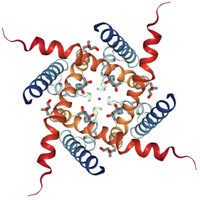
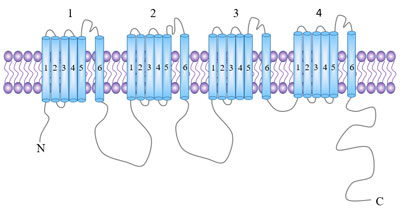
[1] Joel A. Butterwick, Josefina del Mármol, Kelly H. Kim, et al. Cryo-EM structure of the insect olfactory receptor Orco [J]. Nature, 2018, 560: 447–452.
[2] Doyle D, Morais Cabral J, Pfuetzner RA, et al. The structure of the potassium channel: molecular basis of potassium conduction and selectivity [J]. Science, 1998, 280: 69–77.
[3] Dutzler R, Campbell E B, Cadene M, et al. X-ray structure of a ClC chloride channel at 3.0 A reveals the molecular basis of anion selectivity [J]. Nature, 2002, 415(6869): 287-294.
[4] Payandeh J J, Scheuer T T, Zheng N N, et al. The crystal structure of a voltage-gated sodium channel [J]. Nature, 2011, 475(7356): 353-358.
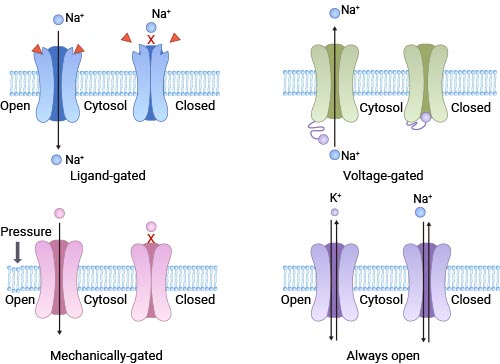
[5] Hille, Bertil. Ionic Channels of Excitable Membranes [J]. Neurology, 1992, 42(7):1439-1439.
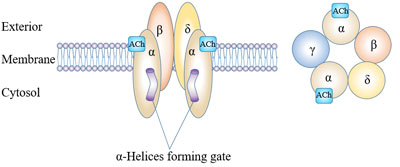
[6] Millar N S, Gotti C. Diversity of vertebrate nicotinic acetylcholine receptors [J]. Neuropharmacology, 2009, 56(1): 237-246.
[7] Changeux, Jean-Pierre. Allosteric Receptors: From Electric Organ to Cognition [J]. Annual Review of Pharmacology and Toxicology, 2010, 50(1): 1-38.
[8] Barnes N M, Hales T G, Lummis S C R, et al. The 5-HT3 receptor--the relationship between structure and function [J]. Neuropharmacology, 2009, 56(1): 273-284.
[9] Walstab J, Rappold G, Niesler B. 5-HT3 receptors: Role in disease and target of drugs [J]. Pharmacology & Therapeutics, 2010, 128(1): 146-169.
[10] Traynelis S F, Wollmuth L P, Mcbain C J, et al. Glutamate Receptor Ion Channels: Structure, Regulation, and Function [J]. Pharmacological Reviews, 2010, 62(3): 405-496.
[11] Surprenant A, North R A. Signaling at Purinergic P2X Receptors [J]. Annual Review of Physiology, 2009, 71(71): 333-359.
[12] Olsen R W, Sieghart W. International Union of Pharmacology. LXX. Subtypes of γ-Aminobutyric AcidA Receptors: Classification on the Basis of Subunit Composition, Pharmacology, and Function. Update [J]. Pharmacological Reviews, 2008, 60(3): 243-260.
[13] Yevenes G E, Zeilhofer H U. Allosteric modulation of glycine receptors [J]. British journal of pharmacology, 2011, 164(2): 224-236.
[14] Louis Ptáček. Ion Channel Diseases: Episodic Disorders of the Nervous System [J]. Seminars in Neurology, 1999, 19(04): 363-369.
[15] Davies N P, Hanna M G. Neurological channelopathies: diagnosis and therapy in the new millennium [J]. Annals of Medicine, 1999, 31(6): 15.
[16] Cotter T G. Apoptosis and cancer: the genesis of a research field [J]. Nature Reviews Cancer, 2009, 9(7): 501-7.
[17] Ramachandran K V, Hennessey J A, Barnett A S, et al. Calcium influx through L-type CaV1.2 Ca2+ channels regulates mandibular development [J]. Journal of Clinical Investigation, 2013, 123(4): 1638-1646.
[18] Daniel Mines, Patricia Tennis, Suellen M Curkendall, et al. Topiramate use in pregnancy and the birth prevalence of oral clefts [J]. Pharmacoepidemiology and Drug Safety, 2014, 23(10): 1017-1025.
[19] Dahal G R, Rawson J, Gassaway B, et al. An inwardly rectifying K+ channel is required for patterning [J]. Development, 2012, 139(19): 3653-3664.
[20] Pai V P, Aw S, Shomrat T, et al. Transmembrane voltage potential controls embryonic eye patterning in Xenopus laevis [J]. Development, 2012, 139(2): 313-23.
[21] Overington J P, Allazikani B, Hopkins A L. How many drug targets are there? [J]. Nature Reviews Drug Discovery, 2006, 5(12): 993-6.