[1] Tomomitsu Tahara, Sayumi Tahara, Noriyuki Horiguchi, Takema Kato, Yasuko Shinkai, Masaaki Okubo, Tsuyoshi Terada, Dai Yoshida, Kohei Funasaka, Mitsuo Nagasaka, Yoshihito Nakagawa, Hiroki Kurahashi, Tomoyuki Shibata, Tetsuya Tsukamoto, Naoki Ohmiya.(2019). Prostate Stem Cell Antigen Gene Polymorphism Is Associated withH. pylori–related Promoter DNA Methylation in Nonneoplastic Gastric Epithelium.
[2] Norihisa Saeki, Hiromi Sakamoto, Teruhiko Yoshida.(2014). Mucin 1 Gene (MUC1) and Gastric-Cancer Susceptibility.
[3] Peixi Liu, Mingxi Zeng.(2020). Role ofMUC1rs4072037 polymorphism in gastric cancer: a meta-analysis.
[4] Xi Gu, Jiaxi Feng, Liyang Liu, Ming Lü, Xiang Ma, Yang Cao, Baolin Wang, Qinghong Zhao.(2018). Association of MUC1 rs4072037 Functional Polymorphism and Cancer Risk: Evidence from 12551 Cases and 13436 Controls.
[5] Fujiao Duan, Chunhua Song, Liping Dai, Shuli Cui, Xiaoqin Zhang, Xia Zhao.(2014). The Effect of MUC1 rs4072037 Functional Polymorphism on Cancer Susceptibility: Evidence from Published Studies.
[6] Fujiao Duan, Chunhua Song, Jiachen Shi, Yanchun Wang, Hua Ye, Liping Dai, Jianying Zhang, Kaijuan Wang.(2021). Identification and epidemiological evaluation of gastric cancer risk factors: based on a field synopsis and meta-analysis in Chinese population.
[7] Hongwei Sun, Xiaoli Wu, Fang Wu, Ying Li, Zhengping Yu, Xiangrong Chen, Yunzhi Chen, Wenjun Yang.(2015). Associations of Genetic Variants in the PSCA, MUC1 and PLCE1 Genes with Stomach Cancer Susceptibility in a Chinese Population.
[8] Xuan Jing, Hongping Liang, Chonghua Hao, Xiaojuan Yang, Xiangrong Cui.(2018). Overexpression of MUC1 predicts poor prognosis in patients with breast cancer.
[9] Andreia Mota Sousa, Margarida Rei, Rita Freitas, Sara Ricardo, Thomas C. Caffrey, Leonor David, Raquel Almeida, Michael A. Hollingsworth, Filipe Santos‐Silva.(2016). Effect of MUC1/β-catenin interaction on the tumorigenic capacity of pancreatic CD133+ cells.
[10] Beom Su Kim, Inchul Lee, Jeong Hwan Yook, Kyuyoung Song, Byung-Sik Kim.(2020). Association between theMUC1rs4072037 Polymorphism and Risk of Gastric Cancer and Clinical Outcomes.
[11] Meng Cai, Shuyang Dai, Wanqing Chen, Changfa Xia, Lingeng Lu, Shuguang Dai, Jun Qi, Minjie Wang, Meilin Wang, Lanping Zhou, Fuhua Lei, Tingting Zuo, Hongmei Zeng, Xiaohang Zhao.(2017). Environmental factors, seven GWAS‐identified susceptibility loci, and risk of gastric cancer and its precursors in a Chinese population.
[12] Luca Giraldi, Maria Benedetta Michelazzo, Dario Arzani, Roberto Persiani, Roberta Pastorino, Stefania Boccia.(2017). MUC1, MUC5AC, and MUC6 polymorphisms, Helicobacter pylori infection, and gastric cancer: a systematic review and meta-analysis.
[13] Miao Li, Liu Huang, Hong Qiu, Qiang Fu, Wen Li, Qianqian Yu, Li Sun, Lihong Zhang, Guangyuan Hu, Junbo Hu, Xianglin Yuan.(2013). Helicobacter pylori Infection Synergizes with Three Inflammation-Related Genetic Variants in the GWASs to Increase Risk of Gastric Cancer in a Chinese Population.
[14] Luis Del Valle, Lu Dai, Hui‐Yi Lin, Zhen Lin, Jungang Chen, Steven R. Post, Zhiqiang Qin.(2021). Role of EIF4G1 network in non‐small cell lung cancers (NSCLC) cell survival and disease progression.
[15] Seiya Yokoyama, Michiyo Higashi, Sho Kitamoto, Monika Oeldorf, Uwe Knippschild, Marko Kornmann, Kosei Maemura, Hiroshi Kurahara, Edwin Wiest, Tomofumi Hamada, Ikumi Kitazono, Yuko Goto‐Koshino, Takashi Tasaki, Tsubasa Hiraki, Kazuhito Hatanaka, Yuko Mataki, Hiroki Taguchi, Shinichi Hashimoto, Surinder K. Batra, Akihide Tanimoto, Suguru Yonezawa, Michael A. Hollingsworth.(2016). Aberrant methylation ofMUC1andMUC4promoters are potential prognostic biomarkers for pancreatic ductal adenocarcinomas.
[16] Xiaocong Wang, Yanmei Li, Huihua Hu, Fangzheng Zhou, Jie Chen, Dongsheng Zhang.(2020). Comprehensive analysis of gene expression and DNA methylation data identifies potential biomarkers and functional epigenetic modules for lung adenocarcinoma.
[17] Ming‐Sheng Teng, Semon Wu, Lung‐An Hsu, Hsin‐Hua Chou, Yu‐Lin Ko.(2021). Pleiotropic Effects of Functional MUC1 Variants on Cardiometabolic, Renal, and Hematological Traits in the Taiwanese Population.
[18] Saumya Mishra, Patrícia Mendonça, Sukhmandeep Kaur, Karam F. A. Soliman.(2025). Silibinin Anticancer Effects Through the Modulation of the Tumor Immune Microenvironment in Triple-Negative Breast Cancer.
[19] Hong Tian, Jiali Yu, Xiaoli Chu, Qi Guan, Juan Liu, Ying Liu.(2024). Unraveling the role of C1GALT1 in abnormal glycosylation and colorectal cancer progression.
[20] Qiang Xie, Shilei Zhao, Wenzhi Liu, Yanwei Cui, Fengzhou Li, Zhuoshi Li, Tao Guo, Wendan Yu, Wei Guo, Wuguo Deng, Chundong Gu.(2021). YBX1 Enhances Metastasis and Stemness by Transcriptionally Regulating MUC1 in Lung Adenocarcinoma.
[21] Yugo Mori, Kaoru Akita, Masakazu Yashiro, Tetsuji Sawada, Kosei Hirakawa, Takeomi Murata, Hiroshi Nakada.(2015). Binding of Galectin-3, a β-Galactoside-binding Lectin, to MUC1 Protein Enhances Phosphorylation of Extracellular Signal-regulated Kinase 1/2 (ERK1/2) and Akt, Promoting Tumor Cell Malignancy.
[22] Yan Li, Yuzhang Zhu, Guiping Dai, Dongjuan Wu, Zhenzhen Gao, Lei Zhang, Yaohua Fan.(2019). Screening and validating the core biomarkers in patients with pancreatic ductal adenocarcinoma.
[23] Seiya Yokoyama, Sho Kitamoto, Michiyo Higashi, Yuko Goto‐Koshino, Taro Hara, Dai Ikebe, Taketo Yamaguchi, Yoshifumi Arisaka, Toru Niihara, Hiroto Nishimata, Sadao Tanaka, Kyoichi Takaori, Surinder K. Batra, Suguru Yonezawa.(2014). Diagnosis of Pancreatic Neoplasms Using a Novel Method of DNA Methylation Analysis of Mucin Expression in Pancreatic Juice.
[24] Yue Geng, Kimberly Yeh, Tait Takatani, Michael R. King.(2012). Three to Tango: MUC1 as a Ligand for Both E-Selectin and ICAM-1 in the Breast Cancer Metastatic Cascade.
[25] Alberto Nicoletti, Federica Vitale, Giuseppe Quero, Mattia Paratore, Claudio Fiorillo, Marcantonio Negri, Angela Carlino, Frediano Inzani, Antonio Gasbarrini, Sergio Alfieri, Lorenzo Zileri Dal Verme.(2023). Immunohistochemical Evaluation of the Expression of Specific Membrane Antigens in Patients with Pancreatic Ductal Adenocarcinoma.
[26] Audrey Bouillez, Viviane Gnemmi, Kelly Gaudelot, Brigitte Hémon, Bélinda Ringot, Nicolas Pottier, François Glowacki, Caroline Butruille, Christelle Cauffiez, Malika Hamdane, Nicolas Sergeant, Isabelle Van Seuningen, Xavier Leroy, Sébastien Aubert, Michaël Perrais.(2014). MUC1-C nuclear localization drives invasiveness of renal cancer cells through a sheddase/gamma secretase dependent pathway.
[27] Rosalia Dieli, Rosa Lioy, Fabiana Crispo, Nicoletta Cascelli, Mara Martinelli Donatella Telesca, Maria Rita Milella, Marco Colella, Simona Loperte, Rosa Lerose. Biomolecules.(2025).The Oncoprotein Mucin 1 in Pancreatic Cancer Onset and Progression: Potential Clinical Implications.

