Early, the journal Nature Reviews Clinical Oncology published a review article titled "Overcoming on-target, off-tumour toxicity of CAR T cell therapy for solid tumours", which summarizes the latest advancements in mitigating OTOT (On-Target, Off-Tumour) toxicity in CAR T cell therapy [1]. This study highlights the promising approach of SynNotch CAR T systems to tackle the OTOT issue. Moreover, a proof-of-principle preclinical study effectively validated the efficacy of this regimen in an xenograft model of ovarian cancer expressing ALPPL2-HER2.
Notably, ALPPL2, known as "germ cell alkaline phosphatase" (ALPG), emerges as a highly specific and targetable antigen for tumor cells. These findings underscore the unique potential of ALPG (ALPPL2) and suggest its significance as a promising target for intelligent cell therapies in the treatment of solid tumors.
1. What is Alkaline Phosphatase Isoenzyme (ALP)?
Alkaline phosphatase (ALP) is an enzyme that functions in alkaline conditions, removing phosphate groups from substrates. In most mammals, there are four different isozymes: placental (PLAP/ALPP), intestinal (IAP/ALPi), germ cell (ALPG/ALPPL2/GCAP) and tissue non-specific (liver/bone/kidney) (ALPL/TNAP). ALP is found throughout the body and is involved in various processes, including the regulation of protein phosphorylation, cell growth, and signaling pathways [2-4]. Changes in ALP activity are associated with diseases like osteoporosis, biliary obstruction, and various cancers such as bone, pancreatic, ovarian, breast, and prostate cancer. Additionally, ALP's role in converting adenosine monophosphate to adenosine highlights the vital value for accurate ALP detection in clinical diagnosis [2-4].
2. What is Alkaline Phosphatase, Germ Cell Type (ALPG)?
2.1 ALPG Structure
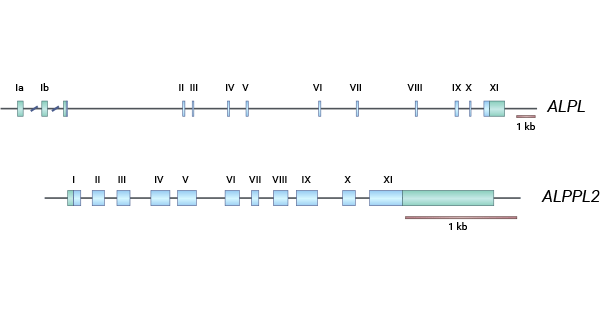
Alkaline Phosphatase, Germ Cell Type (ALPG), known as NAGAO isozyme, GCALP, or ALPPL2, produces ALP-1 protein as part of the alkaline phosphatase isozyme family (ALP). These tissue-specific alkaline phosphatase (TSAP) genes are all located on human chromosome 2q34-q37. Structurally, these TSAP genes are nearly identical, each composed of eleven exons and ten small introns. ALPG, PLAP, and IAP are highly similar, sharing 90-98% genetic homology, while TNAP has about 50% homology with the others. ALPG, like other alkaline phosphatases, is attached to the cell membrane via glycosylphosphatidylinositol (GPI) (Figure 1) [5].
2.2 ALPG Expression and Function
ALPG (ALPPL2/GCAP) is found at low levels in embryonic germ cells, some tumor tissues, and the placenta. It acts as spermatogonial alkaline phosphatase (ALP), a marker in certain cancers like spermatogonial tumors and ovarian cancer. In contrast, GCAP is highly expressed temporarily on pre-implantation primitive state embryonic stem cells' surfaces. Crucially, the accumulating body of evidence reinforces the notion that ALPPL2 serves as a tumor-specific antigen. These findings emphasize the importance of ongoing research to fully understanding ALPPL2's potential in fighting cancer or other diseases [6-8].
Figure 1. ALPG structure [5]
3. ALPG (ALPPL2)-Related Signaling Pathways
3.1 The Mechanisms of ALPG in Maintenance and Establishment of Naïve Pluripotency
Currently, a study reveals the crucial role of ALPG (ALPPL2) in the establishment and maintenance of Naïve pluripotency. Research into Naïve State has become a central focus in the fields of stem cells and reprogramming. The identified surface antigens, as of now, appear to have limited influence on the establishment and maintenance of the Naïve State, and their dynamic changes and related molecular mechanisms remain unclear [6].
