Lately, the journal Nature Communications published a paper entitled "Alk1 acts in non-endothelial VE-cadherin+ perineurial cells to maintain nerve branching during hair homeostasis" [1], emphasizing the importance of TGF-β receptor Alk1 in nerve branching maintenance. Alk1, also known as ACVRL1, belongs to the TGF-β receptor family and has received attention as a newly reported anti-angiogenic target. Although the regulatory mechanisms of Alk1 in multiple signaling pathways have not been fully elucidated, successive studies have revealed that ACVRL1 (Alk1) plays an important role in a variety of solid tumors and angiogenesis.
Currently, targeting strategies for ACVRL1 have been explored and targeting ACVRL1 has demonstrated research potential in improving the symptoms of related diseases. Therefore, ACVRL1 (Alk1), as a novel anti-angiogenic drug target, is expected to be an effective target in a variety of research areas in the future, bringing new possibilities and opportunities for drug research in a variety of diseases!
1. What is the TGF-β Family?
The TGF-β family is a class of structurally and functionally related transforming growth factors, including TGF-β, activin, and bone morphogenic proteins (BMPs). TGF-β is present in the TGF-β1, TGF-β2, and TGF-β3 three subtypes. Among them, TGF-β1 is its main existent form and has the strongest biological activity. BMPs belong to the TGF-β superfamily members. There are about 20 kinds of them, such as BMP2 and 4, 5, 6, 7, 8a and 8b, BMP9 and BMP10, BMP12, BMP13, and BMP14. TGF-β receptors (TβR) are widely found on the surface of normal and tumor cells, including type I (TβRI), type II (TβRII), type III TβRIII and endoglin. Two types of receptors, type I and type II, have been studied more frequently and are involved in a variety of signal transduction. To date, seven type I TGF-beta receptors (TβRI) have been identified, named activin receptor-like kinase 1-7 (ALK1-7) (click here to view "TGF-beta receptor" feature article) [2-4].
2. What is ACVRL1?
2.1 ACVRL1 Structure
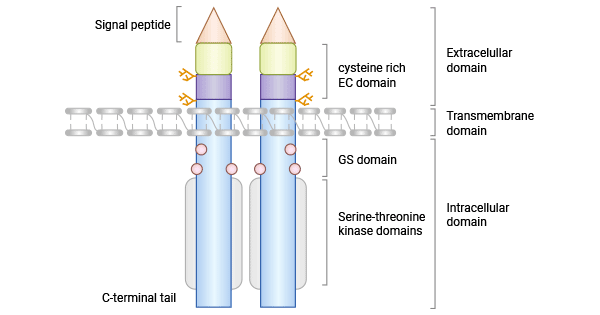
Activin receptor-like kinase-1 (ACVRL1/ALK1) is a member of the TGF-β family's type I receptor (TβRI or ALK), functioning as a serine/threonine kinase receptor. The ACVRL1/ALK1 gene is positioned at 12q13, encompassing 10 exons within a genome length of approximately 15 kilobases. This gene encodes the ACVRL1 receptor protein, a homodimeric transmembrane glycoprotein composed of 503 amino acid residues. Structurally, ACVRL1 comprises three segments: the N-terminal extracellular region, the transmembrane region, and an intracellular segment largely constituted by a serine-threonine kinase region. ACVRL1 exhibits significant similarity with other type I receptors in crucial domains such as the serine-threonine kinase sub-structure, the GS structural domain (a glycine- and serine-rich region preceding the kinase domain), and the C-terminal tail (Figure 1) [5-6].
2.2 ACVRL1 Expression and Function
ACVRL1, a key receptor in the TGF-β family, is notably found in endothelial cells, lungs, and placenta. When activated, it drives endothelial cell growth, crucial for blood vessel formation (angiogenesis) and vascular development. This receptor mainly binds to transforming growth factors (TGF-βs), regulating various cell activities like growth, differentiation, migration, and survival. For example, ACVRL1 aids muscle repair in skeletal cells and accelerates wound healing in fibroblasts [7-8].