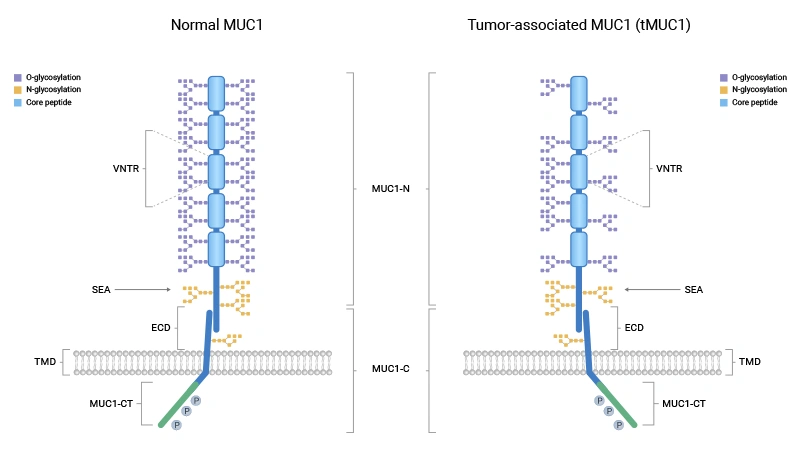


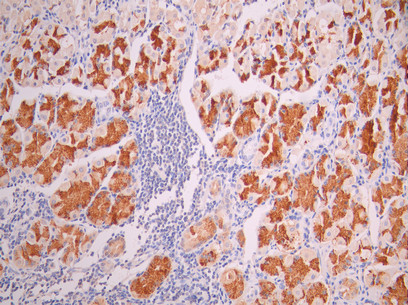


Comments
Leave a Comment