In recent years, immune cell therapy is driving the next-generation treatment for cancer after surgery, drug, and radiotherapy. Undoubtedly, CAR-T therapy is the central component of a host of promising immunotherapy. Currently, seven CAR-T therapies have been approved for hematological tumors, which have greatly stimulated the enthusiasm of researchers to turn their attention to solid tumors. Inspiringly, more and more CAR-T clinical trials have been started for solid tumors. Albeit CAR-T therapy is faced with the inherent obstacles of solid tumors, a growing number of promising coping strategies have gradually emerged. Many studies have focused on improving CAR-T cells and overcoming the unfavorable effects of tumor microenvironment in the hope that CAR-T will also exert an anti-cancer effect in solid tumors. Here, in no particular order, are the most popular targets of CAR-T cell therapy for solid tumors!
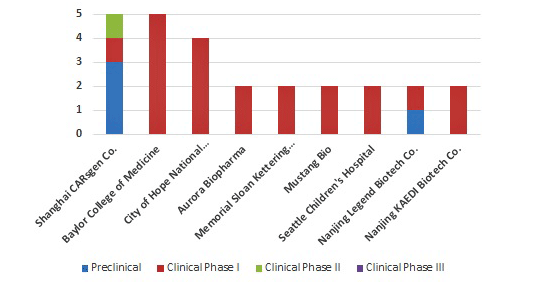
Data from Pharmacodia signify the most favored CAR-T therapy for solid tumors such as Claudin 18.2, GPC3, MSLN, HER2, EGFR, and others. It is important to appreciate that CAR-T development in solid tumors is still in early stages. But, many researchers are involved in investigating the potential use of CAR-T for solid tumor treatment. In terms of the popular CAR-T targets in solid tumors, numerous domestic biotech companies and foreign competitors have constructed product pipelines, including Novartis, Baylor College of Medicine, Atara Biotherapeutics, Cellectis, Shanghai Pharmaceutical Group, Shanghai CARsgen Co., Shanghai Genechem Co., Nanjing Legend Biotech Co., etc.
Numerous biotech companies develop CAR-T cell therapies targeting solid tumors
Source: Pharmacodia
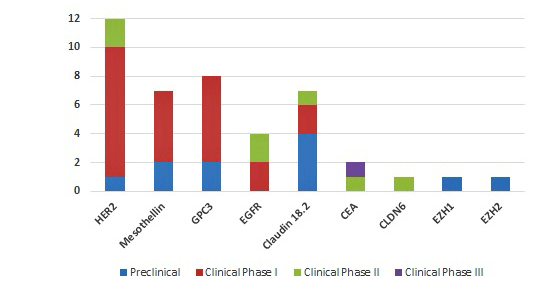
The most popular targets of CAR-T cell therapy for solid tumors
Source: Pharmacodia
1. Claudin 18.2
Claudin 18.2, also known as CLDN18, belongs the Claudin (CLDN) protein family, which is highly specifically expressed in gastric and pancreatic cancers. As a pan-tumor molecular target, Claudin 18.2 was initially found to be highly expressed in a variety of gastric cancer tissues, but subsequent studies suggested that it can also be aberrantly expressed in a variety of primary malignancies, including breast, colon, liver, head and neck, bronchial, and non-small cell lung cancers. Therefore, Claudin 18.2 is considered to be a potent target for immunotherapy of solid tumors. Notably, on September 19, 2021, the latest data on Claudin 18.2 CAR-T (CT041) therapy from Shanghai CARsgen Co. was presented at the EMSO Annual Meeting, demonstrating that CT041 showed a favorable safety profile and promising anti-tumor activity in patients with CLDN18.2-positive gastrointestinal tumor. Because of this, Claudin 18.2 has attracted the interest of researchers and pharmaceutical companies, and many Claudin 18.2 targeted CAR-T therapies have emerged.
