The tumor necrosis factor superfamily (TNFSF) consists of 19 ligands and 29 receptors. TNF superfamily members induce a variety of biological effects, including cell growth, differentiation, and death by the interaction of ligand and receptor. Currently, it is known that tumor necrosis factor (TNF) superfamily members play an important role in the regulation of inflammatory response, immune response to infection and tissue homeostasis. In addition, it may be related to the etiology of some acquired immunodeficiency diseases and genetic diseases.
1. History of The TNFSF Family
The first discovered members of the TNFSF family were two cytotoxic factors isolated by Aggarwal and colleagues in 1984: one from macrophages (molecular weight 17kD), named TNF-α; the other from lymphocytes (molecular weight 20kD), it is named lymphotoxin [1].
Since the amino acid sequence homology of the two cytotoxic factors is 50% [2] and they bind to the same receptor, they are named TNF-alpha and TNF-beta respectively.
All other members of the family, with the exception of TNF-alpha and TNF-beta, have been identified by homology to human cDNA sequences. In this way, 19 members of the TNF superfamily and 29 receptors have been identified. These members play a highly diverse role in the organism. Among these receptors, three TNF receptors (TNFRSF22-mDcTRAILR2/TNFRH2, TNFRSF23-mDcTRAILR1/TNFRH1, and TNFRSF26-mTNFRH3) were found only in mice [3].
2. Structural Characteristics of TNF Ligands
The TNFSF ligand molecule is a trimeric of type II transmembrane protein, and the TNF ligand has a common structural motif, the C-terminal TNF homology domain (THD), through which TNF ligand binds to TNF receptor. THD is a long sequence of 150 amino acids containing a conserved framework of aromatic and hydrophobic residues. THDs have almost identical triple folding and bind to form trimer proteins.
The forms in which the TNF ligand functions include two types: membrane-bound ligands or soluble ligands. Although most ligands are synthesized in the form of membrane-bound proteins, soluble forms can be produced by restriction proteolysis [4].
2.1 Member of Tumor Necrosis Factor Superfamily Ligand
There are 19 members of the tumor necrosis factor super-family ligand, and their specific information is shown in Table 1.
Table 1 Expression profile of ligands of human tumor necrosis factor superfamily [5]
|
Symbol
|
Ligand (alias)
|
Cellular expression
|
|
TNFSF1
|
TNF-α
|
NK, T, and B cells
|
|
TNFSF2
|
TNF-β (LT-α)
|
Macrophages and NK, T, and B cells
|
|
TNFSF3
|
LT-β
|
Activated CD4+ T cells and T, DC, and NK cells
|
|
TNFSF4
|
OX40L (CD252, gp34)
|
B and T cells, DCs, endothelial and smooth muscle cells
|
|
TNFSF5
|
CD40L (CD154, gp39)
|
Activated CD4+ T lymphocytes, NK cells, mast cells, basophils, and eosinophils
|
|
TNFSF6
|
FasL (CD95L, Apo1L)
|
Activated splenocytes, thymocytes, nonlymphoid tissues, and NK cells
|
|
TNFSF7
|
CD27L (CD70)
|
NK, T, B, and mast cells, smooth muscle and thymic epithelial cells
|
|
TNFSF8
|
CD30L (CD153)
|
Activated T cells, B cells, and monocytes, granulocytes, and medullary thymic epithelial cells
|
|
TNFSF9
|
4–1BBL
|
APCs (B cells, macrophages, and DCs), mast cells
|
|
TNFSF10
|
TRAIL (Apo2L)
|
NK and T cells, DCs
|
|
TNFSF11
|
RANKL (TRANCE, OPGL, ODF)
|
T cells, thymus, and lymph nodes
|
|
TNFSF12
|
TWEAK (Apo3L)
|
Monocytes
|
|
TNFSF13
|
APRIL (TALL-2, TRDL-1)
|
Macrophages, lymphoid cells, and tumor cells
|
|
TNFSF13B
|
BAFF (BLYS, THANK)
|
T cells, monocytes, macrophages, and DCs
|
|
TNFSF14
|
LIGHT (HVEML, LT-γ)
|
T cells, granulocytes, monocytes, and DCs
|
|
TNFSF15
|
VEGI (TL1A)
|
Endothelial cells
|
|
TNFSF18
|
GITRL
|
HUVECs
|
|
/
|
EDA-A1
|
Skin
|
|
/
|
EDA-A2
|
Skin
|
3. Structural Characteristics of TNF Family Receptors
The TNF family receptor is a type I or type III membrane protein with a different number of cysteine-rich domains (CRDs). The number of CRDs varies from 1 to 4 for different receptors.
