Presently, ADC competition heats up in global biotech or pharmaceuticals companies. According to the Pharmsnap database, there are nearly 100 partnered or licensed deals based on ADC new drugs, which has an estimated value of about $40 billion. As of now, there are around 17 ADC deals in 2022. Among ADC contenders, SLC39A6 has emerged as an potential candidate for ADC. Albeit SLC39A6 is newly discovered target in cancers, it is widely reported in tumor studies and even employed in ADC.
Earlier, pharmaceutical giant Merck has struck a deal to develop Seagen’ Phase II ADC (ladiratuzumab vedotin, also named MK-6440 or SGN-LIV1A). Merck paid $1.6 billion to co-develop and co-commercialize ladiratuzumab vedotin. SGN-LIV1A is a novel ADC, targeting the zinc transporter SLC39A6 for the treatment of solid tumors. Importantly, accumulating studies are suggesting that SLC39A6 is highly expressed in a variety of tumor cells but limited expression in normal tissues, making it an attractive candidate for ADC therapy! Much more information about SLC39A6 is needed to read this article!
1. What are Zinc Transporter Families?
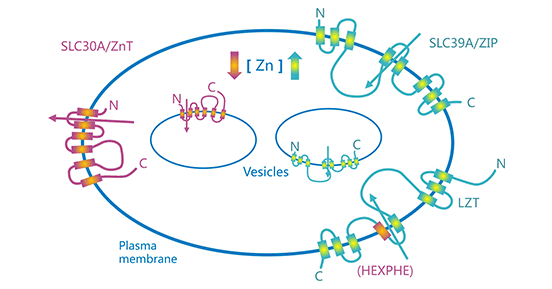
In human, two major zinc transporter families, the SLC39A (ZIP) and SLC30A (ZnT) family control cellular zinc homeostasis, which is zinc influx and efflux between extracellular and intracellular compartments (Figure 1) [1, 6]. The SLC39A/ZIP family has 14 members, namely ZIP1-ZIP14 [2-3]. Furthermore, ZIP channels are classified according to the phylogenetic analysis classifies the fourteen ZIP transporter members into four subfamilies, namely ZIP I (ZIP9), ZIP II (ZIP1, ZIP2, ZIP3), LIV-1 (ZIP4, ZIP5, ZIP6, ZIP7, ZIP8, ZIP10, ZIP12, ZIP13, ZIP14), and gufA (ZIP11). In contrast, the SLC30A/ZnT family, with 10 members, has the opposite role of the SLC39A/ZIP family, with a release zinc function [2-3].
Zinc transporters are distributed differently in various tissues, organelle membranes, cells, and organs, such as SLC39A6, ZIP14, and ZnT1/SLC30A1 are predominantly localized to the cell membrane; in terms of tissue and organ distribution, ZIP5, ZnT1, and ZnT2 are mainly expressed in in pancreatic gland cells, ZIP8 and ZIP10 in blood cells, etc [4-5]. Intriguingly, there is growing evidence that the zinc transporters are not only directly involved in the homeostatic metabolism of zinc in cells, but also influences the metabolic diseases, even in tumors progression [4-5].
Figure 1. Schematic illustration of cellular zinc transport [6]
2. What is SLC39A6?
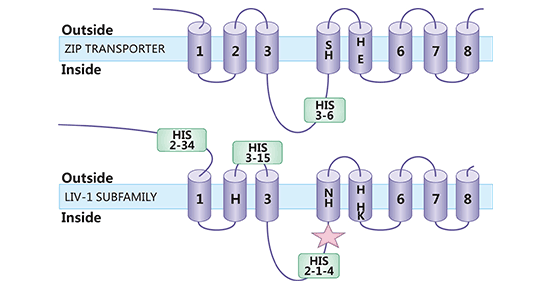
SLC39A6 (also known as ZIP6 or LIV-1) is a member of the SLC39A/ZIP zinc transporter protein family. SLC39A6 exists two isoforms of 755 and 433 amino acids in length. SLC39A6 contains a histidine-rich transcriptional domain that binds to zinc, and aids in its transport into the cell (Figure 2) [7]. The primary function of SLC39A6 is to transfer zinc from the extracellular to the cytoplasm or from the organelle to the cytoplasm. Thus, SLC39A6 plays indispensable roles in maintaining zinc homeostasis by regulating the influx of zinc. SLC39A6 is also regarded as a key protein in the mechanism of action of enzymes, transcription factors, and signaling molecules [8-9].
