Recent years, increasing evidence has showed that transmembrane proteins (also known as multi-pass transmembrane proteins) are great potential targets of antibody drugs. Many human diseases are associated with aberrant function of transmembrane protein. The versatility of transmembrane proteins makes them ideal drug targets, such as PD-L1, CCR8, Claudin-18.2, etc. However, due to the complex structure of multiple transmembrane proteins, it is difficult to prepare high-quality antigens and to design functional experiments. Because High-quality antigens are a key factor in the production of precise antibodies. how to obtain the full-length and natural intact conformation of transmembrane proteins is an urgent problem to be solved in the pre-drug development stage. So what is multi-transmembrane protein? What technology platforms are currently available to express multi-transmembrane proteins?
1. What is Multi-Transmembrane Protein?
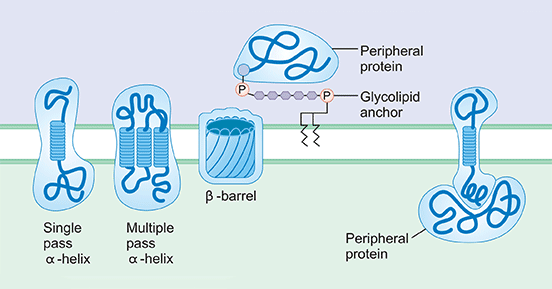
Before understanding multi-transmembrane protein, let's briefly first understand the relationship between transmembrane protein and membrane protein. Transmembrane protein (TP, also known as integral membrane protein) together with peripheral protein make up membrane protein.
As the figure 1 shows, peripheral proteins are distributed on the inner and outer surfaces of the membrane, accounting for about 20% to 30% of the membrane proteins. These proteins are mainly on the inner surface and are water-soluble proteins that bind to the polar heads of membrane lipid molecules through ionic bonds, hydrogen bonds, or indirectly bind to the membrane through interactions with intrinsic proteins.
Transmembrane proteins account for about 70% to 80% of membrane proteins. They are amphiphilic molecules that run through the entire lipid bilayer, with both ends exposed on the inner and outer surfaces of the membrane. The part of the inner membrane protein exposed outside the membrane contains more polar amino acids, which are hydrophilic and are adjacent to the hydrophilic head of the phospholipid molecule; the membrane protein embedded in the lipid bilayer is composed of some non-polar amino acids, which are closely related to the hydrophobic tails of the lipid molecules and are therefore very tightly bound to the membrane. It is estimated that 1/4~1/3 of proteins encoded by human gene are transmembrane proteins.
Figure 1. a diagram of different membrane proteins
Multi-transmembrane proteins, also known as muti-pass membrane proteins, refer to proteins that contain multiple transmembrane domains on the membrane. The interaction of the transmembrane region is an important channel to connect the extramembrane environment and the intracellular environment to perform various activation and response reactions. Signal transduction regulates morphological and functional changes inside and outside cells.
2. What Technology Platforms are Currently Available to Express Multi-Transmembrane Proteins?
Several studies have implied that more than half of all drug targets are transmembrane proteins [1] [2]. This type of proteins plays crucial roles in myriad physiological processes [3]. A key bottle neck in obtaining these proteins is identifying conditions where the protein is stably over-expressed followed by its isolation into an environment that closely mimics its lipidic habitat. Although various prokaryotic to eukaryotic expression systems are available for rapid overexpression of stable membrane proteins now [4], how to obtain the full-length and natural intact conformation of transmembrane proteins (especially multi-transmembrane proteins) is still a popular topic in protein production. Here, we present three recombinant multi-transmembrane protein technology platforms, including virus like particles (VLPs), detergent micelle and Nanodisc platforms.
2.1 Virus Like Particles (VLPs) Platform
Virus Like Particles (VLPs) are nanometer-scale virus-derived structures made up of one or more capsid proteins with the ability to the automatic assemble [5]. VLPs do not contain viral nucleic acid and are non-infectious. VLPs are dispersed nanomaterials that can be produced in a variety of systems, including mammals, plants, insects, and bacteria. VLPs can be exploited as carriers for the delivery of bio- and nanomaterials, such as drugs, vaccines, quantum dots and imaging substances by virtue of the cavity within their structure [6].
