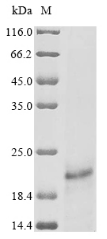
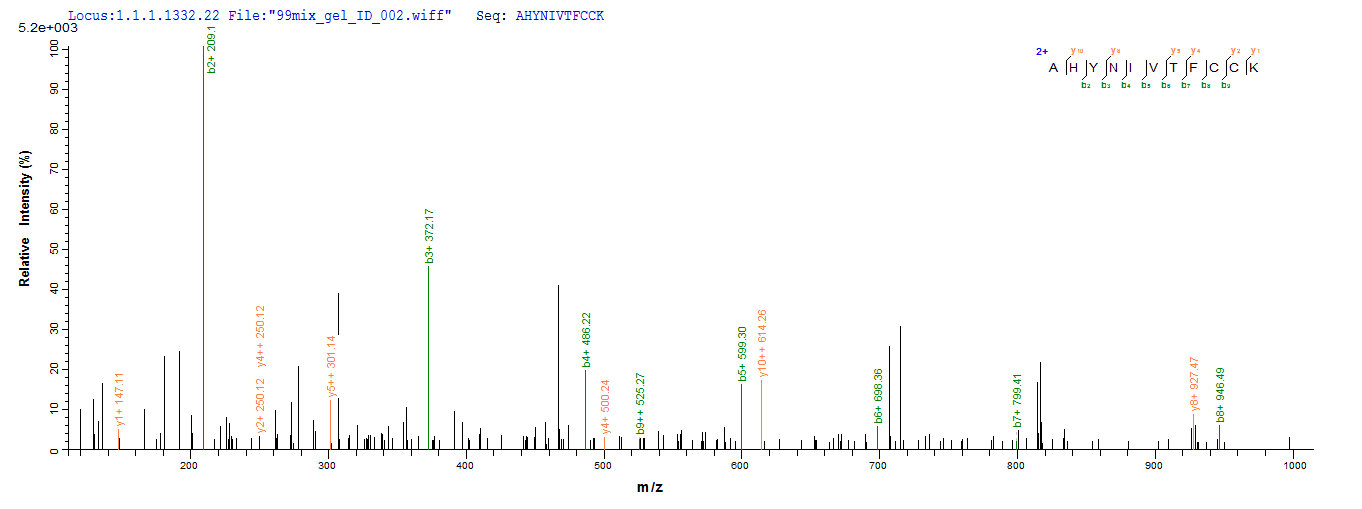
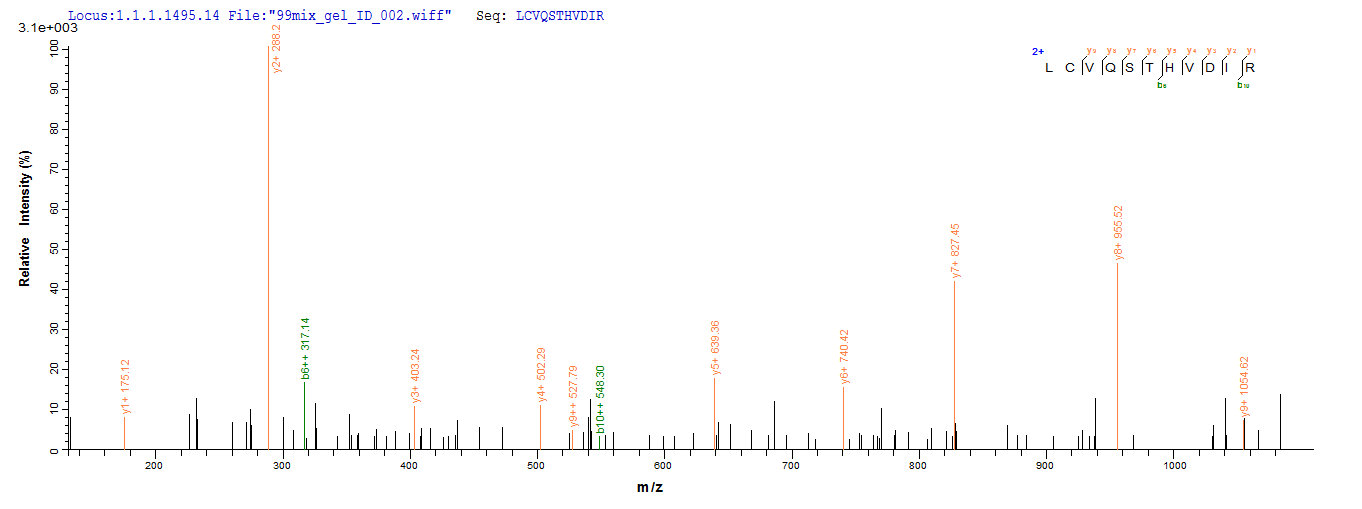
Protein E7 is a crucial oncoprotein found in high-risk human papillomavirus (HPV) types, such as HPV-16. E7 is a small phosphoprotein that interacts with various cellular proteins to promote cellular transformation. It shares sequence homology with adenovirus E1a protein and simian virus 40 large T antigen [1]. E7 exerts its functions by primarily interacting with the retinoblastoma protein (pRB) and other cellular targets, leading to the inactivation of Rb-related proteins p107 and p130 [2][3]. This inactivation of Rb family proteins by E7 disrupts the normal cell cycle regulation, allowing for uncontrolled cell proliferation [4].
