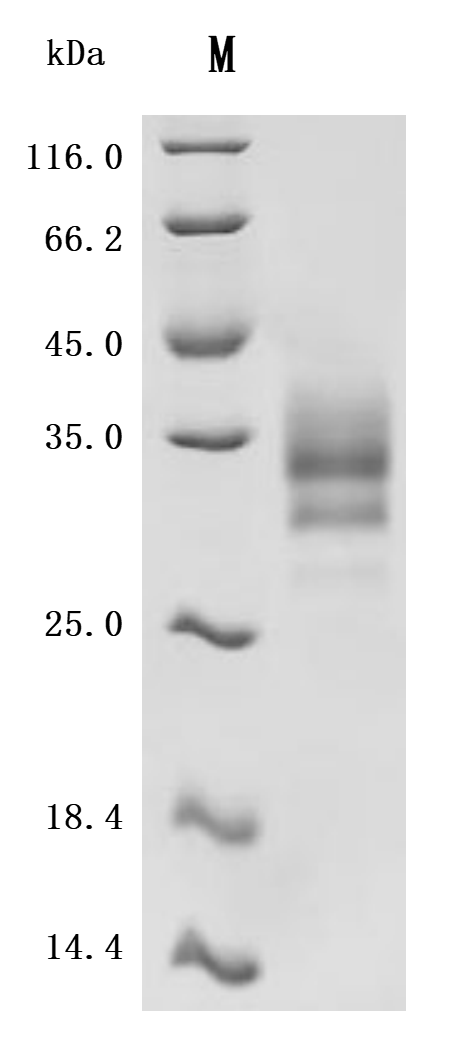
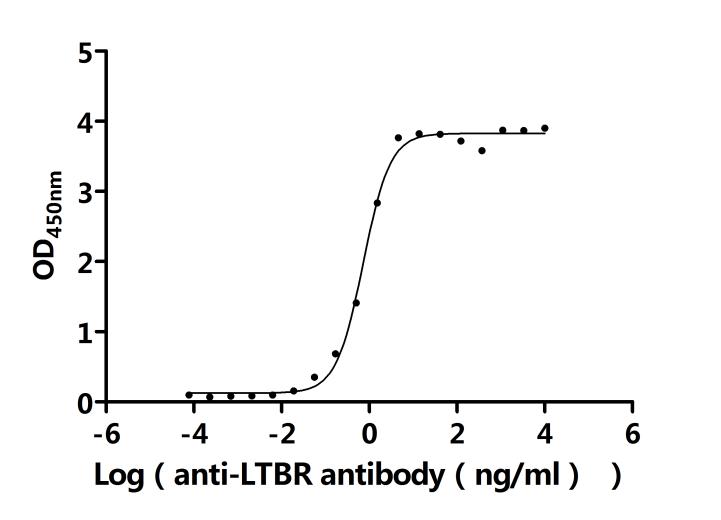
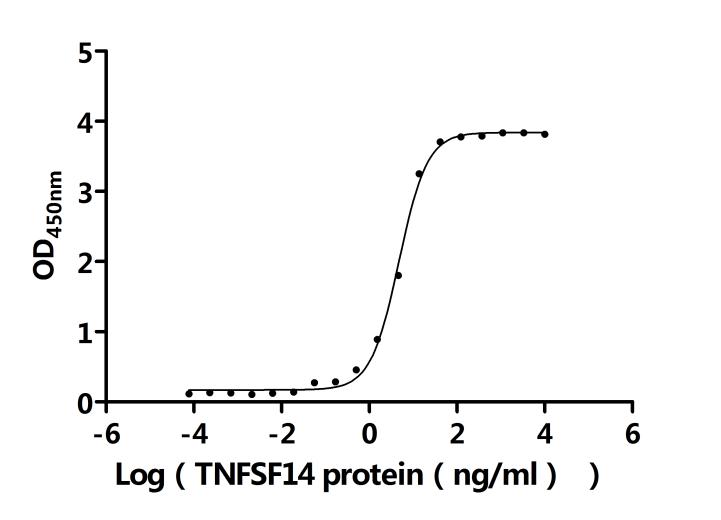
The recombinant human LTBR is an active protein expressed in mammalian cells and consists of the amino acid sequence from positions 31 to 227 of the human LTBR. It is tagged with a C-terminal 10xHis tag, facilitating purification and detection. The endotoxin level of this recombinant LTBR protein is less than 1.0 EU/μg, as determined by the LAL method, ensuring its suitability for various biological applications. LTBR's activity has been assessed through functional ELISA assays. The recombinant LTBR protein, immobilized at a concentration of 2 μg/mL, binds to the anti-LTBR recombinant antibody (CSB-RA013227MA1HU) and human TNFSF14 (CSB-MP023991HUj7-B), with an EC50 of 0.6450 to 0.8200 ng/mL and 4.399 and 5.172 ng/mL, respectively.
The human LTBR protein, also known as TNFR3, is a member of the TNF receptor superfamily and plays a critical role in mediating numerous cellular functions, including survival, proliferation, and apoptosis. TNFR3 is activated by ligands such as Oncostatin M (OSM), which is part of the IL-6 family of cytokines and has been shown to influence immune responses and signaling pathways linked to inflammation and cancer progression [1]. TNFR3 engages in canonical and non-canonical signaling pathways, allowing for diverse effects, such as modulating immune cell responses and contributing to cell survival mechanisms [2].
Evidence indicates that TNFR3 signaling is intricately connected with regulating NF-κB pathways, which are crucial for immune responses and inflammatory processes. Activation of TNFR3 can lead to the activation of NF-κB family members, including RelA and RelB, facilitating transcriptional responses that shape the behavior of various cell types within the immune system [3]. Such interactions underscore TNFR3's pivotal role in maintaining immune homeostasis and responding to pathological challenges.
Dysregulation of TNFR3 signaling has been implicated in various diseases, including cancers and autoimmune disorders, highlighting its potential as a therapeutic target. Studies have shown that aberrant TNFR3 signaling can promote tumor survival and contribute to inflammation within the tumor microenvironment [4].
References:
[1] T. Toyama, S. Xu, R. et al. The nuclear protein hoxb13 enhances methylmercury toxicity by inducing oncostatin m and promoting its binding to tnfr3 in cultured cells. Cells, vol. 9, no. 1, p. 45, 2019. https://doi.org/10.3390/cells9010045
[2] A. Jung, G. Kim, et al. Hmgb1/tnfr1/nf-κb axis in prostate cancer: clinical and biological correlation., 2020. https://doi.org/10.21203/rs.3.rs-40868/v1
[3] M. Jamshidi, R. Fagerholm, et al. Snp-snp interaction analysis of nf-κb signaling pathway on breast cancer survival. Oncotarget, vol. 6, no. 35, p. 37979-37994, 2015. https://doi.org/10.18632/oncotarget.4991
[4] J. Almaden, R. Tsui, et al. A pathway switch directs baff signaling to distinct nfκb transcription factors in maturing and proliferating b cells. Cell Reports, vol. 9, no. 6, p. 2098-2111, 2014. https://doi.org/10.1016/j.celrep.2014.11.024