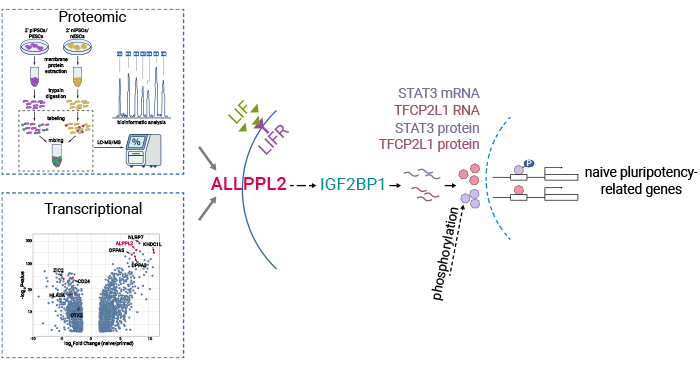
Through a comprehensive examination, the study confirms that ALPPL2 is a critical surface marker for human Naïve state pluripotent stem cells, playing a pivotal role in their initiation and maintenance. Furthermore, ALPPL2 interacts with IGF2BP1, effectively stabilizing mRNA levels of vital pluripotency factors such as TFCP2L1 and STAT3. These findings not only offer valuable insights but also hold the potential to enhance the isolation and cultivation of Naïve state pluripotent stem cells. These highly adaptable cells offer promise for treating various diseases, ranging from heart conditions and diabetes to neurodegenerative disorders, by guiding their differentiation into specific cell types (Figure 2) [6].
Figure 2. The mechanisms of ALPG in maintenance and establishment of Naïve pluripotency [6]
3.2 The Mechanisms of ALPG SynNotch CAR Circuit T Cells in Tumors
SynNotch CAR T is a cutting-edge "smart" cancer therapy designed to target antigens mainly found in healthy tissues [8]. Research suggests that ALPPL2 can act as a guide switch to direct SynNotch CAR T cells towards other tumor-associated antigens, reducing the risk of harming non-cancerous tissues. In preclinical models of solid tumors like mesothelioma and ovarian cancer that express ALPPL2, this approach demonstrated improved precision in tumor control by blocking CAR-mediated signals [9-10].
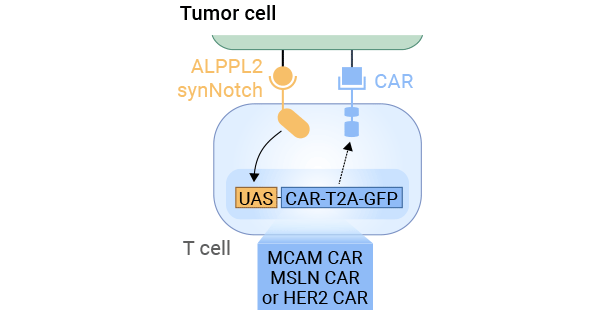
In a specific study, ALPPL2, a tumor-specific antigen, was used to modify T cells, enabling them to produce CAR receptors capable of recognizing and attacking tumor cells when they detect ALPPL2. Three CARs targeting MCAM, mesothelin, and HER2 were employed in the study, and the presence of CAR receptors was confirmed by tracking GFP expression. Two types of mesothelioma tumor cells, M28 (epithelioid) and VAMT-1 (sarcomatoid), were involved in the experiments. These engineered T cells effectively eliminated these tumor cells, and the ALPPL2 SynNotch circuit regulated this process. This circuit can modulate GFP expression, controlling CAR expression and T cell activity. Consequently, the precise control of CAR receptor expression and T cell activity through the ALPPL2 SynNotch circuit has proven effective in killing tumor cells (Figure 3) [9].
Figure 3. The mechanisms of ALPG SynNotch CAR Circuit T Cells in tumors [9]
4. The Roles of ALPG (ALPPL2) in Tumors
Albeit alkaline phosphatase (ALP) is known to be important in various conditions such as bone disorders, gastrointestinal diseases, and cancers, the exact role of ALPG (ALPPL2) in disease is not fully understood. Therefore, ALPPL2 might be involved in the development and progression of these diseases, but more research is needed to determine its specific mechanisms. Presently, most existing studies in the literature focus on ALPG (ALPPL2)'s role in certain types of cancer.
