Nanodisc in Membrane Protein Research
Membrane proteins are proteins that are embedded in the lipid bilayer of cell membranes. They play important roles in cell signaling, transport, and cell-to-cell communication, and other functions. Therefore, membrane proteins have become one of the current research hotspots. However, studying these proteins can be challenging due to their hydrophobic nature, which can cause them to aggregate or denature when removed from their natural lipid environment.
Nanodiscs are small, disc-shaped structures that provide a way to study membrane proteins in a more natural environment, as they mimic the structure and properties of cell membranes. This makes them a valuable tool for studying the structure, function, and interactions of membrane proteins, as well as for drug discovery and development.
1. Composition and Structure of Nanodisc
Nanodiscs refer to small (7-50 nm in diameter) disk-like phospholipid bilayers consisting of two components: phospholipids, which can be synthetic phospholipids or native cell membrane phospholipids and stable belts that closely surround the phospholipids, including membrane scaffold proteins (MSPs) or synthetic polymers.
Compared with the reconstruction in micelle and liposome, integrating the membrane protein into the nanodisc can maintain its structure in the natural state to the maximum extent because the nanodisc is flat and produces no tension that affects the membrane protein conformation. The surface of the micelle and liposome is curved or spherical, which will inevitably affect the conformation of membrane proteins. Nanodisc is also sized to perform many biochemical, biophysical, and functional experiments.
Compared to other methods that study membrane proteins, nanodiscs are characterized by stabilization, flexibility, accessibility, mimicry, and compatibility.
2. Types of Nanodisc
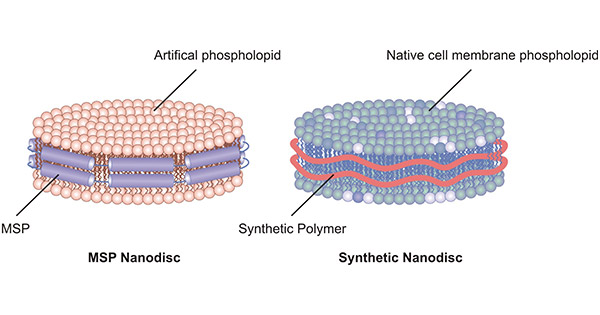
There are two types of nanodiscs: MSP nanodisc and synthetic nanodisc.
Figure 1. MSP nanodisc and synthetic nanodisc
2.1 MSP Nanodisc
MSPs closely wrap around the artificial phospholipids, resulting in the formation of MSP nanodisc. MSPs are truncated forms of apolipoprotein A-I (ApoA-I) and act as stabilizers. Each MSP protein contains 200 amino acid residues and is soft and malleable. MSP provides a hydrophobic surface facing the hydrophobic tail of the lipids, and a hydrophilic surface on the outside. This setup makes nanodiscs highly soluble in aqueous solutions. Once assembled into nanodiscs, membrane proteins can be kept in solution without detergents.
Among MSPs, MSP1D1 and MSP1EE3D1 are the two most widely used granules. The nanodiscs prepared by this method can maximize the preservation of the natural conformation of membrane proteins during purification for further study. The diameter of nanodiscs is determined by the length of the MSP band and when the phospholipid is in the optimum molar ratio to MSP, uniform nanodiscs are formed.
The preparation process of MSP nanodisc usually takes two forms:
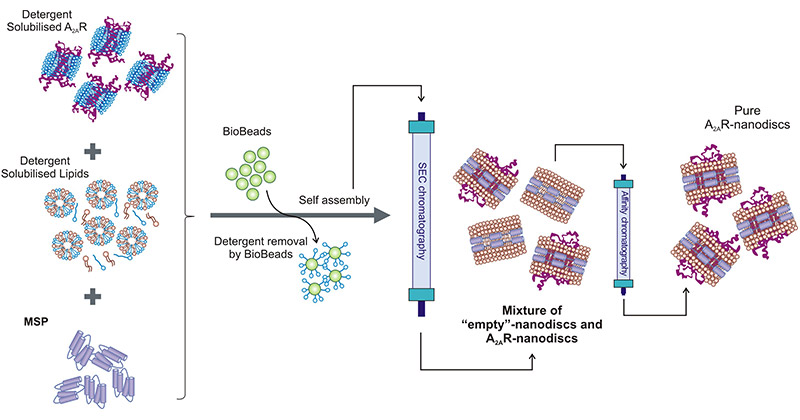
● Assembly of Membrane Proteins Dissolved in the Detergent
The membrane proteins are first purified in the presence of detergents and then spontaneously assemble into nanodiscs with the added MSPs and phospholipids. After removing the detergents by dialysis or hydrophobic adsorbents, the forming nanodiscs can be purified by dimensional exclusion chromatography or affinity purification.
