On November 9, 2020, Quest PharmaTech Inc. announced that Oregovomab, a MUC16 monoclonal antibody, has been dosed in the patients with ovarian cancer in phase III clinical trial. It means that targeting MUC16 drugs will bring new hope in batter against ovarian cancer. It is also worth noting that the role of MUC16 in other tumors has been further explored in recent years, such as an article published in Molecular Therapy (Impact Factor: 8.986) on December 25, 2020, which revealed a novel regulatory mechanism of MUC16 in pancreatic cancer [1]. So what is MUC16? What is its main function? What is the prospect of targeting MUC16 in cancer treatment? After reading this article, you will get something out of it.
1. Structure of MUC16
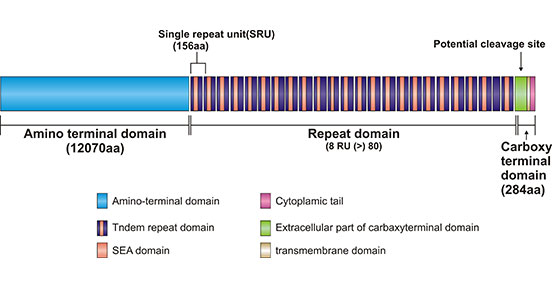
MUC16, also known as CA125, is a member of the mucin (MUC) family and belongs to the transmembrane type I. MUC16, as the largest glycoprotein in the MUC family, is located on human chromosome 19p13.2 and is encoded by 179 kb of genomic DNA, encoding a total of 22,152 amino acids [2]. The protein is composed of the following structural domains: the N-terminal domain, containing approximately 12,070 amino acids, rich in serine or threonine; the tandem repeat domain, containing 60+ repeats of 156 amino acid and 16 repeats of SEA domain; the C- terminal domain, also called the transmembrane domain, containing 284 amino acids, which can be subdivided into extracellular, transmembrane, and cytoplasmic tail (CT). Among them, the CT is composed of 32 amino acids that provide potential sites for phosphorylation, which may be involved in certain signaling pathways [3, 4] (Figure 1).
Figure 1. Schematic diagram of MUC16 structure
*The figure is derived from Biomolecules publication [4]
Unlike other transmembrane mucins, MUC16 does not have an EGF (epidermal growth factor)-like structural domain or 16 SEA domains, whereas some other MUCs that contain only one or a few SEA domains [3] . The particular structure of MUC16 may activate certain tumor signaling pathways [5].
2. Expression and Function of MUC16
MUC16 is a critical element of the ocular surface (including the cornea and conjunctiva), the respiratory tract, and the female reproductive tract epithelia [6]. MUC16 is expressed in normal endometrial tissues, especially in glands and epithelial cells, as well as in cervical mucus [7]. Certainly, MUC16 is produced d in several tissues and organs, like the conjunctiva, cornea, middle ear epithelium, and tracheal surface epidermis [8].
Since MUC16 is highly glycosylated, it creates a hydrophilic environment that acts as a lubricating barrier against foreign particles and infectious agents on the apical membrane of epithelial cells [9] . In the ocular surface epithelia, normally expressed MUC16 provides lubrication that helps in movements such as blinking [10].
However, aberrantly expressed MUC16 is related to multiple diseases. For example, high expression of MUC16 is a key factor in the development of dry eye [11]. In addition, expression levels of MUC16 are elevated in a number of benign conditions, such as endometriosis [12], adenomyosis [13], ovarian cysts [14], liver cirrhosis [15], diabetes [16], pleurisy [17], and peritonitis [18]. It is well-known that MUCl6 contributes to ovarian cancer [19]. More importantly, an increasing number of studies have found that aberrantly expressed MUCl6 is associated with progression in several of cancers [1-3, 5].
3. The Role of MUC16 in Tumor Signaling Pathways
As mentioned earlier, MUC16 is the largest glycoprotein. Given its huge mass, limited knowledge of MUC16 in tumor mechanisms was acquired. One research suggested that the interaction between MUC16 and β-catenin could activate the Wnt/β-catenin signaling pathway by facilitating cytosol-nucleus transportation of β-catenin, consequently induce cell proliferation and the migration [20] in ovarian cancer. Another showed that the interaction of MUC16 with tyrosine kinase (JAK2) induces proliferation of breast cancer cells [21].