4.1 ALPG and Pancreatic Cancer
In a study, a technique called SELEX was employed to identify an RNA molecule known as SQ-2. SELEX screens a library of random single-stranded nucleic acid sequences to find those with a strong affinity for a specific target. In this case, the study pinpointed the target protein of SQ-2, which turned out to be ALPPL2. ALPPL2 is known to be abnormally expressed in pancreatic cancer cells. To understand the impact of ALPPL2, the study used RNA interference to reduce its expression, leading to significant effects on cell growth and invasion [11-13].
What's more, SQ-2 not only recognizes ALPPL2 on the surface of pancreatic cancer cells but also in cell secretions. This discovery is particularly significant because the lack of early sensitive diagnostic markers has contributed to the high mortality rate associated with pancreatic ductal adenocarcinoma (PDAC). These findings suggest that ALPPL2 could have a vital role in the diagnosis of PDAC, both in serum-based and membrane-based approaches [11-13].
4.2 ALPG and Breast Cancer
In a study involving ER+ breast cancer patients, researchers identified genetic variants linked to the levels of Anastrozole in plasma. The most crucial genetic variant (rs11648166) was near the SLC38A7 gene, suggesting it encodes the Anastrozole transporter protein. Another significant variant (rs28845026) was close to the ALPPL2 gene and interacted with the SLC38A7 variant, affecting both genes' expression [7]. Patients carrying both variants had the highest drug concentrations, elevated SLC38A7 expression, and reduced ALPPL2 expression. This study introduces SLC38A7 as a novel gene responsible for encoding the Anastrozole transporter protein. It also highlights interactions between SNPs in the SLC38A7 gene and those near the ALPPL2 gene, impacting both transporter protein expression and Anastrozole levels in plasma [7].
4.3 ALPG and Mesothelioma
ALPPL2 has been found to be widely expressed in mesothelioma, covering a wide range of subtypes and showing high tissue specificity. Mesothelioma is an incurable tumor that can be classified as epithelial, biphasic and sarcomatoid. Researchers developed the M25 antibody to selectively target ALPPL2 on live mesothelioma and normal cells. Innovative ALPPL2-targeted antibody-drug conjugates were designed, demonstrating potent and precise tumor-killing abilities in mesothelioma cells both in vitro and in vivo. These findings present promising strategies for mesothelioma treatment [14].
4.4 ALPG and Gastric Cancer
In this study, ALPPL2 expression in gastric tissues and adjacent normal tissues was analyzed using immunohistochemical techniques and bioinformatics. The objective was to evaluate its potential for early detection and prognosis prediction in gastric cancer. The findings revealed a significant increase in ALPPL2 expression in gastric adenocarcinoma tissues. Notably, ALPPL2 emerged as an independent factor capable of providing reliable prognostic insights for patients with gastric adenocarcinoma [15]. Furthermore, increased ALPPL2 expression demonstrated a strong association with the progression of TNM staging and elevated HER-2 expression levels. As a result, ALPPL2 holds considerable potential as a promising biomarker for revealing crucial prognostic insights in the treatment of gastric cancer [15].
4.5 ALPG and Other Tumors
ALPG, often referred to ALPPL2, is a central focus in cancer research, especially regarding early detection in seminomas and blood-based diagnostics [6]. It's highly expressed in various solid tumors like ovarian, endometrial, and testicular cancers [6], as well as in different cancers such as lung, bladder, ovarian, melanoma, uterine, renal, prostate, central nervous system tumors, colorectal, lymphoma, and leukemia [6, 16-19]. However, its role in these tumors can vary due to factors like tumor subtype, patient characteristics, and experimental conditions. Thus, further studies and validation specific to each tumor type are needed to understand ALPPL2's function fully.
5. The Clinical Potential of ALPG Targeted Therapy
A clinical program, SGN-ALPV, is being developed to target ALPG (ALPPL2). SGN-ALPV is an advanced treatment that uses specific antibodies to identify ALPG and ALPP, releasing a potent agent called MMAE. This process leads to cancer cell death by blocking cell division, triggering apoptosis, and boosting the immune response [20]. Preclinical studies have shown SGN-ALPV's effectiveness in ovarian, lung, pancreatic, and gastric cancers, regardless of ALPG (ALPPL2) and ALPP levels [20]. At present, although researchers don't fully understand its exact functions and regulatory mechanisms, ALPG (ALPPL2) shows promise in playing a crucial role in different cancer types. Ongoing research will provide deeper insights into its functions and potential applications in cancer therapy.
