et al. Hyperresponsive B cells in CD22-de6cient
mice [J]. Science. 1996, 274(5288):798-801.
[20] Nakiri Y, Minowa K, et al. E1pression of CD22 on peripheral B cells in
patients with rheumatoid arthritis: relation to CD5-positive B cells [J]. Clin
Rheumatol. 2007, 26(10):1721-1723.
[21] Thomas F. Tedder, Jonathan C. Poe, et al. CD22: A Multifunctional
Receptor That Regulates B Lymphocyte Survival and Signal Transduction [J]. Advances
in immunology. 2005, 88:1-50.
[22] Carnahan, J., Wang, P., et al. Epratuzumab, a humanized monoclonal
antibody targeting CD22: Characterization of in vitro properties [J]. Clin. Cancer
Res. 2003, 9:3982S–3990S.

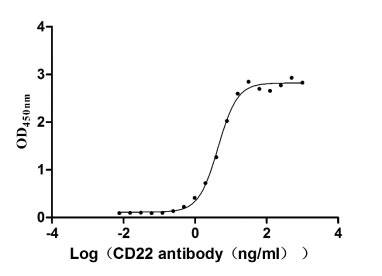


Comments
Leave a Comment