Figure 2. Schematic of a standard nanodisc assembly protocol Nanodisc
This picture is cited from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7696381/
● Nanodisc Combination with and Cell-free Expression System
The assembled nanodiscs are added to the cell-free expression system. Multiple nanodiscs are obtained by introducing MSP particles into the mixture containing membrane proteins and adding excess lipids. The mixture of nanodiscs is purified to obtain nanodiscs only expressing the target membrane proteins. This method does not require the addition of detergents, which can minimize the presence of artifacts.
2.2 Synthetic Nanodisc
The assembly of a synthetic nanodisc begins with a complete cell. It uses synthetic polymers to dissolve cell membranes while using native cell phospholipids to form nanodisc structures around membrane proteins. The polymer acts as both a solvent enhancer and stabilizer. Therefore, no additional detergent is required.
Compared with the MSP nanodisc, synthetic nanodisc omits the screening process of detergent and phospholipid, thus shortening the development cycle. Synthetic nanodisc uses synthetic polymers that are non-protein components and therefore have a higher purity than MSP nanodiscs.
Differences between the two nanodiscs:
|
MSP Nanodisc |
Synthetic Nanodisc |
| Stabilizer |
MSP |
Synthetic polymer |
| Phospholipid Source |
Artificial phospholopid |
Native cell membrane |
| Preparation Method |
Membrane proteins with detergent self-assemble into nanodiscs or nanodiscs combination with cell-free expression system |
Synthetic polymers dissolve cell membrane |
| Whether or not need detergents |
Yes or no |
No |
| Cases |
Pre-assembled nanodisc |
Diisobutylene-maleic acid (DIBMA), styrene-maleic acid (SMA), amphipol |
3. Application of Nanodisc in Membrane Proteins
As a kind of artificial pseudo-membrane structure, nanodisc provides a better platform for the study of membrane proteins, which are affected by structural instability and inconvenient aggregation for a long time.
The size of the nanodisc can be adjusted to meet the expression requirements of the target membrane proteins of different sizes. Nanodiscs can provide a membrane-like environment with a long half-life, strong stability, high solubility, and precise targeting advantages, and have been widely used in proteomics and biomedicine fields, including the production and purification of membrane proteins, receptor- and ligand-binding drug target research, and structural biology.
3.1 Structural Study of Membrane Protein
Nanodiscs can be applied to stabilize membrane proteins for structural studies using techniques such as X-ray crystallography, electron microscopy, and nuclear magnetic resonance spectroscopy. In the absence of a stabilizing environment, membrane proteins can be unstable and prone to aggregation or denaturation, which can make structural studies difficult or impossible.
Nanodiscs provide a native-like environment that can maintain the integrity of the membrane proteins, while also providing a small, uniform size that is compatible with many structural techniques, including nuclear magnetic resonance (NMR), electron microscopy (EM), and X-ray crystallography.
Nanodiscs can also be modified with various chemical and biological tags to allow for easy purification and detection of the protein of interest.
3.2 Functional Study of Membrane Proteins
Nanodiscs are increasingly being used for functional studies of membrane proteins. One application of nanodiscs in functional studies is in the investigation of the activity of membrane transporters, ion channels, and receptors. For example, researchers can incorporate different lipid compositions or ligands into the nanodiscs to study the effect of these factors on the function of the membrane protein. Nanodiscs can also be used to study the kinetics of transport or binding, as well as the effects of mutations on protein function.
Nanodiscs can also be used to study the interaction of membrane proteins with other proteins or small molecules. For example, researchers can incorporate a binding partner or a small molecule into the nanodisc and study its interaction with the membrane protein. This can be useful in drug discovery, where the goal is to develop compounds that modulate the activity of a specific membrane protein.