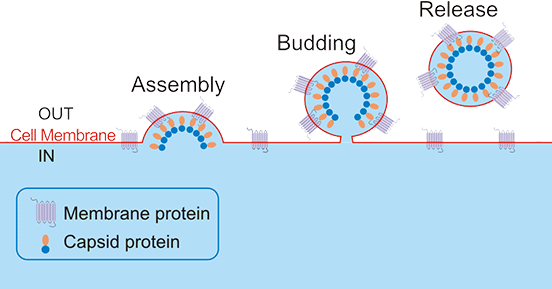
According to the structure of the parental virus, VLPs can be divided into two major categories: non-enveloped VLPs and enveloped VLPs. Non-enveloped viruses are surrounded by a proteinaceous membrane commonly referred to as the capsid. For non-enveloped viruses, this capsid is the tool the virus uses to attach to the host cell. Non-enveloped VLPs are developed based on the features of non-enveloped viruses, usually consisting of one or more components of the pathogen, with the ability to self-assemble into particles, excluding any host components. Not only do enveloped viruses have a protein coat, but the capsid is also surrounded by a lipid membrane. That is, an enveloped virus is a lipid inclusion. These lipids help the virus stick firmly to its target host cells. The viral envelope is antigenic and its main function is to help the virus enter host cells and maintain the structural integrity of the virion. The enveloped VLPs are developed based on the features of enveloped viruses and their structures are relatively complex. After the recombinant expression system self-assembles into particles into the host cell, it is released from the host cell membrane in a budding manner to form particles composed of the host cell membrane and the integrated target antigen displayed on the outer surface.
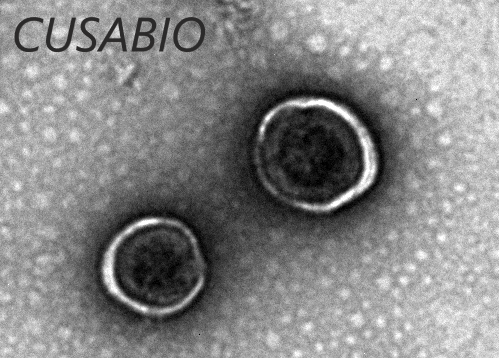
CUSABIO Virus Like Particles (VLPs) Platform is based on the HEK293 expression system by envelope VLPs technology. As the figure 2 shows, after the recombinant expression system self-assembles into particles into the host cell, it is released from the host cell membrane in a budding manner to form particles composed of the host cell membrane and the integrated target antigen displayed on the outer surface.
Figure 2. The Diagram of Virus Like Particles
The prepared enveloped VLPs display correctly folded multiple transmembrane proteins on their native cell membrane, enabling induction and screening of functional antibodies by recognizing the native conformation of the target.. The features of Virus Like Particles (VLPs) Platform are as follows:
- Full-length TPs with complete natural conformation
- Higher immunogenicity
- Higher abundance than that of overexpressing cells
- Can be the best targets for phage display because of their 100-200nm in size
- Can be used in immunization/ELISA/SPR/BLI
Cases Display
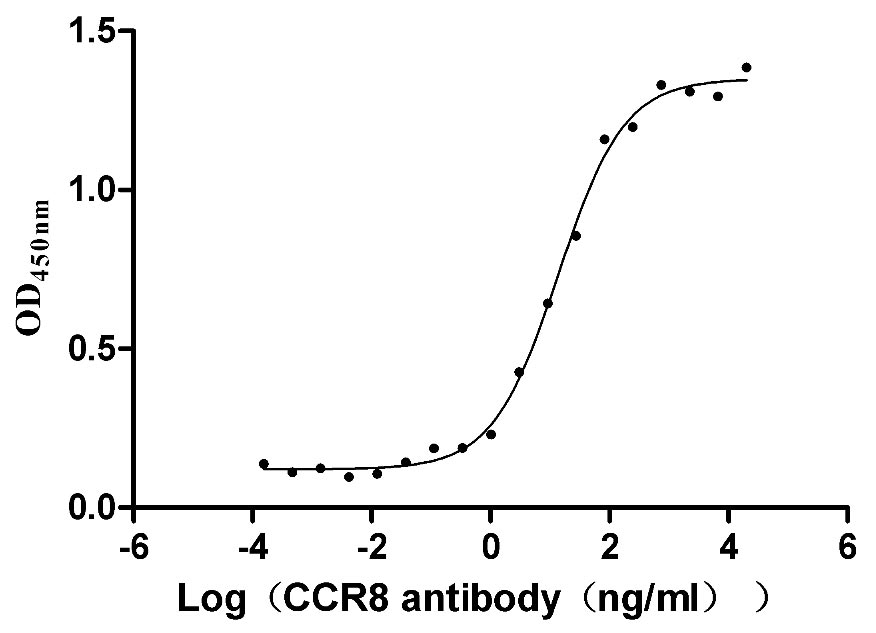
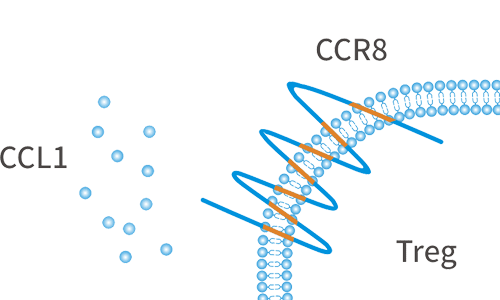
CCR8 is a chemokine receptor that is highly expressed in tumor-infiltrating Tregs, and can be used as a biomarker for Treg cells at tumor sites. CCR8 is specifically expressed on Tregs in tumor sites, but is not expressed on Tregs in peripheral blood or normal tissues. CCR8 is a very potential tumor treatment target.