Moreover, as a vital signaling molecule, ACVRL1 assumes a pivotal role in various physiological and pathological pathways. Ongoing investigations underscore the association between aberrant ACVRL1 expression and the initiation as well as progression of diverse cancers, accentuating its paramount significance in tumor pathogenesis [7-8].
Figure 1. The structure of ACVRL1/ALK1 [5]
3. What is the Ligand for ACVRL1?
The ligands for ACVRL1 originate from the TGF-β family, with TGF-β itself having a low affinity for ALK1. Conversely, BMP and BMP10 are recognized as high-affinity physiological ligands for ACVRL1/ALK1. However, these ligands don't directly trigger the serine/threonine kinase activity of ACVRL1/ALK-1; they rely on TβR-II or activin II-type receptors to complete the activation process [6, 9].
Upon ligand binding to the TGF-β receptor, the type I receptor undergoes phosphorylation and activation, subsequently activating other type I receptors, including ACVRL1/ALK-1. This forms a receptor complex that further stimulates downstream signaling pathways. Consequently, ACVRL1/ALK-1 activation isn't solely dictated by its ligand but rather by its interaction with other receptors. Thus, ACVRL1/ALK-1 is regarded as an "orphan" receptor, regulated by multiple signaling pathways and complexes. This intricate regulatory system enables ACVRL1/ALK1 to undertake diverse and crucial roles in cell signaling and biological effects [6, 9].
4. ACVRL1-Related Signaling Pathways
4.1 ACVRL1-SMAD Signaling Mechanism
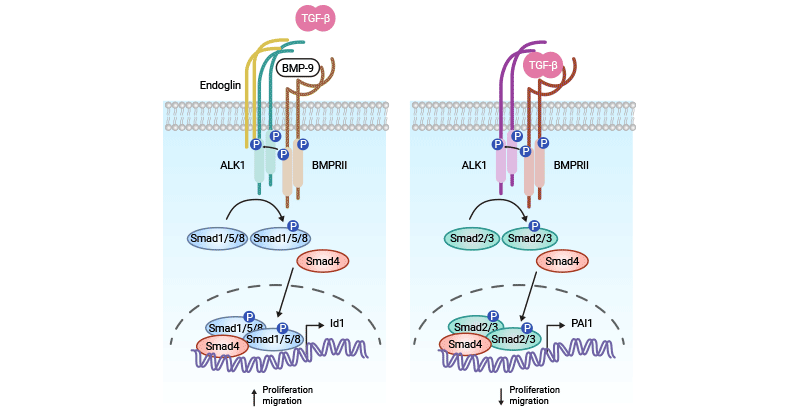
TGF-β engages both ACVRL1 (ALK1) and ALK5 receptors for its functional activity. Initially, TGF-β binds to TβRII, activating TβRI, thus forming a complex that initiates the Smads signaling pathway. Within endothelial cells, this activation leads to divergent effects via two distinct Smad signaling pathways. When BMP-9 (or -10) binds to ACVRL1/ALK1 receptors, it triggers the Smad1/Smad5 signaling cascade, further engaging BMPRII receptors and co-receptors like Endoglin (ENG), culminating in the formation of a cell surface complex. Similarly, active TβRII can recruit and phosphorylate ACVRL1/ALK1, with Endoglin contributing to ligand-ALK1 interactions within the receptor complex [10-11].
The phosphorylation of Smad1/5 (R-Smads) by ALK1 results in the formation of activated R-Smad complexes with Smad4, translocating to the nucleus. Here, these complexes regulate the transcription of specific target genes, such as ID1 and ID3. These regulatory actions prompt increased endothelial cell proliferation and migration. Conversely, TGF-β can activate ALK5 receptors dependent on TβRII, leading to heightened Smad2/Smad3 phosphorylation. Activated Smad2/3 also form complexes with Smad4, entering the nucleus to regulate gene transcription, for instance, affecting genes like PAI1, which exert inhibitory effects on endothelial cell proliferation and migration (Figure 2) [10-11].
