Alternative Names
N; Nucleoprotein; N; Nucleocapsid protein; NC; Protein N
Species Reactivity
Human Novel Coronavirus (SARS-CoV-2/ 2019-nCoV)
Immunogen
Recombinant Human Novel Coronavirus Nucleoprotein (N) (1-419aa) (CSB-EP3325GMY)
Immunogen Species
Human Novel Coronavirus (SARS-CoV-2/ 2019-nCoV)
Isotype
Mouse scFv fusion with human IgG1 Fc
Purification Method
Affinity-chromatography
Concentration
It differs from different batches. Please contact us to confirm it.
Buffer
Preservative: 0.03% Proclin 300
Constituents: 50% Glycerol, 0.01M PBS, pH 7.4
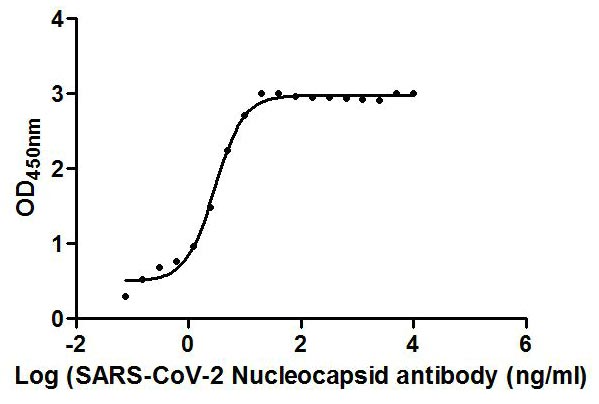
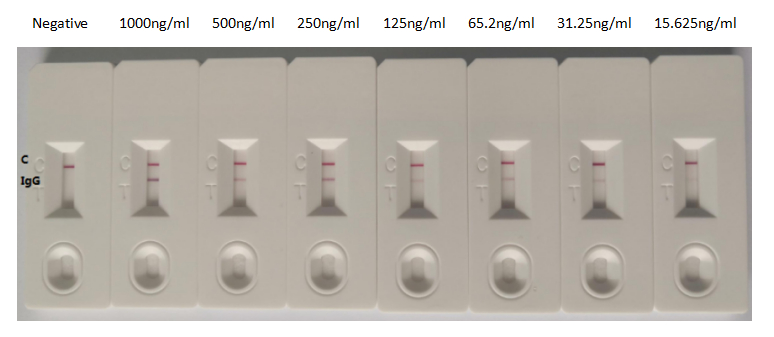
Tested Applications
ELISA, GICA
Recommended Dilution
| Application |
Recommended Dilution |
| ELISA |
1:10000-1:50000 |
| GICA |
1:500-1:5000 |
Storage
Upon receipt, store at -20°C or -80°C. Avoid repeated freeze.
Lead Time
Basically, we can dispatch the products out in 1-3 working days after receiving your orders. Delivery time maybe differs from different purchasing way or location, please kindly consult your local distributors for specific delivery time.
Description
The generation of the N recombinant monoclonal antibody involves a rigorous process to ensure its exceptional quality and specificity. Firstly, B cells are isolated from the spleen of an immunized animal, with the recombinant human SARS-CoV-2 N protein (1-419aa) used as the immunogen. RNA is extracted from the B cells and converted into cDNA through reverse transcription. The N antibody genes are then amplified using specific primers designed for the antibody constant regions and cloned into an expression vector. The human IgG1 Fc is inserted into the vector, downstream of the N antibody. This recombinant vector is transfected into host cells to enable the production of the N recombinant monoclonal antibody. After a period of cell culture, the antibody is collected from the cell culture supernatant and purified using affinity chromatography, resulting in a highly purified form suitable for various applications. To ensure its quality and functionality, the antibody undergoes thorough characterization through assays including ELISA and GICA which validate its specificity and ability to detect human SARS-CoV-2 N protein. The meticulous production process guarantees the development of a reliable and effective N recombinant monoclonal antibody, essential for a wide range of human SARS-CoV-2-related research.
Usage
For Research Use Only. Not for use in diagnostic or therapeutic procedures.