Indeed, the expression of MUC16 in cells is subject to complex regulatory mechanisms. Previous studies pointed that MUC16 expression is usually restricted by cell polarity of epithelial cells. However, as the loss of cell polarity during carcinogenesis, MUC16 is expressed all over the epithelial cells surface, and MUC16 becomes available to interact with multiple growth factors, which could activate downstream signaling pathways and trigger cancer progression [3].
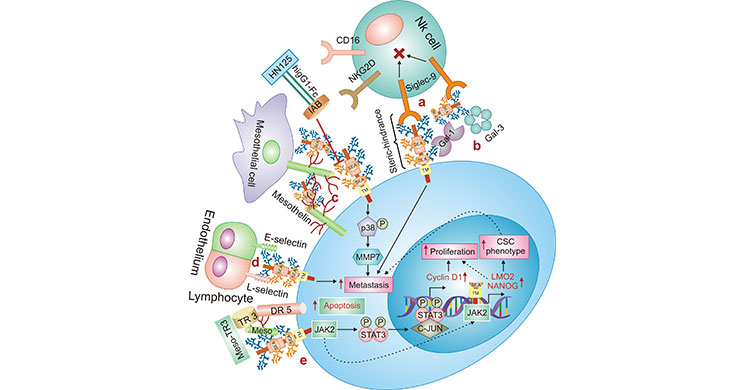
As shown in Figure 2, the main links between MUC16 and tumor signaling pathways are as follows, (a) MUC16-siglec- 9: MUC16 binds to siglec-9, an inhibitory receptor on the surface of NK cells, thus MUC16 could exert immunosuppressive effects and provide immune protection mechanisms for tumor cells; (b) MUC16-galectin-1/3: MUC16 is thought to be a counter-receptor for Galectin, such as Gal-1 and Gal-3, but its role in tumorigenesis remains to be clarified. (c) MUC16-mesothelin: MUC16 interacts with mesothelin, as an important mediator of peritoneal metastasis. (d) MUC16-selectin: MUC16 binds E-selectin or L- selectin to promote tumor cell metastasis. (e) MUC16-JAK2: the interaction of MUC16 with JAK2, on the one hand, it leads to the upregulation of cyclin-D1, which promotes tumor cell proliferation (see solid arrow in the Figure 2); on the other hand, it leads to the upregulation of MO2 and NANOG, which enhances the tumor stem cell phenotype and cell metastasis ability (see dashed arrow in the Figure 2) [22].
Figure 2. Relationship between MUC16 and tumor signaling pathways
* The figure is derived from Cancer research publication [22].
4. Applications and Prospects of Targeting MUC16 in Cancer Treatment
To date, MUC16 is the most widely used and clinically important serum biomarker for the diagnosis of ovarian cancer because it is a tumor-specific antigen overexpressed in ovarian cancer. Recently, the MUC16 monoclonal antibody drug Oregovomab has been used in patients with primary ovarian cancer in phase III clinical trial. Another anti-MUC16 antibody drug (JCAR-020) is in phase I clinical trials for ovarian cancer therapy. Namely, there is no FDA approved MUC16-targeted drug on the market. But the "Ovarian Cancer Drugs Global Market Report 2020" suggested that global ovarian cancer drugs market worth $1.99 billion by 2022-an estimated 55% rise is expected in the number of patients suffering from ovarian cancer by 2035. Therefore, it is believed that more entrants to MUC16-targeted drug research will be attracted to secure their place in future market.
