Presently, ample research suggested that CLDN family members are partly responsible for the development and progression of various malignancies, making them promising target candidates for cancer therapy. For example, CLDN1, CLDN3, CLDN4, CLDN6, CLDN9, CLDN17, and CLDN18.2 have been reported to be associated with poor prognosis, which are often abnormally expressed in various cancers including ovarian cancer, prostate cancer, breast cancer, and others.
To date, researchers are mostly focused on Claudin 18.2 and CLDN6 (click for Claudin 18.2 and CLDN6 related articles), but there are over 100 ongoing clinical trials mainly for tumor treatment, which are involved in the different drug therapies such as bispecific antibodies, CAR-T, ADCs, etc. As a result, CLDN family members have become popular targets for immunotherapy. Among these CLDNs, a host of studies revealed that CLDN9 is crucial to cancers, hearing loss, and HCV diseases. Therefore, CLDN9 is becoming one of the most-studied claudins in cancers or other diseases!
1. What is CLDN9?
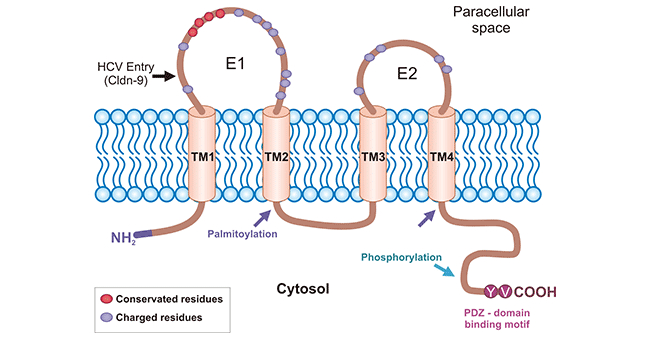
Claudin-9 (CLDN9) is a member of the CLDN family. As an important component of tight junctions (JT), its abnormal expression affects the function of tight junctions. It is widely recognized that CLDNs are similar in structure including four transmembrane structural domains, two extracellular loops, and two intracellular tail regions (Figure 1) [1]. Interestingly, researchers observed the high homology between CLDN6 and CLDN9, which might explain their similar functions in some ways. For example, earlier studies reported that CLDN6 and CLDN9 could mediate hepatitis C virus HCV infection [2]. Subsequent studies found that CLDN6 and CLDN9 are crucial regulators in patients with gastric cancer [3]. In addition, CLDN9 has been discovered to be related with glycolysis levels in endometrial and esophageal adenocarcinomas [4, 5]. More importantly, ample research suggested that aberrant expression of CLDN9 exerts effects on tumors such as lung cancer and liver cancer. Thus, CLDN9 is considered to be the next potent tumor marker from CLDN family [6, 7].
Figure 1. The structure of CLDN9 [1]
2. What's the Mechanism of CLDN9?
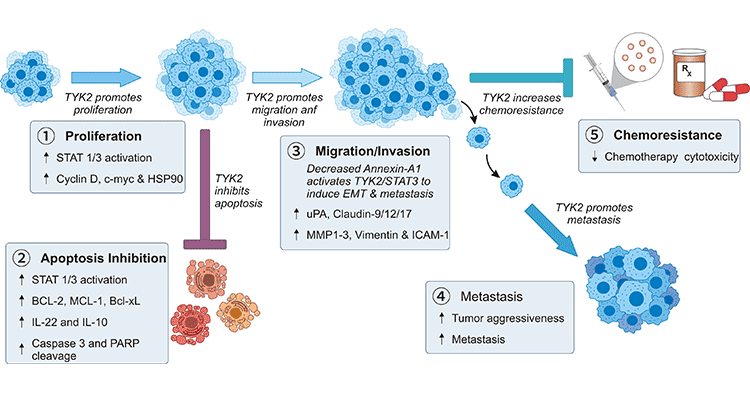
Claudins play a critical role in tight junctions (TJ) by regulating paracellular barrier permeability, but little is known about the signaling pathways. Related studies have shown that CLDNs promote tumor aggressiveness and distant metastasis through the induction of EMT [8]. And the occurrence of EMT is dependent on the aberrant expression and activation of TYK2 [9]. Tyrosine kinase 2 (TYK2), a key member of JAK members (JAK1, JAK2, JAK3 and TYK2), is abnormally expressed in a variety of tumorigenesis and progression. TYK2 is widely considered as an oncogene, contributing to the tumor development (Figure 2) [10].