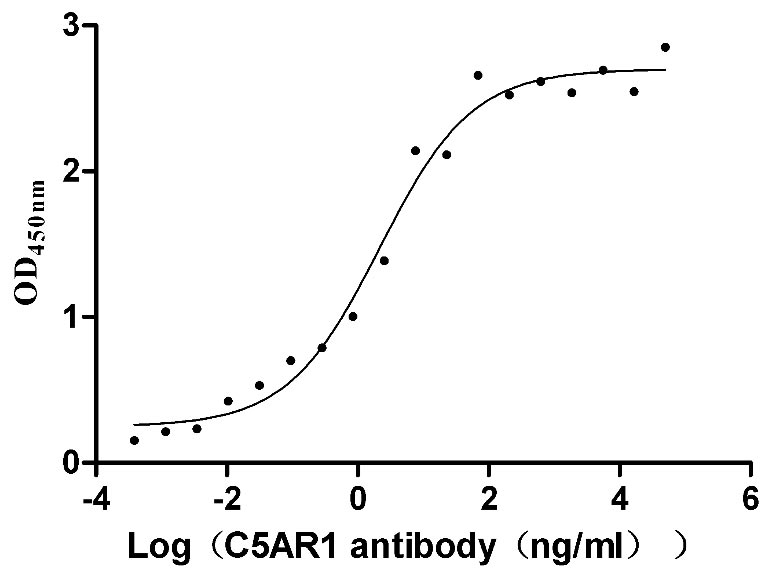
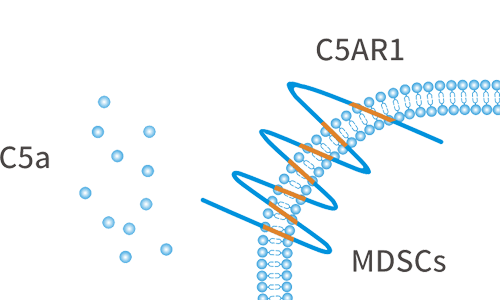
C5AR1, also known as complement fragment C5a receptor, is a classic G protein-coupled receptor, which plays a key role in the innate immune response. C5a is produced in the tumor microenvironment and acts as a chemokine through its C5AR1 receptor to recruit tumor-promoting cells to the tumor site, thereby inhibiting the immune cell attacking the tumor and promoting tumor progression. C5AR1 is a potential new drug target in the field of immuno-oncology.
2.2 Detergent Micelle Platform
Detergents, also known as surfactants, are amphiphilic molecules and able to disintegrate lipid membranes to release membrane proteins, and provide hydrophobic protection for membrane proteins in hydrophilic solutions. Resultant detergent-solubilized membrane proteins can maintain their native transmembrane structure in lipid-free environments. This characteristic is used to solve the problem of natural conformation in TPs expression.
According to the characteristics of the polar groups of detergents, they are able to be divided into three types, including ionic detergents, non-ionic detergents and zwitterionic detergents. Ionic detergents contain charged polar groups and can be further divided into cationic detergents and anionic detergents. Ionic detergents are susceptible to the ionic composition of the solution and may interact with the ion exchange column to interfere with protein purification. Therefore, when purifying membrane proteins using ion exchange columns, non-ionic or zwitterionic detergents are required. Non-ionic detergents contain uncharged hydrophilic polar groups, which are basically unaffected by ionic components in the solution, and can destroy the interaction between lipid molecules and between lipid molecules and membrane proteins, but will not destroy proteins. So it is often used for active separation and purification of membrane proteins. Zwitterionic detergents have characteristics of both ionic and nonionic detergents: on the one hand, the polar groups of zwitterionic detergents are uncharged and do not interact with ion exchange media; on the other hand, zwitterionic detergents can disrupt protein-protein interactions.
