Detergents Applications in Membrane Proteins Research
In recent years, membrane protein research has made significant progress, which is inseparable from the development of membrane-related tools and reagents [1]. Among them, detergents play an important role in the extraction, solubilization, purification, physicochemical properties, and structure study of membrane proteins.
However, how to choose the appropriate detergent is often a complex task. This article mainly describes the important physicochemical properties of detergents and their applications in the study of membrane protein structure and function.
1. Structure of Detergents
Detergents, also known as surfactants, are amphiphilic molecules with hydrophilic polar groups and hydrophobic non-polar groups. They can be quantitatively analyzed by thin-layer chromatography or gas chromatography. Their amphiphilic nature enables them to solubilize hydrophobic membrane proteins from lipid membranes, providing hydrophobic protection for membranes in hydrophilic solutions thus maintaining membrane proteins' native transmembrane structure in a lipid-free environment.
Each detergent molecule has a hydrophilic (polar) head and a long hydrophobic (nonpolar) tail. The hydrophobic portion may be linear or branched hydrocarbons or may have a steroidal structure. The hydrophilic parts are more diverse, they can be ionic or non-ionic, and can be simple structures or relatively complex structures. Their dual nature facilitates the mixing of hydrophobic compounds such as oils and greases with water.
Some detergents also have lenticular shapes, which have polar and non-polar faces, including bile acid derivatives, such as CHAPS and CHAPSO.
2. Classification of Detergents
According to the electrical charge of the surfactants, detergents can be divided into ionic (cationic or anionic), nonionic, and amphoteric detergents.
The ionic detergents include sodium dodecyl sulfate (SDS), N-lauryl sarcosine, CTAB, etc., which are effective in extracting proteins from the membrane. These detergents can effectively disrupt the interaction between intramolecular and intermolecular proteins, but are harsh and tend to denature proteins. Among them, bile acid salts are also ionic detergents, such as sodium cholate and deoxycholic acid, but their skeletons are composed of rigid steroids, which are milder than linear ionic detergents.
Nonionic detergents include maltosides, glucosides, and polyoxyethylene glycols. The feature of this type of detergent is that the hydrophilic head groups are not charged. These detergents are mild, non-denatured, and can disrupt interactions between protein-lipid and lipid-lipid.
The amphoteric detergents include Zwittergents, Fos-Cholines, CHAPS /CHAPSO, etc. Their hydrophilic head groups contain positive and negative charges. These detergents are electrically neutral like nonionic detergents, but they are usually able to disrupt the interaction between proteins like ionic detergents, so intermedium mild. Most successful NMR membrane protein studies use zwitterionic detergents, for example, Fos-Choline 12.
3. Properties of Detergents
Detergents have three key properties: micellization, critical micelle concentration, and cloud point. These characteristics enable detergents to exert important roles in the applications of membrane protein.
3.1 Micellization
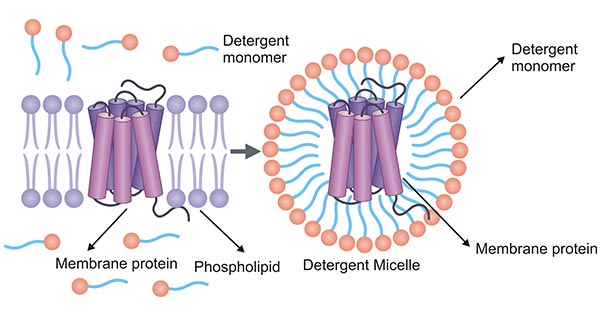
Micelles are aggregates of the detergent monomer in the solution, and the micelle forming process is called micellization [2]. Detergent interacts with the membrane protein and membrane in the form of micelles, and the dissolution of the protein depends on the formation of micelles in the solution. Micelles are usually considered to have a "rough" surface, which is a dynamic structure. The detergent monomer in the micelles rapidly exchanges with the free detergent monomer in the solution. Once the membrane protein is dissolved, we usually think that the detergent molecules form a torus around the hydrophobic transmembrane domain.
Figure 1. Detergent-membrane protein micelle
3.2 The Critical Micelle Concentration (CMC)
CMC is the optimal concentration of the detergent when micelles start to form. The CMC of a particular detergent also depends on several other factors like temperature of solubilization, buffer pH, and ionic strength [3].