Figure 2. ACVRL1-related signaling pathways [11]
4.2 ACVRL1 Anti-Tumor Angiogenesis Mechanism
ALK1, a type I receptor in the TGF-β/BMPs family, significantly influences angiogenesis and vessel formation. Recent research suggests inhibiting the ALK1 pathway as a key strategy against tumor-driven angiogenesis. Studies reveal its vital role in embryonic liver vascular development through interaction with SMAD6 [12].
In embryonic liver tissues, SMAD6 absence leads to ALK1 over-activation, causing vascular malformations and hemorrhaging. Reducing Alk1 gene dosage in experiments rescued the hemorrhaging caused by Smad6 deletion. ALK1 over-activation destabilizes endothelial cell junctions and increases contractility, resulting in fragile vessel walls prone to rupture. This underscores ALK1's crucial role as a "guardian" in embryonic liver vascularization. Loss of ALK1 function leads to vascular malformations and hemorrhaging, highlighting the necessity of balanced ALK1 signaling for proper vascular development [12].
5. The Roles of ACVRL1 in Diseases Research
5.1 ACVRL1 and Hereditary Hemorrhagic Trichotillomania (HHT) Research
ACVRL1 (ALK1), a transmembrane receptor, plays a pivotal role in signaling pathways crucial for angiogenesis and vascular development. Research on inhibiting ALK1 signaling shows promise in addressing various angiogenesis-related conditions, including hereditary hemorrhagic telangiectasia (HHT). HHT, an autosomal dominantly inherited disorder, involves abnormal vascular development and presents clinical phenotypes categorized as HHT1, HHT2, and juvenile polyposis with HHT (JPHT). Mutations in genes such as ALK-1, ENG (endoglin, CD105), and SMAD4 have been identified as causative factors for HHT. Specifically, mutations in the ENG gene lead to the HHT1 phenotype, ALK-1 gene mutations relate to the HHT2 phenotype, and SMAD4 gene mutations correspond to the JPHT phenotype. Hence, targeting ACVRL1 (ALK1) inhibition holds promise as a significant approach in researching various angiogenesis-related diseases, including HHT [13].
5.2 ACVRL1 and Systemic Sclerosis (SSc) Research
A study examined ACVRL1 (ALK1) expression in skin fibroblasts from individuals with systemic sclerosis (SSc), a disorder causing excessive tissue matrix deposition, fibrosis, and organ dysfunction. SSc often involves vascular damage and immune system activation. The research revealed notably higher ALK1 levels in SSc patients compared to healthy individuals. ALK1 was primarily found in the cytosol and cytoplasm of fibroblasts using immunofluorescence. Experimentally reducing ALK1 levels in fibroblasts resulted in decreased expressions of ALK1, fibronectin, and PAL-1. Conversely, treating control groups with TGFβ1 significantly increased these protein levels. These findings suggest TGFβ1 influences fibronectin and PAI-1 expression through ALK1 in fibroblasts, indicating ALK1's significant role in the fibrotic process linked with systemic sclerosis [14].
5.3 ACVRL1 and Tumor Research
Presently, there's limited research concerning the ACVRL1 (ALK1) gene in tumors, but diverse experiments confirm its close association with tumor development and progression. In colorectal cancer (CRC) treatment, the efficacy of multi-target tyrosine kinase inhibitors (mTKIs) like regorafenib and sorafenib remains constrained. Recent experiments propose an ACVRL1-mediated resistance mechanism contributing to this limitation. Manipulating ACVRL1 expression significantly influences CRC cell sensitivity to mTKIs ex vivo, suggesting that targeting ACVRL1 could overcome mTKI resistance effectively [15].
In a phase Ib study evaluating the safety and efficacy of Dalantercept combined with Sorafenib for advanced hepatocellular carcinoma (HCC), Dalantercept, a soluble ALK1 receptor fusion protein, hampers ALK1 signaling by binding to BMP9 and BMP10, impeding mature blood vessel formation. While the combination's antitumor activity is modest, the novelty of targeting ALK1 in HCC has captured researchers' interest [16]. Moreover, in non-small cell lung cancer (NSCLC), studies indicate specific upregulation of ALK1 mRNA and protein in NSCLC tissues compared to normal lung tissues. This suggests the potential utility of the ALK1 gene as a molecular target or a serological tumor marker for targeted therapy in NSCLC [17].