Most receptors of the TNF superfamily can be divided into two categories: one with an intracellular death domain (DD) and the other without. For information on TNF superfamily receptors, see the article: Tumor Necrosis Factor Receptor Superfamily and Apoptosis.
4. Tumor Necrosis Factor Ligand and Receptor Interactions
The 19 ligand members of the TNF superfamily interact with 29 different receptors, indicating that the sites of contact between the receptor and the ligand are diverse. Different ligands can bind to the same receptor (such as TNF-alpha and TNF-beta, which both bind to the same receptor with similar affinity), and the same ligand can bind to different receptors.
For example, TNF-α interacts with two different receptors, tumor necrosis factor receptor 1 (TNFR1) and TNFR2. BAFF on B cells binds to three different receptors, including transmembrane activators and CAML interactors (TACI), B cell mature protein (BCMA) and BAFF receptor (BAFFR). TRAIL binds to five different receptors, including death receptor 4 (DR4), DR5, decoy receptor 1 (DcR1), DcR2, osteoprotegerin (OPG) [6].
Another feature of some ligand receptor interactions in the TNF superfamily is the phenomenon of “reverse signaling”: the transfer of signals from receptors to cells with transmembrane ligands [7]. Reverse signaling has been reported in CD30 ligands [8].
Receptor-ligand interactions usually result in three outcomes:
-
Cell activation mediated by activation of NF-κB and MAP kinase activities by TRAF family proteins.
-
Caspase-dependent apoptosis mediated by FADD and caspase-8.
-
Caspase-independent necrosis mediated by RIP1 and RIP3 kinase.
Through the binding of receptors and ligands, downstream signal transduction is initiated to induce cell growth, differentiation and apoptosis, which plays an important role in the immune regulation of tissue self-stability and inflammation.
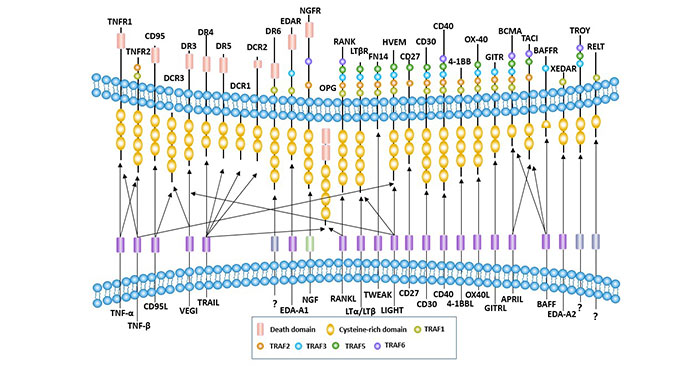
Figure 1 Interaction between TNF superfamily receptor and ligand
5. Cell Signaling
Most cytokine signaling is mediated by interactions between soluble ligands and transmembrane receptors. However, there are many types of ligands in the TNF superfamily. Some ligands are mainly expressed as transmembrane proteins on the cell surface (FasL, CD27L, CD30L, CD40L, OX40L, and 4-1BBL). Some ligands are expressed in both transmembrane and soluble forms (TNF- alpha). Ligands such as TNF- beta lack transmembrane domain and can only be expressed as soluble proteins.
TNF superfamily ligands bind to receptors and activate different types of signals, including NF- kappa B, extracellular signal regulating kinase (ERK), p38 mitogen activated protein kinase (p38 MAPK), and c-Jun N-terminal kinase signaling pathways.
Taking TNF-α as an example, when TNF-α binds to TNFR1, its death domain (DD) recruits a protein called TNFR associated death domain (TRADD) [9]. TRADD subsequently recruited a protein called fas-associated protein with death domain (FADD), which activates caspase-8 and caspase-3, leading to apoptosis [10]. In addition, TNF-alpha can activate mitochondria, releasing ROS, cytochrome C and Bax successively, thereby activating caspase-9 and caspase-3, leading to apoptosis [11]. TNF-α also activates NF-κB, which regulates the expression of proteins involved in cell survival and cell proliferation [12]. TNF-α also activates cell proliferation by activating another transcription factor, activator protein-1 (AP-1) [13].