SLC39A6 is mainly localized in the cell membrane, acting as a zinc-influx transporter. In recent years, many studies on SLC39A6 have indicated that SLC39A6 is abnormally expressed in many cancers, such as esophageal, cervical, lung and breast cancers, etc., suggesting that SLC39A6 might play a critical role in the process of tumor development [10-15].
Figure 2. SLC39A6 structure [7]
3. SLC39A6-Related Regulatory Mechanisms
SLC39A6 have recently received a great deal of attention since its involvement in tumor-related mechanisms, such as JAK/STAT, PI3K/AKT, EGFR-Ras-ERK, MAPK/ERK, and other signaling pathways. SLC39A6 as a newly discovered target in tumors, the complex mechanism of SLC39A6 in tumorigenesis process needs to be further elucidated.
3.1 SLC39A6 and STAT3 Signaling
STAT3 and zinc finger protein (Snail) are major markers of the epithelial-mesenchymal transition (EMT) pathway. SLC39A6 was identified as a downstream target of STAT3 that promotes the entry of Snail into the nucleus. In turn, the Snail reduces the expression of genes related to cell adhesion, which plays an important role in the transformation of epithelial cells into mesenchymal cells [16-17]. Therefore, SLC39A6 can regulate the cell differentiation process by affecting Snail expression.
Another study pointed out that in breast cancer, cells transfected with ZIP6 show inhibition of GSK-3β. The inactivated GSK3β cannot phosphorylate snail, which makes snail stay in the nucleus to inhibit cellular cross-linking gene E-cadherin transcription and promote cell migration [18]. SLC39A6 as a downstream target of STAT3 in esophageal squamous carcinoma, SLC39A6 is involved in the EMT process of esophageal squamous carcinoma by regulating the SNAIL2 gene [19].
3.2 SLC39A6 and EGFR Signaling
EGFR belongs to the membrane receptor tyrosine kinase (RTK) family. EGFR is known receptor for EGF and TGFα. Amphiregulin, betacellulin, heparin-binding EGF (HBEGF), epiregulin, and vaccinia virus growth factor (VGF) also bind to EGFR. The binding between EGFR and its ligands can recruit other receptor substrate proteins and activate a series of downstream signaling pathways, such as Ras/MAPK and PI3K/AKT pathway, which is linked to cancer.
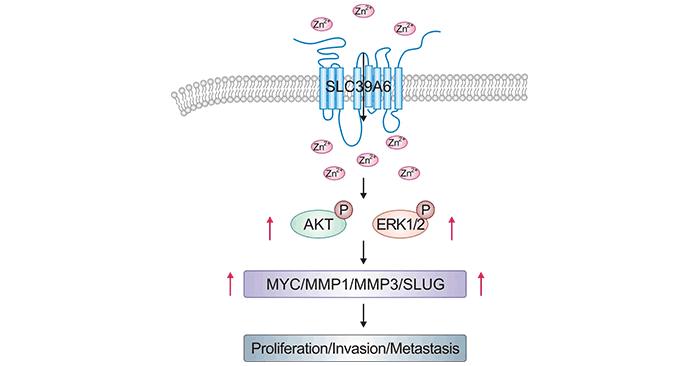
In addition, SLC39A6 in the tumor microenvironment could be activated by EGF, IGF-1, and other growth factors [20]. In prostate tumor cell lines, SLC39A6 overexpression induces EGFR and ERK phosphorylation. As a result, its downstream molecules promote cell migration and invasion [21]. In esophageal squamous cell carcinoma (ESCC), upregulated SLC39A6 activate AKT and ERK pathways that regulate MMP1, MMP3, MYC, and SLUG expression (Figure 3) [22]. Currently, many studies revealed that SLC39A6 can act as an important cancer-related gene, but the specific mechanism of SLC39A6's role in cancer still needs to be further investigated.