In Summary:
ALPG (ALPPL2) is a versatile target protein with applications in various aspects of cancer research. It has been identified as a crucial biomarker in multiple tumors, including seminomas and solid tumors such as ovarian, endometrial, and testicular cancers. ALPG's potential extends to early cancer detection and blood-based diagnostics. Moreover, ALPG shows promise in targeted therapies, like antibody-drug conjugates, offering novel treatment options. All in all, its significance lies in its potential as a valuable tool in cancer diagnosis, prognosis, and therapy.
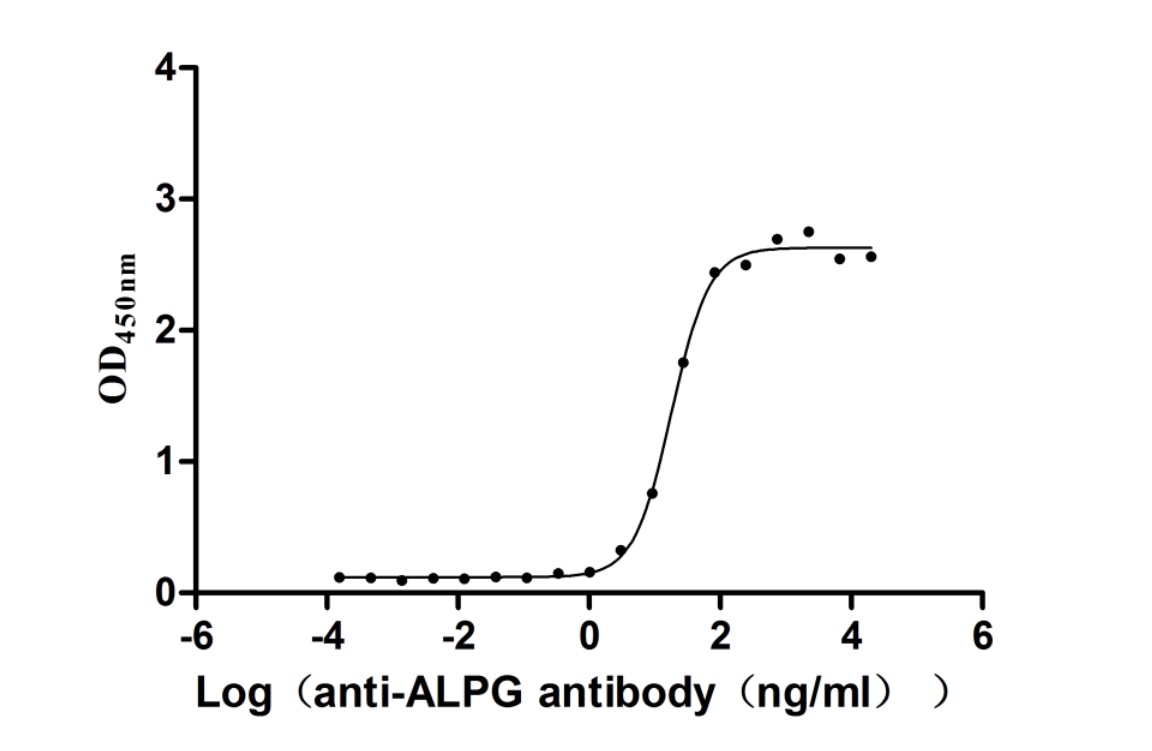
6. CUSABIO ALPG Recombinant Proteins & Antibodies for Research Use
To fully support researchers and pharmaceutical companies in their research on ALPG (ALPPL2) in tumors and other diseases, CUSABIO presents ALPG active proteins & antibodies to support your research on the mechanism of ALPG or its potential clinical value.
References
[1] Flugel, Christian L., et al. "Overcoming on-target, off-tumour toxicity of CAR T cell therapy for solid tumours." Nature Reviews Clinical Oncology 20.1 (2023): 49-62.