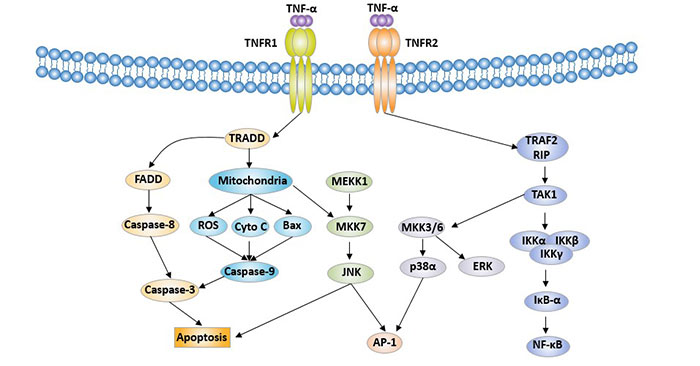
Figure 2 Cell signaling mediated by TNF superfamily
6. Biological Role of TNF Superfamily
TNF superfamily ligands bind to receptors and mediate apoptosis [14] [15] (eg TNF, LT, CD95L, TRAIL, VEGI, TWEAK and LIGHT), survival (eg RANKL and BAFF), differentiation (such as TNE, RANKL and DR6) or proliferation (such as TNF, CD27L, CD30L, CD40L, OX40L, 4-1BBL, APRIL and BAFF) and other physiological processes.
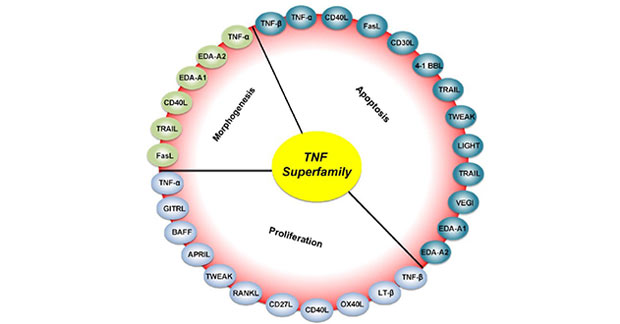
Figure 3 Physiological processes mediated by TNF superfamily [5]
These superfamily members modulate the immune response through a costimulatory, maturation, and cell death signaling pathway. In addition, they play an important role in intraosseous balance, exocrine gland development, and the nervous system.
Most members of the TNF superfamily have both beneficial and potentially harmful effects [16].
6.1 Beneficial Effect
-
Anticancer potential: tumor necrosis factor was initially considered as an effective anticancer drug. But its toxicity limits its ability to inhibit cancer. Inhibition of this systemic toxicity can better exert its anticancer effect. Studies have shown that matrix metalloproteinases can block TNF-induced lethal hepatitis and apoptosis, making anti-tumor therapy safe. TRAIL has been found to specifically kill tumor cells without harming normal cells.
-
Regulate the immune system: The immune system is regulated by cell proliferation and differentiation and apoptosis. Ligands of the TNF superfamily control and coordinate immune responses at multiple levels. TNF, LT, LTβ and RANKL provide a key signal for the morphogenesis of secondary lymphoid organs. The CD95-CD95L interaction is important in several aspects of regulating the immune system. TRAIL also plays an important role in the regulation of the immune system, which regulates lymphocyte function mainly by inhibiting the cell cycle progression of T cells.
-
Prevent microbial infection: TNF signaling is involved in the host's effective response to bacterial infections.
6.2 Harmful Effect
Some of the deleterious activities of TNF and its superfamily members are thought to be mediated through activation of NF-κB.
-
Cancer: Although TNF has been discovered as a cytokine that kills tumor cells, current research indicates that TNF can promote tumorigenesis by mediating tumor cell proliferation, invasion and metastasis. The TNF superfamily induces the expression of various genes involved in invasion and metastasis through activation of NF-κB (including adhesion molecules, urokinase plasminogen activator (UPA), matrix metalloproteinase 9 (MMP9), cyclooxidase 2 (COX2) and vascular endothelial growth factor (VEGF). In addition, activation of NF-κB can inhibit apoptosis and further promote tumorigenesis.
-
Autoimmunity: Several TNF ligands are involved in the development of autoimmunity. Transgenic overexpression of BAFF results in many autoimmune symptoms in mice. The abnormal expression of LIGHT in the body is also related to some autoimmune diseases, indicating that LIGHT plays an important role in the body's autoimmunity, tumor immunity and rejection of organ transplantation.