Intriguingly, more and more studies implied that MUC16 is overexpressed in other tumors, including endometrial cancer [23], fallopian tube cancer [24], pancreatic cancer [25], colon cancer [26], peritoneal cancer [27], nasopharyngeal cancer [28], lung cancer [29], breast cancer [30] and gastric cancer [31]. It suggests that MUC16 may be an attractive target for anti-tumor treatment. Due to the short history of MUC16 discovery, the importance of MUC16 in the tumor warrants the need for in-depth research. But so far, the findings suggest that MUC16 is expected to serve as a new predictive marker for more tumors and provide new strategies for anti-cancer therapy.
MUC16 protein
● Recombinant Human Mucin-16(MUC16),partial (Active) (Code: CSB-MP704410HU3c7)
Excellent Bioactivity Validated by Functional ELISA

(Tris-Glycine gel) Discontinuous SDS-PAGE (reduced) with 5% enrichment gel and 15% separation gel.
Excellent Bioactivity Validated by Functional ELISA
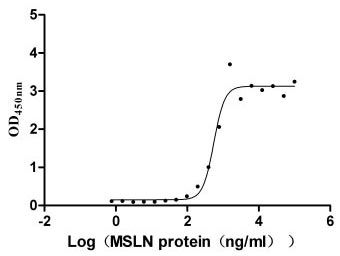
Immobilized MUC16 at 10 μg/ml can bind MSLN(CSB-MP015044HUc9), the EC50 is 460.7-662.2 ng/ml.
References
[1] Thomas, Divya, et al . "Isoforms of MUC16 Activate Oncogenic Signaling Through EGF Receptors to Enhance the Progression of Pancreatic Cancer." Molecular Therapy (2020).
[2] Haridas, Dhanya, et al . "MUC16: molecular analysis and its functional implications in benign and malignant conditions." The FASEB Journal 28.10 (2014): 4183-4199.
[3] Aithal, Abhijit, et al . "MUC16 as a novel target for cancer therapy." Expert opinion on therapeutic targets 22.8 (2018): 675-686.
[4] Hanson, Ryan L., and Michael A. Hollingsworth. "Functional consequences of differential O-glycosylation of MUC1, MUC4, and MUC16 (downstream effects on signaling)." Biomolecules 6.3 (2016): 34.
[5] Haridas, Dhanya, et al . "MUC16: molecular analysis and its functional implications in benign and malignant conditions." The FASEB Journal 28.10 (2014): 4183-4199.
[6] Argüeso, Pablo, et al . "MUC16 mucin is expressed by the human ocular surface epithelia and carries the H185 carbohydrate epitope." Investigative ophthalmology & visual science 44.6 (2003): 2487-2495.
[7] Gipson, Ilene K., et al . "Comparison of the transmembrane mucins MUC1 and MUC16 in epithelial barrier function." PloS one 9.6 (2014): e100393.
[8] Choi, Sangdun, ed. Encyclopedia of Signaling Molecules. Vol. 337. New York: Springer, 2012.
[9] Argüeso, Pablo, et al . "MUC16 mucin is expressed by the human ocular surface epithelia and carries the H185 carbohydrate epitope." Investigative ophthalmology & visual science 44.6 (2003): 2487-2495.
[10] Aithal, Abhijit, et al . "MUC16 as a novel target for cancer therapy." Expert opinion on therapeutic targets 22.8 (2018): 675-686.
[11] Setälä, Niko. "Interrelations between Dry Eye Syndrome and Tear Fluid Phospholipid Transfer Protein." (2011).
[12] Muyldermans, M., F. J. Cornillie, and P. R. Koninckx. "CA125 and endometriosis." Human reproduction update 1.2 (1995): 173-187.
[13] Kil, Kicheol, et al . "Usefulness of CA125 in the differential diagnosis of uterine adenomyosis and myoma." European Journal of Obstetrics & Gynecology and Reproductive Biology 185 (2015): 131-135.
[14] Kurata, Hitoshi, et al . "Elevated serum CA125 and CA19-9 due to the spontaneous rupture of ovarian endometrioma." European Journal of Obstetrics & Gynecology and Reproductive Biology 105.1 (2002): 75-76.
[15] DiBaise, John K., and Jeremiah P. Donovan. "Markedly elevated CA125 in hepatic cirrhosis: two case illustrations and review of the literature." Journal of clinical gastroenterology 28.2 (1999): 159 -161.