Figure 3. SLC39A6 promotes ESCC via AKT and ERK pathways [22]
4. Roles of SLC39A6 in Cancer Therapy
SLC39A6 partly regulates cellular zinc homeostasis. Zinc is a key secondary messenger that can regulate multiple signaling pathways within tumor cells. Currently, SLC39A6 is found to be aberrantly expressed in digestive system tumors (eg. esophageal, liver, and pancreatic cancer), breast cancer, glioma, prostate cancer, lung cancer and bladder cancer. Therefore, SLC39A6 is considered as a promising target for cancer immunotherapy.
4.1 SLC39A6 and Esophageal Cancer
In esophageal cancer, down-regulation of SLC39A6 inhibited the proliferation and invasion of cells. Up-regulation of SLC39A6 was associated with tumor aggressiveness, intracellular zinc levels, and patient survival time. Further analysis showed that with upregulated SLC39A6 expression in esophageal squamous cell carcinoma (ESCC), researchers observed the increased expression levels in matrix metalloproteinase 1, matrix metalloproteinase 3. Successive studies concluded that SLC39A6 has a tumor-promoting role in esophageal cancer, suggesting that SLC39A6 may serve as a promising marker for early detection, prognosis in high-risk patients. Taken together, targeting SLC39A6 may be a potential therapeutic strategy for blocking esophageal cancer [22, 23-24].
4.2 SLC39A6 and Hepatocellular Carcinoma
The miRNA microarray analysis revealed that miR-192 expression was decreased in hepatocellular carcinoma cell lines MHCC-97L, MHCC-97H, and HCC-LM3. Furthermore, miR-192 expression level was decreased in the vascular cell infiltration group compared with the non-infiltrated group. In contrast, SLC39A6 is a direct target of miR-192 and SLC39A6 was negatively correlated with miR-192. Thus, SLC39A6 may promote the migration and invasion of hepatocellular carcinoma cells [25-27].
4.3 SLC39A6 and Pancreatic Cancer
A study implied that SLC39A6 expression level was correlated with tumor size and lymphatic infiltration in 72 pancreatic cancer tissues. By downregulating SLC39A6 expression and constructing a nude mouse model, the results of in vivo and in vitro experiments showed that inhibition of SLC39A6 suppressed pancreatic cancer cell proliferation and migration. SLC39A6 expression in pancreatic cancer is associated with cell proliferation, whereas inhibition of SLC39A6 significantly reduces cell metastasis [28-29].
4.4 SLC39A6 and Breast Cancer
SLC39A6 has been originally described as an estrogen-inducible gene that is upregulated in estrogen receptor-positive breast cancer (ER+BC). Besides, the results of the related RNAseq analysis identified that in the luminal-type breast cancer, SLC39A6, PGR, ESR1, GATA3, BCL2, GATA3, and NAT1 were downregulated in metastatic tumors, suggesting that SLC39A6 may be associated with the grading, size and staging of breast tumors [30-32]. Ladiratuzumab vedotin (SGN-LIVI1), an ADC drug targeting SLC39A6, is currently being used in combination with pembrolizumab (Keytruda) in clinical studies for the treatment of metastatic triple-negative breast cancer [18, 33].
4.5 SLC39A6 and Other Cancers
In addition, aberrant expression of SLC39A6 has been found in a variety of other tumors, including gliomas, ovarian, bladder, prostate, head and neck, lung, and gastric cancers [15, 21, 34-38]. As a key mediator of intracellular zinc levels, SLC39A6 is highly expressed in steroid hormone-sensitive tissues such as the placenta and prostate [1, 21, 39]. In gastric cancer, the single nucleotide polymorphism rs1050631 of SLC39A6 is strongly implicated in the prognosis of patients with surgically removed gastric adenomas [38]. In glioma, SLC39A6 was highly expressed and significantly correlated with tumor grade and stage [34]. In non-small cell lung cancer, knockdown of SLC39A6 restrained cell proliferation, invasion, and migration and cells accumulated in the G1 phase [15, 40].