3.3 Drug Carrier
As drug carriers, nanodiscs can effectively dissolve drugs with poor water solubility and load some drugs with low bioavailability, which strongly promotes the rapid development of drug diagnosis and therapeutics. At present, a large number of experiments have proved that nanodisc materials have good loading capacity for lipophilic drugs.
At the same time, the drug preparation constructed by the nano drug delivery system has a small size and narrow particle size distribution. After surface modification, the nanodisc-carrying drugs can carry out targeted specific positioning to achieve the purpose of targeted drug delivery. Besides, it can protect drug molecules, improve stability and biocompatibility, and combine imaging and treatment to realize disease diagnosis and treatment. Great progress has been made in the application of chemotherapy in the treatment of tumor diseases.
So far, nanodiscs as drug carriers have made great progress in application research due to their special advantages, such as drug-controlled release, targeting function, high drug loading rate, etc. However, at present, relevant tests are only carried out in animal models. Further exploration and verification are needed before they are expected to better play their advantages as drug carriers in clinical use.
Meanwhile, in the field of clinical medicine, nanodiscs have also made important contributions to cardiovascular disease treatment, tumor therapy in combination with chemotherapy immunotherapy, and vaccine preparation.
Table1. Methods and tools used with membrane proteins incorporated into Nanodiscs [1]
| Features and applications |
Experimental methods |
Membrane proteins |
| Solubilization and stabilization |
X-ray crystallography |
GPCR |
| NMR |
Various membrane proteins |
| Resonance Raman |
Cytochrome P450, rhodopsins |
| Optical absorption |
| EPR |
Transporters, rhodopsin |
| Cell-free expression |
Various |
| Surface immobilization using affinity tags on scaffold protein or lipids |
Single-molecule studies SPR, LSPR, sensing and detection |
P450, GPCR, Factor VII |
| Isolation of monomers, oligomers, and functional complexes of multiple proteins |
Structural and functional studies |
Receptors, rhodopsins, cytochrome P450 with CPR, ATPase |
| Structurally homogeneous preparations |
SAXS, SANS, EM |
Cytochrome P450 and reductase Receptors, channels |
| Fractionation by size, charge, density. |
Chromatography, Electrophoresis, Ultracentrifugation |
|
| Protein-membrane binding |
SPR |
K-ras protein, TF:VIIa, PgP
transporter |
| Membrane effect on protein properties |
Redox potential, Kinetics |
Cytochrome P450 reductase Factor VIII Respiratory Complex |
4. Nanodisc Application Case Study by CUSABIO
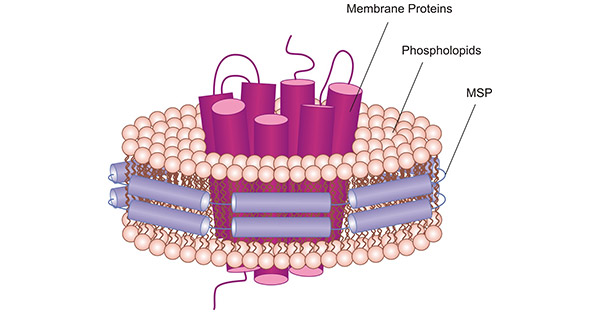
Cusabio incorporates both in vitro E.coli protein expression and nanodiscs technology, adding pre-assembled nanodiscs to in vitro E.coli protein expression systems. The membrane proteins are assembled into nanodiscs while they are translated, forming membrane protein-nanodisc complexes (Figure 3). This simulated artificial lipid environment provides new avenues for analyzing the effects of different lipids on membrane proteins, allowing us to better understand membrane proteins.
Figure 3. Schematic illustration of a MSP nanodiscs with a 7-transmembrane protein embedded.
CUSABIO provides some membrane proteins expressed through nanodisc and in vitro E.coli expression system, as follows:
In summary, nanodiscs are novel membrane-like structures that mimic the native phospholipid bilayer of cells for target membrane proteins whilst maintaining the structure and conformation of membrane proteins in a physiologically relevant state. Nanodiscs have become an essential tool for the study of membrane proteins, allowing researchers to better understand the roles these proteins play in biological systems and to develop new therapies for diseases that involve membrane proteins.
References
[1] Ilia G. Denisov and Stephen G. Sligar Nanodiscs for structural studies of membrane proteins [J]. Nat Struct Mol Biol. 2016 Jun; 23(6): 481–486.