Previous research found that the expression levels of CLDN9 and TYK2 in pituitary adenomas were correlated with tumor aggressiveness. In pituitary adenomas, high expression of CLDN9 may induce EMT through low expression of TYK2, thereby promoting pituitary adenomas [11]. Another study suggested that upregulation of CLDN9 in hepatocellular carcinoma enhance the cell invasion and metastasis through TYK2/STAT3 signaling pathway [7]. It has also been shown that CLDN9 is associated with glycolysis in esophageal adenocarcinoma. Overexpression of CLDN9 upregulates GLUT-1, PKM2, and LDHA expression levels [12]. In conclusion, CLDN9 is involved in complicated tumor processes, but its precise mechanism needs to be further explored.
Figure 2. The mechanism of CLDN9 and TYK2 in cancer [10]
3. The Roles of CLDN9 in Tumors and Other Diseases
3.1 CLDN9 and Tumors
Accumulated evidence have shown that CLDN9 is positively correlated with tumor aggressiveness. In hepatocellular carcinoma, high expression of CLDN9 activates the TYK2/STAT3 signaling pathway and promotes tumor proliferation [7]; CLDN9 overexpression is also closely associated with metastasis in lung cancer, and CLDN9 knockdown significantly inhibits metastasis in lung cancer [13]. Other data suggested that CLDN9 was upregulated in gastric cancer tissues and correlated with clinicopathological features such as tumor infiltration depth and lymph node metastasis [14]. Investigators further found that highly expressed CLDN9 is also involved in the regulation of tumor glycolysis. Given that, researchers speculated that CLDN9 inhibition may prolong patient survival time and improve patient prognosis. A recent study implied that CLDN9 is associated with pituitary adenomatosis. In the pituitary tumor GT1-1 cell line, CLDN9 expression is higher than in normal pituitary tissue, while TYK2 is degraded, considering that high expression of CLDN9 may promote its aggressiveness through down-regulated TYK2 [11]. These studies concluded that the abnormal expression of CLDN9 may reflect the malignant potential of tumor cells, suggesting that CLDN9 is likely to be a biological marker in tumors.
3.2 CLDN9 and Hepatitis C Virus (HCV)
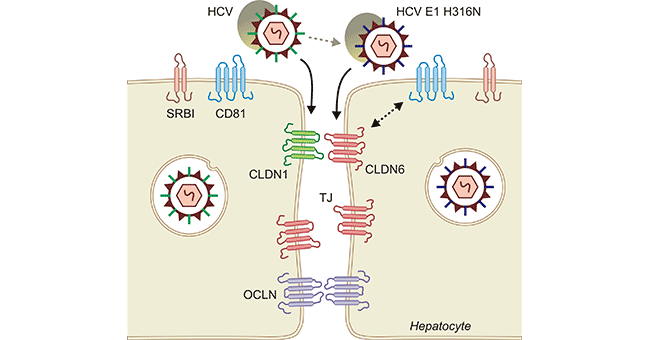
In previous studies, CLDN1 served as family’s only entry factor for hepatitis C virus (HCV) (Figure 3) [15]. However, later studies found that among these CLDNs, CLDN6 and CLDN9 were also able to mediate the entry of HCV into target cells as efficiently as CLDN1 [16, 17]. Hepatitis C virus is the pathogen that causes hepatitis C. Globally, an estimated 58 million people have chronic hepatitis C virus infection, with about 1.5 million new infections occurring per year. HCV is transmitted primarily through parenteral exposures to infectious blood or body fluids. Those patients may exhibit fever, fatigue, decreased appetite, nausea, and vomiting or whatnot. In severe cases, it can cause cirrhosis, endocrine, kidney, and skin diseases. Therefore, the discovery of CLDN6 and CLDN9 as HCV receptors will help to further explore the link between CLDNs and HCV pathogenesis. It also provides new insights for drugs design to inhibit HCV infection in the future.