CUSABIO has specially built a detergent technology platform based on cell free expression. Synthesize proteins by adding DNA templates, ATP, amino acids, various substrates, and enzymes in cell extracts in vitro, and then extract the target protein by screening different detergents. At present, we have screened Brij78, FOS12 and DDM. A good detergent can dissolve the purified membrane protein well and help it form the correct conformation in solution. The features of detergent micelle platform are as follows:
- Full-length TPs, not only contain complete ECD epitopes
- Can be accurately quantified
- Compatible with immunization/ELISA/SPR/BLI
Cases Display
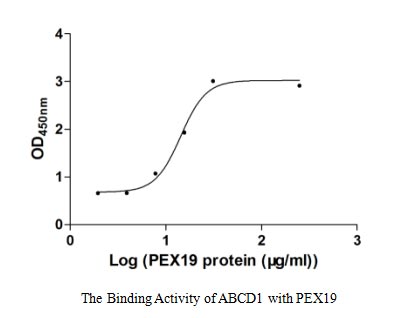
ABCD1 protein is a member of the ALD subfamily, which is involved in peroxisomal import of fatty acids and/or fatty acyl-CoAs in the organelle. Diseases associated with ABCD1 include Adrenoleukodystrophy and Hypoadrenocorticism, Familial.
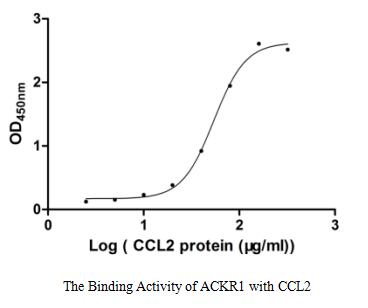
ACKR1 is an atypical and non-specific receptor for several chemokines, such as inflammatory chemokines of both the CXC and the CC subfamilies. The protein encoded by ACKR1 gene, also called interceptor or chemokine-scavenging receptor, controls chemokine levels and localization via high-affinity chemokine binding that is uncoupled from classic ligand-driven signal transduction cascades, resulting instead in chemokine sequestration, degradation, or transcytosis.
2.3 Nanodisc Platform
A few TPs that are sensitive to detergents are not suitable for buffer containing detergents, which are specifically characterized as being unable to be purified, easily degraded and inactive. For this type of membrane protein, we suggest to try the nanodiscs technology.
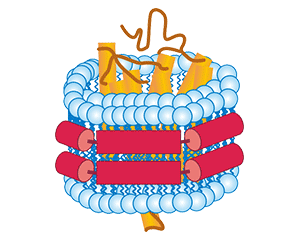
Nanodisc is a membrane-like structure formed by encapsulating a phospholipid bilayer by membrane scaffold protein (MSP). Its structure is similar to that of pie-shaped HDL in vivo. Different studies have proposed 3 structural models of pie-shaped HDL, including the molecular ribbon model, the Helix hairpin model and the guardrail model. Accumulating evidence has implied that the molecular ribbon model is the most probable Nanodisc model. The molecular ribbon model proposes that two MSP proteins surround the phospholipid bilayer head-to-tail, and their Helix conformation is parallel to the phospholipid molecule. The hydrophobic residues of MSP protein are located on the inside of Helix and combine with the hydrophobic tail of phospholipid molecules through hydrophobic interaction, and the hydrophilic residues of the protein are located on the outside of Helix to maintain the soluble state of Nanodisc.
CUSABIO incorporates both in vitro E.coli protein expression and nanodiscs technology, adding pre-assembled nanodiscs to in vitro E.coli protein expression systems. The membrane proteins are assembled into nanodiscs while they are translated, forming membrane protein-nanodiscs complexes. This simulated artificial lipid environment provides new avenues for analyzing the effects of different lipids on membrane proteins, allowing us to better understand membrane proteins. The features of detergent Nanodisc platform are as follows:
- Full-length TPs with complete natural conformation
- Can be accurately quantified
- Detergent-free, suitable for experiments that interfere with detergents
- Can be used directly for phage screening
- Compatible with immunization/ELISA/SPR/BLI/cell experiments
In addition to providing multiple transmembrane proteins routinely produced by these three platforms, CUSABIO also offers accepts customized services with traditional protein expression technologies, including E. coli expression system, yeast expression system, insect baculovirus expression system and mammalian cell expression system.