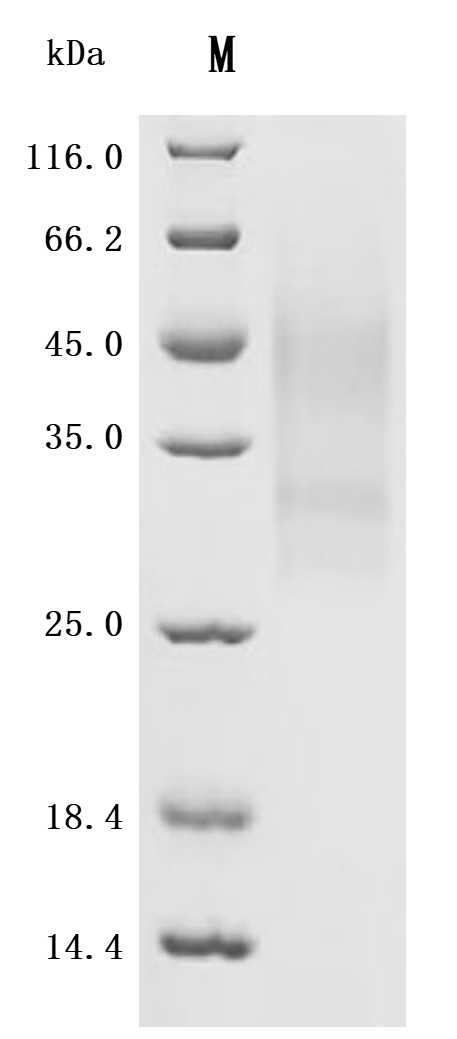
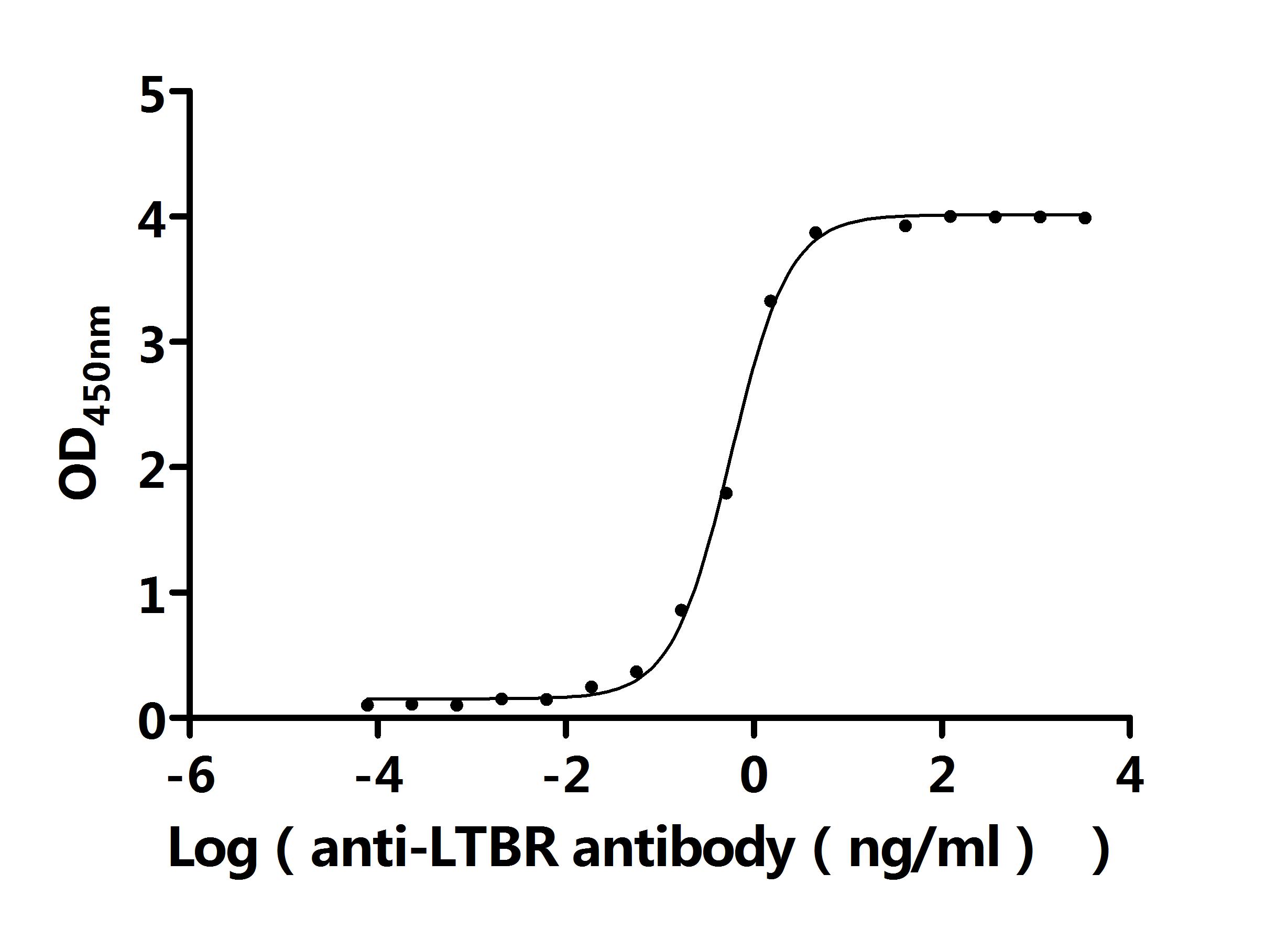
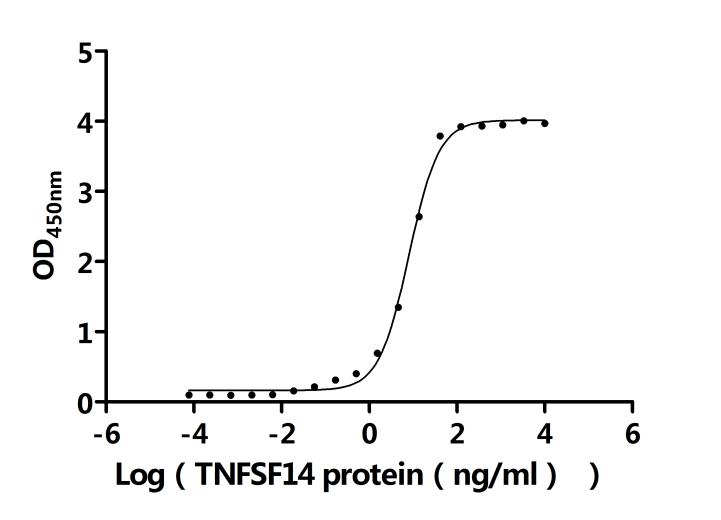
The recombinant human LTBR protein is an active, lyophilized product expressed in a yeast system, designed to preserve its structural integrity and functionality. It comprises amino acids 31 to 227 of the native LTBR sequence, corresponding to its extracellular domain, and is tagged with an N-terminal 6xHis for efficient purification and detection. Endotoxin levels are strictly controlled and kept below 1.0 EU/µg, as verified by the LAL assay. Functional assays confirm its bioactivity. When immobilized at 2 μg/mL, LTBR binds specifically to both the anti-LTBR recombinant antibody (CSB-RA013227MA1HU), with an EC50 of 0.5282–0.6120 ng/mL, and to human TNFSF14 (CSB-MP023991HUj7-B), with an EC50 of 7.283–8.859 ng/mL. These results demonstrate the protein's preserved bioactivity and suitability for receptor-ligand interaction studies, immune pathway research, and therapeutic antibody development.
The LTBR is a crucial member of the tumor necrosis factor (TNF) receptor superfamily, specifically identified as TNFRSF3. The human LTBR gene is located on chromosome 12, near other TNF receptor genes, and encodes a protein structure consisting of an extracellular domain, a transmembrane region, and an intracellular domain [1]. This receptor is involved in various biological processes, including immune responses, inflammation, and cancer progression [1],[2].
