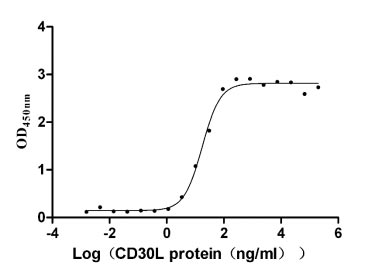
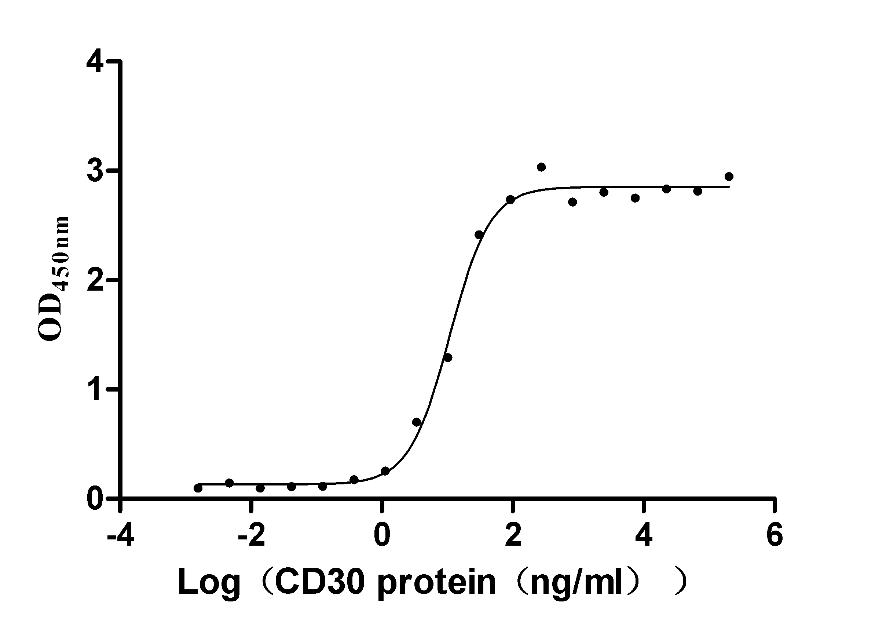
CD30 (TNFRSF8) serves as a critical costimulatory receptor in Hodgkin lymphoma biology and T-cell activation, making precise characterization of its ligand interactions essential for oncology-focused drug discovery. This recombinant human CD30 ectodomain, spanning residues 19–379, is produced in mammalian cells to preserve native glycosylation and disulfide-dependent folding required for authentic CD30L engagement. Functional ELISA confirms robust binding to human CD30L with an EC50 of 14.96–20.25 ng/ml, providing a validated basis for ligand-receptor interaction assays, competitive blocking antibody screening, therapeutic antibody epitope mapping, and affinity characterization by SPR or BLI. The protein carries N-terminal 10×His and C-terminal Myc tags for flexible immobilization strategies, and with purity exceeding 95% by SDS-PAGE alongside endotoxin levels below 1.0 EU/μg, it meets the quality thresholds commonly required for cell-based functional assays and sensitive binding studies in cancer research.