[2] Sharma, Ujjawal, Deeksha Pal, and Rajendra Prasad. "Alkaline phosphatase: an overview." Indian journal of clinical biochemistry 29 (2014): 269-278.
[3] Orimo, Hideo. "The mechanism of mineralization and the role of alkaline phosphatase in health and disease." Journal of Nippon Medical School 77.1 (2010): 4-12.
[4] Zhang, Hongyi, et al. "Recent progress of fluorescent probes for the detection of alkaline phosphatase (ALP): A review." Dyes and Pigments 194 (2021): 109569.
[5] Buchet, René, José Luis Millán, and David Magne. "Multisystemic functions of alkaline phosphatases." Phosphatase modulators (2013): 27-51.
[6] Bi, Yan, et al. "Identification of ALPPL2 as a naive pluripotent state-specific surface protein essential for human naive pluripotency regulation." Cell Reports 30.11 (2020): 3917-3931.
[7] Dudenkov, Tanda M., et al. "Anastrozole Aromatase Inhibitor Plasma Drug Concentration Genome‐Wide Association Study: Functional Epistatic Interaction Between SLC 38A7 and ALPPL 2." Clinical Pharmacology & Therapeutics 106.1 (2019): 219-227.
[8] Su, Yang, et al. "ALPPL2 is a highly specific and targetable tumor cell surface antigen." Cancer research 80.20 (2020): 4552-4564.
[9] Hyrenius-Wittsten, Axel, et al. "SynNotch CAR circuits enhance solid tumor recognition and promote persistent antitumor activity in mouse models." Science translational medicine 13.591 (2021): eabd8836.
[10] Hyrenius-Wittsten, Axel, et al. "Enhanced solid tumor recognition and T cell stemness with SynNotch CAR circuits." bioRxiv (2021): 2021-01.
[11] Shin, Hye-Su, et al. "Alppl2 is a potential diagnostic biomarker for pancreatic cancer-derived extracellular vesicles." Molecular Therapy-Methods & Clinical Development 15 (2019): 204-210.
[12] Dua, Pooja, et al. "Alkaline phosphatase ALPPL-2 is a novel pancreatic carcinoma-associated protein." Cancer research 73.6 (2013): 1934-1945.
[13] Dua, Pooja, Soyoun Kim, and Dong-ki Lee. "Alppl2 aptamer-mediated targeted delivery of 5-fluoro-2′-deoxyuridine to pancreatic cancer." nucleic acid therapeutics 25.4 (2015): 180-187.
[14] Li, Yuyang, Min Liu, and Songbo Xie. "Harnessing phage display for the discovery of peptide-based drugs and monoclonal antibodies." Current Medicinal Chemistry 28.40 (2021): 8267-8274.
[15] Liu, Shuang, et al. "High expression of ALPPL2 is associated with poor prognosis in gastric cancer." Human Pathology 86 (2019): 49-56.
[16] Lei, Sibei, et al. "ALPPL2‐binding peptide facilitates targeted mRNA delivery for efficient hepatocellular carcinoma gene therapy." Advanced Functional Materials 32.43 (2022): 2204342.
[17] Zhou, Shuchang, et al. "Present Status and Advances in Chimeric Antigen Receptor T Cell Therapy for Glioblastoma." Frontiers in Bioscience-Landmark 28.9 (2023): 206.
[18] Chen, Laurence C., Andrew J. Hou, and Yvonne Y. Chen. "Getting better mileage with logically primed CARs." Med 2.7 (2021): 785-787.
[19] Halder, Kuntal, et al. "Abstract A041: Extracellular vesicle based ALPPL2 and THBS2 as biomarkers for monitoring disease burden in patients with pancreatic cancer." Cancer Research 82.22_Supplement (2022): A041-A041.
[20] Lakhani, Nehal J., et al. "Phase 1 study of SGN-ALPV, a novel, investigational vedotin antibody–drug conjugate directed to ALPP/ALPPL2 in advanced solid tumors (SGNALPV-001, trial in progress)." (2022): TPS3159-TPS3159.
CUSABIO team. ALPG: a Germ Cell-Associated Alkaline Phosphatase (ALP), a Highly Specific Cell Surface Antigen in Cancers!. https://www.cusabio.com/c-21148.html



Comments
Leave a Comment