-
Other diseases: The ligands of the TNF superfamily have been associated with diseases including chronic heart failure, bone resorption, AIDS, Alzheimer's disease, transplant rejection, atherosclerosis and hepatotoxicity.
7. TNF Superfamily and Disease
Members of the TNF family are associated with a range of pathophysiology, including cancer, neurological, cardiovascular, pulmonary, autoimmune, and metabolic diseases.
Studies have shown that TNFSF15 level is decreased in tumor cells, including breast cancer, bladder cancer, ovarian cancer, prostate cancer, liver cancer and colorectal cancer. This suggests a link between TNFSF15 and cancer.
TWEAK and its receptor Fn14 signaling pathway are associated with the development of lung diseases such as acute lung injury, asthma and lung cancer. Fn14 antibody or TWEAK neutralizing antibody can alleviate renal interstitial fibrosis caused by many factors.
TWEAK/FN14 interaction can stimulate the release of IL-8 and GM-CSF from human bronchial epithelial cells, aggravating airway inflammation, while FN14 neutralizing antibody can inhibit the release of inflammatory factors, suggesting that TWEAK/Fn14 may play a role in airway inflammation. TNF-α also plays an important role in these lung diseases.
TNF-α is expressed in nerve cells and has an important influence on the survival of these cells by activating NF-κB signaling. TNF-α is currently known to be associated with depression, bipolar disorder, epilepsy, Alzheimer's disease, Parkinson's disease, and multiple sclerosis. In addition, TNF-α also plays an important role in the occurrence and development of cardiovascular diseases.
The role of TNFSF in autoimmune diseases is the most studied. For example, TNFβ plays a crucial role in rheumatoid arthritis (RA). It can activate the phosphorylation of ERK and p38 in the downstream signaling pathway of MAPK, which will activate the downstream signaling pathway and eventually lead to the occurrence of RA.
We use systemic lupus erythematosus (SLE) as an example to illustrate the relevance of TNFSF in autoimmune diseases.
SLE is an autoimmune disease that is associated with the development of autoreactive T and B cells and autoantibody formation.
Systemic lupus erythematosus is associated with members of the TNF superfamily.
BAFF is a lymphocyte co-stimulatory factor. It has important immunomodulatory effects in B cell growth and development, antibody type switching, and T cell activation. BAFF plays an important role in the pathogenesis of SLE.
Studies have shown a negative correlation between APRIL levels in serum and anti-dsDNA antibody titers and severity of SLE [17].
TRAIL levels are elevated in SLE patients, and TRAIL can accelerate the apoptosis of PMNs in lupus patients and participate in T cell-induced monocyte apoptosis.
CD27/CD70 can promote T cell proliferation and differentiation, induce the formation of CTL, and promote the differentiation of B cells into plasma cells.
4-1BB/4-1BBL can cause T lymphocyte proliferation and cytokine production. 4-1BB and 4-1BBL activate nuclear factor NF-κB, JNK and p38MAPK activation pathways to regulate T cell apoptosis.
OX40/OX40L is a pair of important costimulatory molecules in the immune response process, mainly in the late stage of T cell immune response. It plays an important role in the development of Th1 and Th2 cells.
TNFSF plays different roles in SLE. They act as synergistic molecules to stimulate lymphocyte activation, proliferation, or induce apoptosis, and participate in the occurrence and development of diseases.
8. Therapy Based on The TNF Superfamily
TNF has been considered for many years to be a pro-inflammatory molecule that plays a role in inflammation, apoptosis, proliferation, invasion, angiogenesis, metastasis and morphogenesis. Members of the TNF superfamily play an important role in the pathogenesis of a variety of human diseases, and their role in immune, cardiovascular, neurological, pulmonary, and metabolic diseases is now increasingly apparent. This superfamily is therefore an active target for drug development. TNF blockers have recently been approved to treat rheumatoid arthritis, psoriasis, Crohn's disease and osteoporosis.
Several antagonists targeting TNF family members have been approved by FDA and some are in clinical trials. The drugs currently approved are targeting TNF-alpha. Drugs targeting other members of the TNF superfamily are in various stages of research.