5. SLC39A6 Clinical Research Prospects
Data from Pharmsnap exhibited that there is one SLC39A6-based ADC antibody drug (Ladiratuzumab vedotin) in clinical phase II for the treatment of adenocarcinoma, esophageal cancer, breast cancer, and many other tumors. There are 11 clinical trials in progress for the treatment of solid tumors such as non-small cell lung cancer and triple-negative breast cancer. As of now, a total of 14 ADC drugs have received the marketing approval. Early efforts at ADCs focused on HER2 and TROP2. Subsequent ADCs have expanded to FRα, VEGF, CD19, CD22, CD30, EGFR, KAAG1, NaPi2b, c-MET, CLDN18.2, HER3, LIV-1/SLC39A6, Nectin-4, and MSLN etc. SLC39A6, as a new tumor-related molecule, is an emerging ADC target that is expected to make breakthroughs in the treatment of esophageal cancer and breast cancer, or other tumors.
To fully support researchers and pharmaceutical companies in their research on SLC39A6 in tumors such as esophageal and breast cancers, CUSABIO presents the SLC39A6 active protein product (Code: CSB-BP621669HU1) to assist you in your research on the mechanism of SLC39A6 or its potential clinical value.
Recombinant Human SLC39A6, partial (Active) (CSB-BP621669HU1)
High Purity Validated by SDS-PAGE
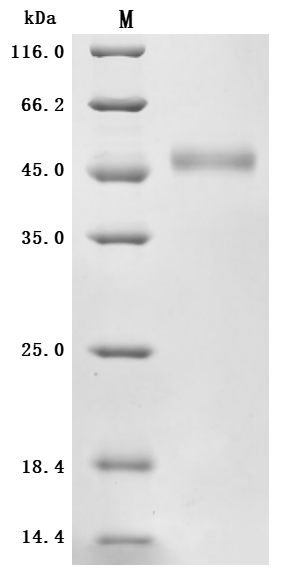
The purity was greater than 95% as determined by SDS-PAGE.
Excellent Bioactivity Validated by Functional ELISA Blot
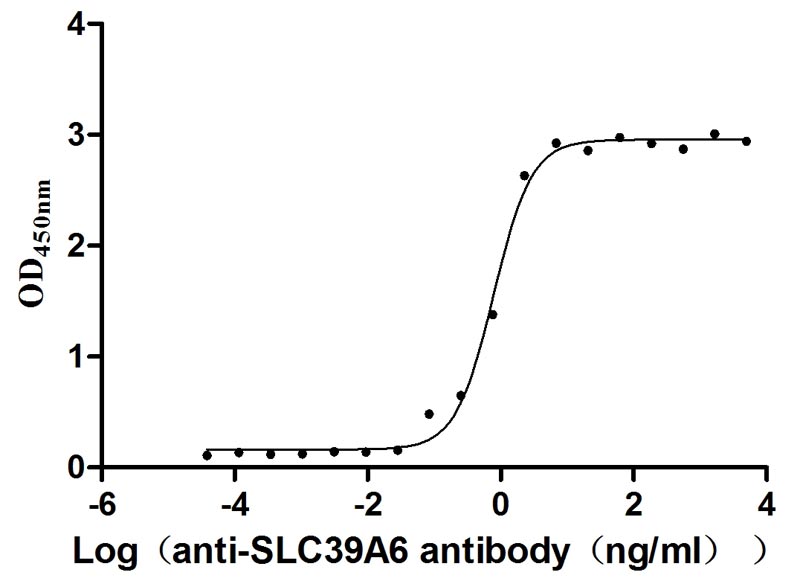
Immobilized Human SLC39A6 at 1 μg/ml can bind Anti-SLC39A6 recombinant antibody (CSB-RA621669MA1HU), the EC50 is 0.6873-0.9010 ng/mL.
References
[1] Prasad, Ram R., et al. "Stage-specific differential expression of zinc transporter SLC30A and SLC39A family proteins during prostate tumorigenesis." Molecular Carcinogenesis 61.5 (2022): 454-471.
[2] Singh, Chandra K., et al. "Role of zinc transporters in prostate cancer and a potential association with racial disparity." Cancer Research 77.13_ Supplement (2017): 4139-4139.
[3] Meng, Jie, et al. "Accumulation of different metals in oyster Crassostrea gigas: significance and specificity of SLC39A (ZIP) and SLC30A (ZnT) gene families and polymorphism variation." Environmental Pollution 276 (2021): 116706.
[4] Nagamatsu, Shino, et al. "Sophisticated expression responses of ZNT1 and MT in response to changes in the expression of ZIPs." Scientific reports 12.1 (2022): 1-13.