E7 is considered the major transforming protein among HPV oncoproteins and is structurally and functionally similar to adenovirus E1A and SV40 Large T antigen [5]. It binds and destabilizes pRB, p107, and p130, inhibiting their tumor-suppressive functions and promoting cell cycle progression [6]. E7 contains conserved regions, including CR1 at the N-terminal end, CR2 with the LxCxE motif, and CR3 at the C-terminal end, which are crucial for its interactions with molecular targets [7][8].
Moreover, E7 binds to specific domains in the Rb protein, namely RbAB and RbC domains, further interfering with Rb's tumor-suppressive activities [9]. The interaction of E7 with pRB is essential for overcoming cell cycle arrest and promoting cell proliferation [6]. Additionally, E7 can induce abnormal centrosome duplication independently of Rb inactivation, further contributing to its oncogenic potential [4].
References:
[1] N. Gammoh, H. Grm, P. Massimi, & L. Banks, "Regulation of human papillomavirus type 16 e7 activity through direct protein interaction with the e2 transcriptional activator", Journal of Virology, vol. 80, no. 4, p. 1787-1797, 2006. https://doi.org/10.1128/jvi.80.4.1787-1797.2006
[2] A. Helt and D. Galloway, "Destabilization of the retinoblastoma tumor suppressor by human papillomavirus type 16 e7 is not sufficient to overcome cell cycle arrest in human keratinocytes", Journal of Virology, vol. 75, no. 15, p. 6737-6747, 2001. https://doi.org/10.1128/jvi.75.15.6737-6747.2001
[3] A. Hall and K. Alexander, "Rna interference of human papillomavirus type 18 e6 and e7 induces senescence in hela cells", Journal of Virology, vol. 77, no. 10, p. 6066-6069, 2003. https://doi.org/10.1128/jvi.77.10.6066-6069.2003
[4] S. Duensing and K. Münger, "Human papillomavirus type 16 e7 oncoprotein can induce abnormal centrosome duplication through a mechanism independent of inactivation of retinoblastoma protein family members", Journal of Virology, vol. 77, no. 22, p. 12331-12335, 2003. https://doi.org/10.1128/jvi.77.22.12331-12335.2003
[5] T. Prathapam, C. Kühne, & L. Banks, "The hpv-16 e7 oncoprotein binds skip and suppresses its transcriptional activity", Oncogene, vol. 20, no. 52, p. 7677-7685, 2001. https://doi.org/10.1038/sj.onc.1204960
[6] M. Giarrè, S. Caldeira, I. Malanchi, F. Ciccolini, M. Leão, & M. Tommasino, "Induction of prb degradation by the human papillomavirus type 16 e7 protein is essential to efficiently overcome p16 ink4a-imposed g1 cell cycle arrest", Journal of Virology, vol. 75, no. 10, p. 4705-4712, 2001. https://doi.org/10.1128/jvi.75.10.4705-4712.2001
[7] H. Yun, M. Kim, H. Lee, W. Kim, J. Shin, H. Kimet al., "Structural basis for recognition of the tumor suppressor protein ptpn14 by the oncoprotein e7 of human papillomavirus", Plos Biology, vol. 17, no. 7, p. e3000367, 2019. https://doi.org/10.1371/journal.pbio.3000367
[8] C. Bello-Rios, S. Montaño, O. Garibay-Cerdenares, L. Araujo-Arcos, M. Leyva-Vázquez, & B. Illades-Aguiar, "Modeling and molecular dynamics of the 3d structure of the hpv16 e7 protein and its variants",, 2020. https://doi.org/10.20944/preprints202012.0671.v1
[9] L. Chemes, I. Sánchez, C. Smal, & G. Prat‐Gay, "Targeting mechanism of the retinoblastoma tumor suppressor by a prototypical viral oncoprotein", Febs Journal, vol. 277, no. 4, p. 973-988, 2010. https://doi.org/10.1111/j.1742-4658.2009.07540.x