Figure 3. CLDN1, CLDN6, and CLDN9 might act as HCV receptors [15]
3.3 CLDN9 and Hearing Loss
CLDN9 is an essential component of the tight junctions of epidermal cells, which form a semi-permeable cellular barrier between the ectolymph and endolymph of the inner ear. Abnormal expression of CLDN9 can affect the physiological functions of the body. In some cases, it suggested that CLDN9 plays a crucial role in human hearing. CLDN9 dysregulation can lead to sensorineural hearing loss. CLDN9 expression was observed in the mouse inner ear, which showed mouse CLDN9 expression increases from embryonic to postnatal development and persists in adult inner ears coinciding with prominent CLDN9 immunoreactivity in TJs of epithelia outlining the scala media. However, variants of CLDN9, CLDN11, and CLDN14 can lead to hearing loss and even deafness [18, 19]. Hearing impairment is one of the most common and frequently diagnosed sensory disorders worldwide, with 50% to 70% of cases attributable to genetic causes. Hereditary hearing disorders exhibit great heterogeneity. The identification of deafness-associated genes is one of the most influential advances in hearing biology in recent years, which help researchers understand the pathogenesis involved. In a nutshell, hearing loss induced by CLDN9 mutant genes may serve as deafness-associated genes, which will provide more appropriate and precise treatment options for patients.
4. CLDN9 Clinical Prospects
Curently, many researchers reported the high expression of CLDN1, 3, 4, 6, 9 and 18.2 in different cancers, which represent promising targets for tumor therapy. Albeit no CLDN family targets have been approved yet, the CLANs are contributing the one of the highest-impact research in immunotherapy. As of now, over 70 companies are developing the CLDNs targeting drugs, including Astellas, BioNTech, Amgen, Daiichi Sankyo, I-MAB Biopharma, Novarock Biotherapeutics, Shanghai Genechem Co., Ltd., and others.
Besides Claudin18.2 and CLDN6, researchers are still exploring more CLDN potential targets, expecting to find the best strategies for more disease treatments. Numerous studies have confirmed that CLDN9 participates in complicated diseases including multiple cancers, HCV, and hearing loss. Therefore, CLDN9 target might boast the great potential for the tumor or other diseases diagnosis or therapy.
To assist researchers and pharmaceutical companies in their research on the CLDN9 targeted drug research in tumors, hearing loss, HCV or other diseases. CUSABIO presents CLDN9 active protein (Code: CSB-MP005511HU) to assist you in your research on the mechanism of CLDN9 or its potential clinical value.
● Recombinant Human Claudin-9 (CLDN9)-VLPs (Active)
High Specifity Validated by Western Blot

The high specifity was validated by western blot. CSB-MP005511HU is detected by Mouse anti-6*His monoclonal antibody.
Excellent Bioactivity Validated by Functional ELISA
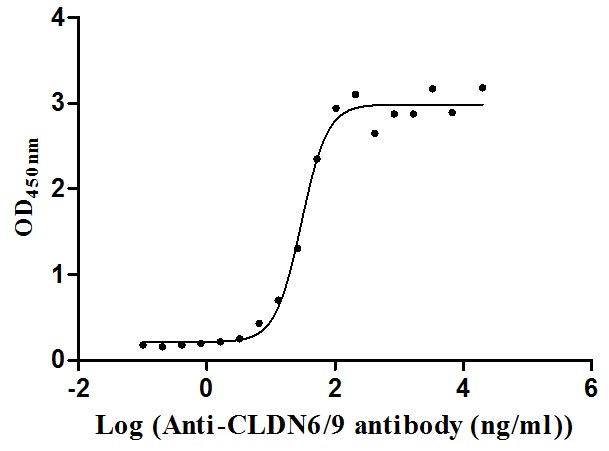
Immobilized Human CLDN9 at 10 μg/ml can bind Anti-CLDN6/9 recombinant antibody (CSB-RA005508MA1HU), the EC50 is 24.93-35.47 ng/mL.