Each kind of detergent has a CMC value, when the detergent concentration is higher than CMC, the monomer is self-assembled into a non-covalent aggregate, also called micelles. The micellization does not actually occur at a single concentration but occurs within a narrow concentration range.
When applying detergents to membrane proteins, one rule of thumb is that the working concentration of the detergent should be at least 2xCMC and the weight ratio of detergent to protein is at least 4: 1. When the membrane protein is dissolved from the original membrane, the working concentration of the detergent is much higher than that of CMC, and the molar ratio of detergent to lipid is 10: 1. Therefore CMC determines the amount of detergent to be added to various proteins and membrane products [4].
3.3 Cloud Point
Cloud point refers to the temperature at which detergent solution is separated into two phases when the concentration is close to or higher than CMC. The micelle aggregates to form a turbid phase with high detergent concentration, and the detergent will be exhausted when the solution reaches the equilibrium point. The obtained two-phase solution can be separated, and the extracted protein is located in the phase rich in detergent.
Because a high cloud point temperature will cause denaturation of dissolved protein, it is recommended to use a detergent with a low cloud point in the presence of protein. The cloud point is affected by factors such as changes in detergent concentration and temperature, and the addition of salts or polymers, such as dextran and polyethylene glycol. The cloud point is affected by factors such as changes in detergent concentration and temperature, and the addition of salts or polymers, such as dextran and polyethylene glycol.
4. Applications of Detergents in Membrane Proteins
The application of detergents in the study of membrane proteins mainly involves the in vitro expression, separation and purification, and structure research of membrane proteins.
In the process of membrane protein research, detergents can disintegrate lipid membranes and release membrane protein, and maintain the stability of structure and function of membrane protein in the state of membrane removal. The state and the behavior of detergents in solution are related to their own concentration, purity, non-polar water transport groups, pH and ionic strength of the solution, temperature, and other organic matter in the solution.
4.1 Detergents used in solubilization of membrane proteins case study
When the detergent interacts with the lipid membrane containing membrane proteins, the membrane protein can be released from the membrane lipid by competing with and weakening the hydrophobic binding between the membrane protein and the membrane lipid molecules.
Studies on cytochrome c showed that there were many lipid molecule-specific binding sites on the surface of membrane proteins. Detergents could replace lipid molecules to bind to these sites on the surface of membrane proteins and maintain the hydrophobic structure of membrane proteins.
The solubilization efficiency of the detergent on membrane protein is related to the structure of detergents and the critical micelle concentration, which can be optimized by changing the type and concentration of the detergent.
When selecting a detergent for the study of membrane proteins, it is not only necessary to improve the solubilization efficiency of the detergent, but also to consider reducing the concentration of the detergent below its critical micelle concentration so that the membrane proteins can be removed from the effect of the detergent and reassembled on the lipid membrane for structural and functional study.
Cusabio adopts E.coli cell-free expression technology. The technology is not limited by cell structure and is suitable for the expression of membrane proteins and toxic proteins that are toxic to cells, with a yield of up to mg/ml level. The open E.coli cell-free expression system can optimize expression yield in vitro in multiple ways, and the expressed membrane proteins can be immediately enveloped by the detergents after translation during the expression, to maximum avoid exposure to the aqueous solution. Now we have already successfully developed the following active aquaporins.
● Recombinant Escherichia coli Aquaporin Z (aqpZ)
Function: Channel that permits osmotically driven movement of water in both directions. It is involved in osmoregulation and in the maintenance of cell turgor during volume expansion in rapidly growing cells. It mediates the rapid entry or exit of water in response to abrupt changes in osmolarity.
Figure 2. aqpZ in detergent micelles
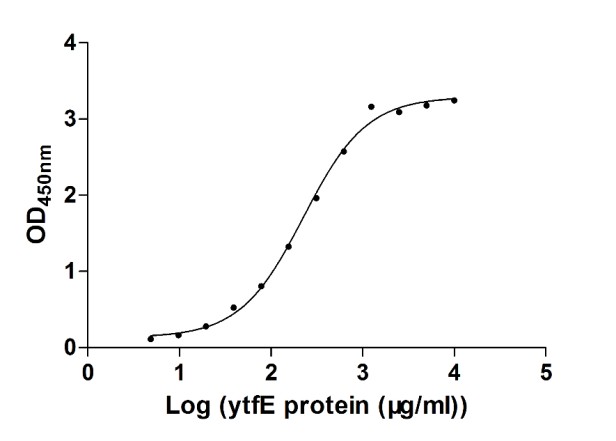
Figure 3. The binding activity of aqpZ with ytfE
Activity: Measured by its binding ability in a functional ELISA. Immobilized aqpZ at 5 μg/ml can bind human ytfE.