[5] Satarug, Soisungwan, et al. "Aberrant expression of ZIP and ZnT zinc transporters in UROtsa cells transformed to malignant cells by cadmium." Stresses 1.2 (2021): 78-89.
[6] Fukada, T., & Kambe, T. (2011). Molecular and genetic features of zinc transporters in physiology and pathogenesis. metallomics: integrated biometal science, 3(7), 662 -674.
[7] Taylor, Kathryn M., and Robert I. Nicholson. "The LZT proteins; the LIV-1 subfamily of zinc transporters. "Biochimica et Biophysica Acta (BBA)- Biomembranes 1611.1-2 (2003): 16-30.
[8] Ghaderi, Hajarossadat, et al. "Preparation of heavy chain polyclonal antibody against zinc transporter SLC39A6 and its diagnostic application." (2021): 274-280.
[9] Ghaderi, Hajarossadat, et al. "Development of camelid monoclonal nanobody against SLC39A6 zinc transporter protein." Iranian Journal of Basic Medical Sciences 24.12 (2021): 1726.
[10] Bagheri, Sajedeh, et al. "Recombinant expression of Zinc transporter SLC39A6 and its functional antibody production." Monoclonal Antibodies in Immunodiagnosis and Immunotherapy 38.2 (2019): 70-74.
[11] Zhao, Lei, et al. "SLC39A6/ZIP6 is essential for zinc homeostasis and T-cell development in zebrafish." biochemical and biophysical research communications 511.4 (2019): 896-902.
[12] Cui, Xiao-Bin, et al. "SLC39A6: a potential target for diagnosis and therapy of esophageal carcinoma." journal of translational medicine 13.1 (2015): 1 -16.
[13] Zhao, Le, Wei Chen, and Xu Li. "Expression of LIV-1 mRNA in human cervical carcinoma and endometrial carcinoma." Nan Fang yi ke da xue xue bao= Journal of Southern Medical University 27.10 (2007): 1590-1592.
[14] Takatani-Nakase, Tomoka, et al. "ZIP6-centered zinc regulatory and malignant characteristics of breast cancer cells." Metallomics Research 2.1 (2022): rev-29.
[15] Wan, Xuechao, et al. "Co-expression analysis revealed PTCH1-3'UTR promoted cell migration and invasion by activating miR-101-3p/SLC39A6 axis in non- small cell lung cancer: implicating the novel function of PTCH1." oncotarget 9.4 (2018): 4798.
[16] Hogstrand, Christer, et al. "A mechanism for epithelial-mesenchymal transition and anoikis resistance in breast cancer triggered by zinc channel ZIP6 and STAT3 (signal transducer and activator of transcription 3)." Biochemical Journal 455.2 (2013): 229-237.
[17] Brethour, Dylan, et al. "A ZIP6-ZIP10 heteromer controls NCAM1 phosphorylation and integration into focal adhesion complexes during epithelial-to-mesenchymal transition." Scientific reports 7.1 (2017): 1-19.
[18] Saravanan, Roshni, et al. "Zinc transporter LIV1: A promising cell surface target for triple negative breast cancer." Journal of Cellular Physiology (2022).
[19] Cheng Xinxin, et al. "The expression of STAT3, SLC39A6 and SNAIL family genes in esophageal squamous cell carcinoma." Proceedings of the National Academic Conference on Tumor Epidemiology and Tumor Etiology. 2015.
[20] Grattan, Bruce J., Kavitha Sankavaram, and Hedley C. Freake. "Regulation of LIV-1 expression in breast cancer cells. "The FASEB Journal 23 (2009): 897-25.
[21] Sussman, Django, et al. "LIV-1 antibody-drug conjugate: A novel therapeutic agent for breast and prostate cancer." Cancer Research 71.8_Supplement (2011): 3620-3620.
[22] Cheng X, Wei L, Huang X, et al. Solute Carrier Family 39 Member 6 Gene Promotes Aggressiveness of Esophageal Carcinoma Cells by Increasing Intracellular Levels of Zinc, Activating Phosphatidylinositol 3-Kinase Signaling, and Up-regulating Genes That Regulate Metastasis. 2021 May;160(6):2228-2229]. Gastroenterology. 2017;152(8):1985-1997.e12.