References
[1] Rendón-Huerta, Erika P. et al. "Atlas of Genetics and Cytogenetics in Oncology and Haematology CLDN 9 ( claudin 9 )." (2013).
[2] Zheng, Aihua, et al. "Claudin-6 and claudin-9 function as additional coreceptors for hepatitis C virus." journal of virology 81.22 (2007): 12465-12471 .
[3] Zavala-Zendejas, Veronica Elizabeth, et al. "Claudin-6, 7, or 9 overexpression in the human gastric adenocarcinoma cell line AGS increases its invasiveness, migration, and proliferation rate." Cancer Investigation 29.1 (2011): 1-11.
[4] Kang, Huafeng, et al. "A glycolysis-related gene signature predicts prognosis of patients with esophageal adenocarcinoma." Aging (Albany NY) 12.24 ( 2020): 25828.
[5] Zhu, Jianyou, et al. "Expression of claudin-5,-7,-8 and -9 in cervical carcinoma tissues and adjacent non-neoplastic tissues." International Journal of Clinical and Experimental Pathology 8.8 (2015): 9479.
[6] Sharma, Rajesh, et al. "Enhanced Claudin-9 expression is associated with lung cancer metastasis (173.8)." (2012): 173-8.
[7] Liu, Hongyu, et al. "Claudin-9 enhances the metastatic potential of hepatocytes via Tyk2/Stat3 signaling." The Turkish Journal of Gastroenterology 30.8 (2019): 722.
[8] Morel, Anne-Pierre, et al. "EMT inducers catalyze malignant transformation of mammary epithelial cells and drive tumorigenesis towards claudin-low tumors in transgenic mice." PLoS genetics 8.5 (2012): e1002723.
[9] Majeski, Hannah Elisabeth. The role of Tyk2 in matrix stiffness-driven EMT and metastasis. diss. uc San Diego, 2020.
[10] Borcherding, Dana C., et al. "TYK2 in cancer metastases: genomic and proteomic discovery." Cancers 13.16 (2021): 4171.
[11] Abudukadier YASEN, BA Tu, et al. Expressions of CLDN9 and TYK2 in non-functioning pituitary adenoma correlated with invasiveness[J]. Chinese Journal of Practical Nervous Diseases, 25(5): 552-558.
[12] Kang, Huafeng, et al. "A glycolysis-related gene signature predicts prognosis of patients with esophageal adenocarcinoma." Aging (Albany NY) 12.24 ( 2020): 25828.
[13] Sharma, Rajesh K., et al. "A spontaneous metastasis model reveals the significance of claudin-9 overexpression in lung cancer metastasis." clinical & amp; experimental metastasis 33.3 (2016): 263-275.
[14] Sharma, Rajesh K., et al. "A spontaneous metastasis model reveals the significance of claudin-9 overexpression in lung cancer metastasis." clinical & amp; experimental metastasis 33.3 (2016): 263-275.
[15] Colpitts, Che C., Mirjam B. Zeisel, and Thomas F. Baumert. "When one receptor closes, another opens: claudins and the hepatitis C virus E1 glycoprotein." Hepatology 62.4 (2015): 991-993.
[16] Meertens, Laurent, et al. "The tight junction proteins claudin-1,-6, and-9 are entry cofactors for hepatitis C virus." journal of virology 82.7 (2008): 3555-3560.
[17] Fofana, Isabel, et al. "Functional analysis of claudin-6 and claudin-9 as entry factors for hepatitis C virus infection of human hepatocytes by using monoclonal antibodies." journal of virology 87.18 (2013): 10405-10410.
[18] Sineni, Claire J., et al. "A truncating CLDN9 variant is associated with autosomal recessive nonsyndromic hearing loss." Human genetics 138.10 (2019): 1071-1075.
[19] Ramzan, Memoona, et al. "Variants of human CLDN9 cause mild to profound hearing loss." Human Mutation 42.10 (2021): 1321-1335.
CUSABIO team. CLDN9: Another Key Regulator for Tumors or Hearing Loss from the CLDN Family!. https://www.cusabio.com/c-21074.html





Comments
Leave a Comment