[16] Yao, Litong, et al . "Serum CA125 Level Is Associated with Diabetic Retinopathy in Chinese Patients with Type 2 Diabetes." Diabetes, metabolic syndrome and obesity: targets and therapy 13 (2020): 1803.
[17] Nakanishi, Yoichi, et al . "Clinical significance of serum CAl25 in patients with tuberculous pleurisy." Kekkaku (Tuberculosis) 66.8 (1991): 525-530.
[18] Taniwaki, A., et al . "Tuberculous peritonitis with elevated serum CA125 levels mimicking peritoneal cancer." Clinical and Experimental Obstetrics & Gynecology 47.6 (2020): 994-996.
[19] Funston, Garth, et al . "The diagnostic performance of CA125 for the detection of ovarian and non-ovarian cancer in primary care: A population-based cohort study." PLoS medicine 17.10 (2020): e1003295.
[20] Liu, Qi, et al . "C-terminus of MUC16 activates Wnt signaling pathway through its interaction with β-catenin to promote tumorigenesis and metastasis." Oncotarget 7.24 (2016): 36800.
[21] Lakshmanan, I., et al . "MUC16 induced rapid G2/M transition via interactions with JAK2 for increased proliferation and anti-apoptosis in breast cancer cells." Oncogene 31.7 (2012): 805- 817.
[22] Das, Srustidhar, and Surinder K. Batra. "Understanding the unique attributes of MUC16 (CA125): potential implications in targeted therapy." Cancer research 75.22 (2015): 4669-4674.
[23] Hu, Jing, and Jing Sun. "MUC16 mutations improve patients'prognosis by enhancing the infiltration and antitumor immunity of cytotoxic T lymphocytes in the endometrial cancer microenvironment." Oncoimmunology 7.10 (2018): e1487914.
[24] Koneru, Mythili, et al . "A phase I clinical trial of adoptive T cell therapy using IL-12 secreting MUC-16 ecto directed chimeric antigen receptors for recurrent ovarian cancer." Journal of translational medicine 13.1 (2015): 102.
[25] Fan, Kun, et al . "MUC16 C terminal-induced secretion of tumor-derived IL-6 contributes to tumor-associated Treg enrichment in pancreatic cancer." Cancer letters 418 (2018): 167- 175.
[26] Liu, Tong, et al . "Increased serum CA125 II, but not CEA, CA19-9, AFP, or CA72-4 in colon cancer compared to rectal cancer." British Journal of Biomedical Science (2020).
[27] Suidan, Rudy S., et al . "A multicenter prospective trial evaluating the ability of preoperative computed tomography scan and serum CA-125 to predict suboptimal cytoreduction at primary debulking surgery for advanced ovarian, fallopian tube, and peritoneal cancer." Gynecologic oncology 134.3 (2014): 455-461.
[28] Tang, Yong-Fei, et al . "Diagnostic values of serum tumor markers CA72-4, SCCAg, CYFRA21-1, NSE, AFU, CA125, CA19-9, CEA and FER in nasopharyngeal carcinoma." Transl Cancer Res 7 (2018): 1406-12.
[29] Zang, Ruochuan, et al . "ERO1L promotes IL6/sIL6R signaling and regulates MUC16 expression to promote CA125 secretion and the metastasis of lung cancer cells." Cell death & disease 11.10 (2020): 1-15.
[30] Feng, C., et al . "Postoperative expressions of TRACP5b and CA125 in patients with breast cancer and their values for monitoring bone metastasis." Journal of BU ON.: Official Journal of the Balkan Union of Oncology 25.2 (2020): 688-695.
[31] Li, Xiangchun, et al . "Association of MUC16 mutation with tumor mutation load and outcomes in patients with gastric cancer." JAMA oncology 4.12 (2018): 1691-1698.
CUSABIO team. MUC16 (CA125): a Promising Predictive Marker and Therapeutic Target in Cancer. https://www.cusabio.com/c-21013.html





Comments
Leave a Comment