5.4 ACVRL1 and Other Diseases Research
ACVRL1's significance extends beyond tumors to various diseases. In neurological studies, research targeting ALK1 in brain-related aspects revealed that TGF-β1-ALK1p-Smad1 contributes to neuroprotection, while TGF-β1-ALK5-p-Smad2/3 promotes epilepsy. In damaged brain tissue, neurons and astrocytes engage through the TGF-β1-ALK1-p-Smad1 and TGF-β1-ALK5-p-Smad2/3 pathways [18]. Moreover, investigations explored LRG-TGF-β-Alk-1/TGFβRII-Smads as predictive biomarkers for chronic hydrocephalus post-aneurysmal subarachnoid hemorrhage [19].
Regarding BMP9-induced osteogenesis, in vitro and in vivo experiments underscored the importance of ALK1 and ALK2. In vitro, siRNA targeting ALK1 and ALK2 effectively inhibited alkaline phosphatase, calcium salts, and luciferase activities, key indicators validating BMP9 signaling and osteogenesis. Remarkably, inhibiting ALK1 and ALK2 not only hampered BMP9 signaling but also impeded the differentiation of bone marrow mesenchymal stem cells into osteoblasts. These findings highlight ALK1's crucial role in modulating BMP9-mediated osteoblast differentiation, providing a strong basis for future therapies targeting bone-related conditions [20].
6. The Prospects for Clinical Drug Research on ACVRL1
Three drugs currently in development target ACVRL1 (ALK1): Ascrinvacumab, a monoclonal antibody; ALK-1/VEGF bispecific antibody, a bispecific antibody; and TATX-21, another monoclonal antibody. Suzhou Pioneering Pharmaceuticals and Pfizer collaborate on the first two, while TATX-21 is a joint effort of ImmunoPrecise Antibodies and Talem Therapeutics. The earlier drug, Dalantercept, is now in the termination phase. Ascrinvacumab is in clinical trials in China, while the other two are in early research stages overseas. These drugs have diverse focuses: Ascrinvacumab and ALK-1/VEGF bispecific antibody target solid tumors, whereas TATX-21 aims to address atherosclerosis and heart disease. Overall, targeting ALK1 for anti-tumor angiogenesis signifies potential advancements in cardiovascular diseases, immune system disorders, and tumor research!
In conclusion:
ACVRL1 (ALK1) a crucial receptor within the TGF-β/BMPs family, holds diverse roles in angiogenesis, vascular development, and tumor pathophysiology. Its implications span various diseases like systemic sclerosis, neurological conditions, and osteogenesis. Current research explores ALK1 as a therapeutic target in multiple diseases. Several drugs targeting ALK1 are in development stages, showing promise against solid tumors, atherosclerosis, and heart disease. ALK1's intricate involvement in disease pathways signifies its potential in advancing research and interventions across cardiovascular diseases, immune disorders, and tumor-related angiogenesis.
7. CUSABIO ACVRL1 Recombinant Proteins & Antibodies for Research Use
To fully support researchers and pharmaceutical companies in their research on ACVRL1 (ALK1) in cardiovascular diseases, immune disorders, and tumor research, CUSABIO presents ACVRL1 active proteins & antibodies to support your research on the mechanism of ACVRL1 or its potential clinical value.
References
[1] Chovatiya, Gopal, et al. "Alk1 acts in non-endothelial VE-cadherin+ perineurial cells to maintain nerve branching during hair homeostasis." Nature Communications 14.1 (2023): 5623.
[2] Bierie, Brian, and Harold L. Moses. "TGF-β and cancer." Cytokine & growth factor reviews 17.1-2 (2006): 29-40.
[3] Tzavlaki, Kalliopi, and Aristidis Moustakas. "TGF-β Signaling." Biomolecules 10.3 (2020): 487.
[4] Vander Ark, Alexandra, Jingchen Cao, and Xiaohong Li. "TGF-β receptors: In and beyond TGF-β signaling." Cellular signalling 52 (2018): 112-120.