The EC50 of human ytfE protein is 197.90-259.70 μg/ml.
4.2 Detergents used in membrane proteins isolation and purification
Recombinant membrane proteins expressed by in vivo expression systems usually occur on the plasma membrane or in inclusion bodies. Membrane proteins expressed on the plasma membrane can be purified by a fusion affinity labeling system. After the membrane proteins have been dissolved from the plasma membrane using a suitable detergent, the affinity purification test can be performed by adsorption of free membrane proteins using affinity chromatography columns or affinity media.
The purification procedure is basically similar to that for soluble proteins, but the detergent in the solution needs to be kept above the critical micelle concentration to maintain and stabilize the membrane protein structure.
For membrane proteins expressed in an in vitro system, if the system does not contain lipid molecules or detergent, the membrane proteins will exist as aggregates. Due to the different solubilization efficiency of membrane proteins in different detergents, the pretreatment of protein aggregates with different detergents during the purification of target membrane proteins can remove and reduce the contamination of impurity proteins to a certain extent.
Membrane protein isolation and purification can sometimes be affected by the detergent. For example, during affinity purification of recombinant membrane protein containing maltose-bing protein (MBP) fusion label, detergent-containing maltose, such as DDM, may compete with affinity medium for maltose binding sites on MBP protein, thus reducing purification efficiency.
It was found that different detergents could significantly affect the activity of transporter Tytl. For different detergents with maltopyranoside polar groups, the activity of Tytl was related to the length of the non-polar hydrocarbon chain of the detergent. However, other detergents such as LDAO, C12E8, or detergents with polar groups of glucopyranoside can inhibit the activity of Tytl. Triton X-100 is the only detergent that does not contain polar groups of maltopyranoside and can maintain a certain degree of Tytl activity.
5. Transmembrane Proteins Developed on Detergent Micelle Platform
Membrane proteins are embedded in the phospholipid bilayer of the cell membrane to bridge the connection between the inside and outside of the cell membrane environment. The areas exposed to membrane proteins outside the cell are potential targets for many drug effects. It has been estimated that 60% of drug targets are membrane proteins. Full-length membrane proteins are valuable in research and development, but difficult to express and purify.
The conventional membrane protein preparation process has the characteristics of low protein content, low yield, low activity, and a complex purification process. Due to the obvious tendency of aggregation and denaturation of membrane proteins in an aqueous solution, it is difficult to simulate the membrane-like environment that maintains the correct conformation of membrane proteins in vitro. As a result, the research on the structure and function of membrane proteins and related ligand drugs lags far behind that of water-soluble proteins.
Therefore, the membrane protein-detergent technology platform helps maintain membrane proteins' conformation in solution during the separation and purification process. Suitable detergents can extract membrane proteins and effectively increase the water solubility of multiple transmembrane proteins.
In an aqueous solution, the cell membrane exists in the form of the lipid bilayer, and detergent molecules exist in the form of micelles at higher than the critical micellar concentration (CMC). When the cell membrane and detergent micelle are mixed, detergent molecules first bind to the surface of the cell membrane in a non-synergistic way.
With the increase of detergent quantity, detergent molecules are incorporated from the surface of the cell membrane into the interior of the lipid bilayer, changing the non-synergistic binding to synergistic binding. The synergistic and non-synergistic binding of detergents to cell membranes is called the transbilayer mechanism.
As more and more detergent is incorporated into the membrane, the lipid-detergent mixed micelles form. The membrane is lysed, and the detergent successfully extracts membrane proteins to form micelles. Increasing water solubility can also effectively help the membrane proteins to form a naturally correct conformation in solution.
References
[1] R.M. Garavito, S. Ferguson-Miller, Detergents as tools in membrane biochemistry, J. Biol. Chem. 276 (2001) 32403–32406.
[2] From Wikipedia, the free encyclopedia.
[3] M. le Maire, P. Champeil, J.V. Mbller, Interaction of membrane proteins and lipids with solubilizing detergents, Biochim. Biophys.Acta 1508 (2000) 86–111.
[4] Anatrace, Detergents and Their Uses in Membrane Protein Science.