[23] Cheng, Xinxin, et al. "Solute carrier family 39 member 6 gene promotes aggressiveness of esophageal carcinoma cells by increasing intracellular levels of zinc, activating phosphatidylinositol 3-kinase signaling, and up-regulating genes that regulate metastasis." Gastroenterology 152.8 (2017): 1985-1997.
[24] Wang, An-Hui, et al. "Epidemiological studies of esophageal cancer in the era of genome-wide association studies. "World Journal of Gastrointestinal Pathophysiology 5.3 (2014): 335.
[25] Lian, Junwei, et al. "miR-192, a prognostic indicator, targets the SLC39A6/SNAIL pathway to reduce tumor metastasis in human hepatocellular carcinoma." Oncotarget 7.3 (2016): 2672.
[26] Pascut, Devis, et al. "HCV Proteins Modulate the Host Cell miRNA Expression Contributing to Hepatitis C Pathogenesis and Hepatocellular Carcinoma Development." Cancers 13.10 (2021): 2485.
[27] Wan, Zhen, and Xuzhen Wang. "Role of SLC39A6 in the development and progression of liver cancer." Oncology letters 23.3 (2022): 1-13.
[28] Zhu, Bo, et al. "Increased expression of zinc transporter ZIP4, ZIP11, ZnT1, and ZnT6 predicts poor prognosis in pancreatic cancer." Journal of Trace Elements in Medicine and Biology 65 (2021): 126734.
[29] Liu, Mingyang, et al. "ZIP4 Promotes Pancreatic Cancer Progression by Repressing ZO-1 and Claudin-1 through a ZEB1-Dependent Transcriptional MechanismZIP4 Regulates Pancreatic Cancer Invasion and Metastasis." Clinical Cancer Research 24.13 (2018): 3186-3196.
[30] Garcia-Recio, Susana, et al. "FGFR4 regulates tumor subtype differentiation in luminal breast cancer and metastatic disease." the Journal of clinical investigation 130.9 (2020): 4871-4887.
[31] Taylor, Kathryn M. "A distinct role in breast cancer for two LIV-1 family zinc transporters." (2008): 1247-1251.
[32] Lim, Wai Feng, et al. "Significantly decreased expressions of CaN, VEGF, SLC39A6 and SFRP1 in MDA-MB-231 xenograft breast tumor mice treated with Moringa oleifera leaves and seed residue (MOLSr) extracts." Nutrients 12.10 (2020): 2993.
[33] Pegram, Mark D., et al. "HER2-Overexpressing/Amplified Breast Cancer as a Testing Ground for Antibody-Drug Conjugate Drug Development in Solid TumorsDrug Development of HER2 Antibody-Drug Conjugates." Clinical Cancer Research 26.4 (2020): 775-786.
[34] Do, Minchenko, et al. "Hypoxic regulation of the expression of genes encoded estrogen related proteins in U87 glioma cells: eff ect of IRE1 inhibition." Endocrine Regulations 51.1 (2017): 8-19.
[35] Burgetová, Lenka. "Investigating the role of zinc transporter ZIP 6 and STAT3 in mitosis." (2013).
[36] SU, Juan. "Expression of SLC39A6 in Hnman Bladder Cancer. "Journal of Medical Research (2018): 35-38.
[37] Ressnerova, Alzbeta, et al. "Zinc and copper homeostasis in head and neck cancer: review and meta-analysis." current medicinal chemistry 23.13 (2016): 1304-1330.
[38] Gao, Jian, et al. "Involvement of SLC39A6 in gastric adenocarcinoma and correlation of the SLC39A6 polymorphism rs1050631 with clinical outcomes after resection." bmc cancer 19.1 (2019): 1-15.
[39] Piqué Borràs, Maria Riera. "Identification of molecular subtypes and gene expression patterns of breast cancer analysing RNA-seq data."(2014).
[40] Zhou, Heng, et al. "Evaluation of the prognostic values of solute carrier (SLC) family 39 genes for patients with lung adenocarcinoma." Aging (Albany NY) 13.4 (2021): 5312.
CUSABIO team. SLC39A6/ZIP6: a Key Member of Zinc Transporters, a Novel Targeting Antigen of ADC!. https://www.cusabio.com/c-21096.html





Comments
Leave a Comment