[5] Munoz-Felix, Jose M., Maria Gonzalez-Nunez, and Jose M. Lopez-Novoa. "ALK1-Smad1/5 signaling pathway in fibrosis development: friend or foe?." Cytokine & growth factor reviews 24.6 (2013): 523-537.
[6] Bhave, Shreyas, et al. "ALK1 Deficiency Impairs the Wound-Healing Process and Increases Mortality in Murine Model of Myocardial Infarction." Journal of Cardiovascular Translational Research (2023): 1-9.
[7] Li, Kefei Nina, et al. "Blood endothelial ALK1‐BMP4 signaling axis regulates adult hair follicle stem cell activation." The EMBO Journal (2023): e112196.
[8] Johnston, E. D., W. Wang, and K. Lyons. "A single-cell rna sequencing analysis of alk1 and related bmp pathway proteins in osteoarthritis progression." Osteoarthritis and Cartilage 31.5 (2023): 673-674.
[9] Chu, Kit Yee. Elucidating the Role of the TGF-β Superfamily Signaling in Pulmonary Vascular Cell Proliferation. Diss. Michigan State University, 2023.
[10] Finnson, Kenneth W., et al. "ALK1 opposes ALK5/Smad3 signaling and expression of extracellular matrix components in human chondrocytes." Journal of bone and mineral research 23.6 (2008): 896-906.
[11] Hawinkels, Lukas JAC, Amaya Garcia de Vinuesa, and Peter Ten Dijke. "Activin receptor-like kinase 1 as a target for anti-angiogenesis therapy." Expert opinion on investigational drugs 22.11 (2013): 1371-1383.
[12] Kulikauskas, Molly R., et al. "Endothelial Cell SMAD6 Balances ACVRL1/Alk1 Function to Regulate Adherens Junctions and Hepatic Vascular Development." bioRxiv (2023): 2023-03.
[13] Duerig, Inga, et al. "Nonfunctional TGF-β/ALK1/ENG signaling pathway supports neutrophil proangiogenic activity in hereditary hemorrhagic telangiectasia." Journal of Leukocyte Biology 114.6 (2023): 639-650.
[14] Morris, Erin, et al. "Endoglin promotes TGF‐β/Smad1 signaling in scleroderma fibroblasts." Journal of cellular physiology 226.12 (2011): 3340-3348.
[15] Lu, Xiaolin, et al. "ACVRL1 drives resistance to multitarget tyrosine kinase inhibitors in colorectal cancer by promoting USP15-mediated GPX2 stabilization." BMC medicine 21.1 (2023): 366.
[16] Abou‐Alfa, Ghassan K., et al. "A phase Ib, open‐label study of dalantercept, an activin receptor‐like kinase 1 ligand trap, plus sorafenib in advanced hepatocellular carcinoma." The Oncologist 24.2 (2019): 161-e70.
[17] Casaluce, Francesca, et al. "ALK inhibitors: a new targeted therapy in the treatment of advanced NSCLC." Targeted oncology 8 (2013): 55-67.
[18] Cacheaux, Luisa. A Novel Role for TGF-Beta Signaling in Epileptogenesis. Diss. UC Berkeley, 2010.
[19] Ma, Dongying, et al. "The LRG-TGF-β-Alk-1/TGFßRII-Smads as Predictive Biomarkers of Chronic Hydrocephalus after Aneurysmal Subarachnoid Hemorrhage." Journal of Neurological Surgery Part A: Central European Neurosurgery (2023).
[20] Seong, Chang‐Hwan, et al. "Early induction of Hes1 by bone morphogenetic protein 9 plays a regulatory role in osteoblastic differentiation of a mesenchymal stem cell line." Journal of Cellular Biochemistry 124.9 (2023): 1366-1378.
CUSABIO team. ACVRL1 (Alk1): a Member of the TGF-β Receptor Family, a Novel Target for Anti-Angiogenic Drug Research!. https://www.cusabio.com/c-21159.html



Comments
Leave a Comment