Table 2 Selected FDA-approved drugs targeting the TNF superfamily
|
Cytokine
|
Drug
|
Mechanism
|
Disease
|
|
TNF-α
|
Infliximab
|
mAbs
|
Rheumatoid arthritis(RA), Psoriatic arthritis(PA), psoriasis, Amyotrophic lateral sclerosis (ALS), Ulcerative colitis, Crohn disease
|
|
Etanercept
|
receptor derivative (RD)
|
RA, PA, psoriasis, ALS, juvenile RA
|
|
Adalimumab
|
mAbs
|
RA, PA, psoriasis, ALS, juvenile RA, Crohn disease
|
|
Certolizumab
|
mAbs
|
RA, Crohn disease
|
|
Golimumab
|
mAbs
|
RA, PA, Ankylosing spondylitis (AS)
|
References
[1] Kelker H C, Oppenheim J D, Stone-Wolff D, et al. Characterization of human tumor necrosis factor produced by peripheral blood monocytes and its separation from lymphotoxin [J]. International Journal of Cancer, 2010, 36(1): 69-73.
[2] Aggarwal B B, Kohr W J, Hass P E, et al. Human tumor necrosis factor: Production, purification, and characterization [J]. Journal of Biological Chemistry, 1985, 260(4): 2345-2354.
[3] Schneider P, Olson D, Tardivel A, et al. Identification of a new murine tumor necrosis factor receptor locus that contains two novel murine receptors for tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) [J]. Journal of Biological Chemistry, 2003, 278(7): 5444-54.
[4] Bodmer J L, Schneider P, Tschopp J. The molecular architecture of the TNF superfamily [J]. Trends in Biochemical Sciences, 2002, 27(1): 19-26.
[5] Aggarwal Bharat B, Gupta Subash C, Kim Ji Hye, Historical perspectives on tumor necrosis factor and its superfamily: 25 years later, a golden journey [J]. Blood, 2012, 119: 651-65.
[6] Aggarwal B B, Bhardwaj U, Takada Y. Regulation of TRAIL-Induced Apoptosis by Ectopic Expression of Antiapoptotic Factors [J]. Vitamins & Hormones, 2004, 67: 453-483.
[7] Watts A D. A casein kinase I motif present in the cytoplasmic domain of members of the tumour necrosis factor ligand family is implicated in “reverse signalling” [J]. The EMBO Journal, 1999, 18(8): 2119-2126.
[8] Wiley S R, Goodwin R G, Smith C A. Reverse Signaling via CD30 Ligand [J]. The Journal of Immunology, 1996, 157(8): 3635-3639.
[9] Hsu H, Xiong J, Goeddel D V. The TNF receptor 1-associated protein TRADD signals cell death and NF-κB activation [J]. Cell, 1995, 81(4): 495.
[10] Hsu H, Shu H B, Pan M G, et al. TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal transduction pathways [J]. Cell, 1996, 84(2): 299-308.
[11] Morgan M J, Liu Z G. Reactive oxygen species in TNFα-induced signaling and cell death [J]. Molecules and Cells, 2010, 30(1): 1-12.
[12] Aggarwal B B. Nuclear factor-kappaB: the enemy within [J]. Cancer Cell, 2004, 6(3): 203./p>
[13] Natoli G, Costanzo A, Moretti F, et al. Tumor necrosis factor (TNF) receptor 1 signaling downstream of TNF receptor-associated factor 2. Nuclear factor kappaB (NF-kappaB)-inducing kinase requirement for activation of activating protein 1 and NF-kappaB but not of c-Jun N-terminal kinase/stress-activated protein kinase [J]. Journal of Biological Chemistry, 1997, 272(42): 26079-26082.
[14] Bhardwaj A, Aggarwal B B. Receptor-Mediated Choreography of Life and Death [J]. Journal of Clinical Immunology, 2003, 23(5): 317-332.
[15] Upasna G, Aggarwal B B. Regulation of proliferation, survival and apoptosis by members of the TNF superfamily [J]. Biochemical Pharmacology, 2003, 66(8): 1403-1408.
[16] Aggarwal, Bharat B. Signalling pathways of the TNF superfamily: a double-edged sword [J]. Nature Reviews Immunology, 2003, 3(9): 745-756.
[17] Stohl W. Inverse association between circulating APRIL levels and serological and clinical disease activity in patients with systemic lupus erythematosus [J]. Annals of the Rheumatic Diseases, 2004, 63(9): 1096-1103.
CUSABIO team. Does TNF Superfamily Inhibit Cancer or Promote Its Development?. https://www.cusabio.com/c-20950.html






Comments
Leave a Comment