What are Aquaporins?
Aquaporin (AQPs) is a kind of membrane protein, which widely exists in prokaryotic and eukaryotic cell membranes and has the characteristics of highly selective transport of water molecules. In addition, AQP is also involved in various physiological and pathological processes of the body, including cell proliferation, apoptosis, migration, phagocytosis and nerve signal transduction.
The Discovery of Aquaporins
In 1985, Benga et al. first discovered the existence of water-permeating channel proteins in erythrocyte membrane. In 1988, Agre et al. discovered a transmembrane protein in the erythrocyte membrane while identifying the human Rh blood group antigen. The protein's molecular weight is 28 KD, so it named "channel-forming integral membrane protein (CHIP28)". Subsequently, Agre et al. identified the function of CHIP28 to transfer water and renamed it aquaporin1 (AQP1).
At present, A total of 13 AQP have been found in human bodies, which are AQP0-AQP12, belonging to the major intrinsic protein (MIP) family, composed of 250 ~ 295 amino acids.
Classification of Aquaporins
Aquaporins found in mammals can be divided into three subfamilies based on differences in gene structure and transport function.
The first subfamily is aquaporin, including AQP 0, AQP 1, AQP 2, AQP 4, AQP 5, AQP 6 and AQP 8. They are only permeable to water, except for AQP 6 and AQP 8. AQP 6 is permeable to anions, while AQP 8 is permeable to urea.
The second subfamily is aquaglyceroporin, including AQP 3, AQP 7, AQP 9 and AQP 10. The second subfamily can transport water and other small molecular solutes such as glycerin and urea.
The third subfamily is known as the superaquaporins, including AQP 11 and AQP 12.
Molecular Structure and Biochemical Properties of AQPs
The typical AQP gene structure contains a large exon (exon 1) encoding the amino terminus of AQP and three smaller exons (exons 2 ~ 4) encoding the hydroxy-terminus of AQP.
AQP1 is a glycoprotein. Its primary structure is a single peptide chain that spans the membrane 6 times. The amino and hydroxyl terminals are all located in the cell, and it contains 3 extracellular rings (A, C, E) and 2 intracellular rings (B, D). B ring and E ring are hydrophobic, and any variation will cause the decrease of the activity of aquaporin. The asparagine-valine-alanine (NPA) motifs located on the B and E rings are a common feature of members of the AQP family.
The tertiary structure of aquaporin is the "hourglass" model proposed by Jung et al. The hydrophobic B ring and E ring are located in and outside the cell respectively, and fold into the membrane lipid bilayer. The two NPAs are folded to form a single water channel, each of which is approximately a water molecule in size. The concentrated electrostatic field around the NPA prevents the passage of charged protons and other ions.
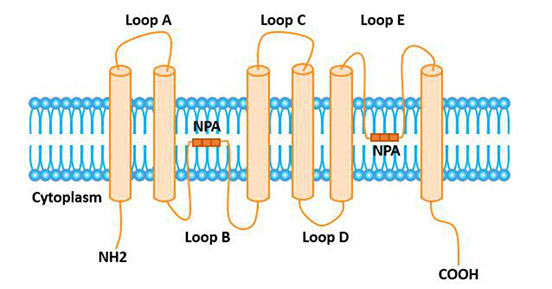
Figure 1 The structure of the AQP
Distribution of Aquaporins
Aquaporin is widely distributed in various tissues and organs of the body, including nervous system, cardiovascular system, respiratory system, kidney, digestive system and reproductive system.
Nervous system: Aquaporin is widely expressed in central nervous system and peripheral nervous system. Aquaporin in the brain are mainly AQP1, AQP4 and AQP9.
Cardiovascular system: Human heart expressions include AQP1, AQP3, AQP4, AQP5, AQP7, AQP9, AQP10 and AQP11.
Respiratory system: Four aquaporins, AQP1, AQP3, AQP4 and AQP5, are mainly expressed in the respiratory system.
Kidney: The reabsorption of water by the kidneys is mainly mediated by AQP, which in turn completes the urine concentration process. The AQP types in the kidney are mainly AQP1-4, AQP6-8 and AQP11.
Digestive system: The main functions of the digestive system include digestion and absorption, both of which require liquid transport across the cell membrane. Aquaporins in the digestive system include AQP1, AQP3, AQP4, AQP8, and AQP9.
Reproductive system: Recent studies on the human reproductive system AQP have shown that aquaporins play an important physiological role in the reproductive system. AQP is associated with diseases of the reproductive system, such as polycystic ovary syndrome, ovarian tumors, etc.
In addition, aquaporins are also distributed in the skin system and musculoskeletal system.
The Function of the AQP
With the isolation and identification of a large number of aquaporins, aquaporins have been found to be not only water-selective channel proteins, but also many other physiological and biochemical functions, and are a class of multifunctional proteins. The new functions of different members are mainly related to the cells they are located in.
-
For example, AQP1 is mainly located on vascular endothelial cells and lymphatic vascular endothelial cells, which are mainly involved in the formation of blood vessels and lymphatic vessels.
-
AQP3 located on the surface of keratinocytes and dendritic cells of the skin is associated with skin moisturization, innate immunity, and acquired immunity; mutations in AQP3 localized on renal tubular epithelial cells are associated with the pathogenesis of polycystic kidney disease.
-
AQP4 is mainly located in glial cells and ependymal cells in the central nervous system, which is involved in the occurrence and maintenance of cerebral edema.
-
AQP5 locates on the surface of ovarian epithelial cells, which is related to the occurrence of ovarian epithelial tumors.
-
AQP8 and AQP9 were located in liver cancer cells, and their decreased expression was related to their anti-apoptotic properties.
AQP plays an important role in the pathogenesis of many diseases and can be used as a new target for pharmacological treatment.
CUSABIO provides the products you need for your research. At present, we can produce aquaporin-related proteins, antibodies and kit products.