The primary ligand for LTBR is lymphotoxin-beta (LTB), which, upon binding, activates several downstream signaling pathways. This interaction induces the expression of various cytokines such as IL-8 [3]. Research indicates that LTBR signaling can enhance the production of cytokines like IL-22BP in human monocyte-derived dendritic cells, thereby linking it to immune modulation and potential anti-tumor effects [4]. Moreover, LTBR's signaling functions also significantly influence tumor biology. For instance, evidence shows that LTBR activation can enhance oncogenic signaling in liver cancer, particularly by sustaining AKT activation in cancer cells [2].
LTBR has also been implicated in the development of physiological conditions like chronic hepatitis C infection. Studies report that LTBR signaling can activate the noncanonical NF-kB pathway, influencing liver disease progression and tumorigenesis [5], [6]. The receptor modulates cell survival and apoptosis, relevant features in various cancers, by interacting with several adapter proteins and pathways, including TRAF2, which is crucial for NF-kB signaling [7].
LTBR expression is associated with distinct cellular states. For instance, its upregulation has been noted in particular immune cell types, influencing their functional capabilities, such as enhancing T cell effector functions while preventing exhaustion [8]. The functional context of LTBR thus continues to be a significant area of research, particularly concerning its therapeutic potential in oncology and immune-related therapies [9].
References:
[1] M. Fernandes, E. Dejardin, & N. Santos. Context-dependent roles for lymphotoxin-β receptor signaling in cancer development. Biochimica Et Biophysica Acta (Bba) - Reviews on Cancer, vol. 1865, no. 2, p. 204-219, 2016. https://doi.org/10.1016/j.bbcan.2016.02.005
[2] A. Scarzello, Q. Jiang, et al. Ltβr signalling preferentially accelerates oncogenic akt-initiated liver tumours. Gut, vol. 65, no. 10, p. 1765-1775, 2015. https://doi.org/10.1136/gutjnl-2014-308810
[3] Y. Mikami, H. Matsuzaki, et al. Lymphotoxin β receptor signaling induces il-8 production in human bronchial epithelial cells. Plos One, vol. 9, no. 12, p. e114791, 2014. https://doi.org/10.1371/journal.pone.0114791
[4] J. Kempski, A. Giannou, et al. Il22bp mediates the antitumor effects of lymphotoxin against colorectal tumors in mice and humans. Gastroenterology, vol. 159, no. 4, p. 1417-1430.e3, 2020. https://doi.org/10.1053/j.gastro.2020.06.033
[5] Q. Chen, X. Lu, & X. Zhang. Noncanonical nf-κb signaling pathway in liver diseases. Journal of Clinical and Translational Hepatology, vol. 000, no. 000, p. 1-9, 2020. https://doi.org/10.14218/jcth.2020.00063
[6] D. Tai, S. Tsai, et al. Activation of nuclear factor κb in hepatitis c virus infection: implications for pathogenesis and hepatocarcinogenesis. Hepatology, vol. 31, no. 3, p. 656-664, 2000. https://doi.org/10.1002/hep.510310316
[7] Q. Xiang, L. Chen, et al. Traf5 and traf3ip2 gene polymorphisms are associated with behçet's disease and vogt-koyanagi-harada syndrome: a case-control study. Plos One, vol. 9, no. 1, p. e84214, 2014. https://doi.org/10.1371/journal.pone.0084214
[8] J. Kotov, Y. Xu, N. Carey, & J. Cyster. Ltβr overexpression promotes plasma cell accumulation. Plos One, vol. 17, no. 8, p. e0270907, 2022. https://doi.org/10.1371/journal.pone.0270907
[9] X. Wang, T. Zhang, et al. Lymphotoxin-β promotes bone colonization and osteolytic outgrowth of indolent bone metastatic cells of breast cancer., 2023. https://doi.org/10.1101/2023.08.15.553179