Data from Pharmacodia show that there are seven Claudin 18.2 CAR-T clinical drug programs. Three Claudin 18.2 CAR-T solid tumor treatments are in the clinical stage, from Shanghai CARsgen Co., Shanghai Oriental Hospital, and Nanjing KAEDI Biotech Co.
| Drug name |
Clinical stage |
Company |
Indications |
Target |
Last update date |
| CLDN18.2 CAR-T (Carsgen Therapeutics) |
Clinical Phase II |
Shanghai CARsgen Co.;
Carsgen Therapeutics Ltd;
Shanghai CARsgen Co.; |
Esophageal cancer ;
Pancreatic cancer;
Stomach cancer;
Adenocarcinoma;
Solid tumors ; |
Claudin 18.2 |
2021-11-23 |
| LCAR-C18S CAR-T cell therapy |
Clinical Phase I |
Shanghai Oriental Hospital; |
Stomach cancer; |
Claudin 18.2 |
2021-10-19 |
| KD-022 (Nanjing KAEDI Biotech) |
Clinical Phase I |
Nanjing Kaiji Biotechnology Co; |
Tumors ; |
Claudin 18.2 |
2020-11-06 |
| BNT-212 |
preclinical |
Biontech Se; |
Solid tumors ;
Pancreatic cancer ; |
Claudin 18.2 |
2021-03-16 |
| PR401 |
preclinical |
Shandong Boan Biotechnology Co; |
Stomach cancer ;
Pancreatic cancer; |
Claudin 18.2 |
2021-11-01 |
| P2-003 |
preclinical |
Shandong Boan Biotechnology Co; |
Stomach cancer ;
Pancreatic cancer; |
Claudin 18.2 |
2021-11-01 |
| KJ-C1807 |
preclinical |
Koji Biomedical (Shanghai) Co; |
Stomach cancer ;
Pancreatic cancer; |
Claudin 18.2 |
2021-11-01 |
2. MSLN
Mesothelin (MSLN) is a cell surface glycoprotein that is highly expressed in a variety of tumors, such as malignant pleural mesothelioma, pancreatic cancer, ovarian cancer, and lung cancer. But MSLN is low expressed in the normal pleura, peritoneum, and pericardial tissues, making MSLN a potential target for CAR-T cell therapy of solid tumors. On November 12, 2021, French Cellectis Co. announced the first preclinical data of UCARTMESO, an allogeneic CAR-T cell candidate targeting MSLN for the treatment of pancreatic cancer and mesothelioma, at the annual meeting of the Society for Immunotherapy of Cancer (SITC). The study evaluated the in vivo activity of TGFβR2 knockout MESO CAR-T cells using immunodeficient mesothelioma mouse models. The increased tumor regression and survival were observed in mouse treated with CAR-T. In addition, a novel target MSLN CAR-T was designed from a domestic clinical trial for the treatment of relapsed refractory epithelial ovarian cancer. Currently, the clinical trial has officially started recruiting patients. Thus, MSLN CAR-T also plays a key role in the field of solid tumor treatment research.
Data from Pharmacodia show that there are currently seven MSLN CAR-T clinical drug programs. Five are in the clinical stage, involving pharmaceutical companies such as Shanghai CARsgen Co. and Nanjing Legend Biotech Co, as well as Novartis and Cellectis.
| Drug name |
Clinical stage |
Company |
Indications |
Target |
Last update date |
| LCAR-M23 |
Clinical Phase I |
Nanjing Legend Biotech Co; |
Ovarian epithelial carcinoma ; |
Mesothelin |
2021-11-24 |
| UCARTMESO |
preclinical |
Cellectis; |
Solid tumors ; |
Mesothelin |
2021-11-16 |
| KJ-C2113 |
preclinical |
Koji Biomedical (Shanghai) Co; |
Solid tumors ; |
Mesothelin |
2021-11-1 |
| KD-021 |
Clinical Phase I |
Nanjing Kaiji Biotechnology Co; |
Tumors ; |
Mesothelin |
2020-12-18 |
| MCY-M11 |
Clinical Phase I |
Maxcyte; |
Ovarian cancer ;
Peritoneal cancer ;
Mesothelial tissue tumors;
Fallopian tube cancer ;
Adenocarcinoma; |
Mesothelin |
2020-10-27 |
| iCasp9M28z |
Clinical Phase I |
Memorial Sloan Kettering Cancer Center; |
Lung cancer;
Mesothelial tissue tumors;
Breast cancer;
Metastatic cancer ; |
Mesothelin |
2020-8-28 |
| RNA-Meso-CIR-T |
Clinical Phase I |
Novartis;
University of Pennsylvania ; |
Mesothelial tissue tumors; |
Mesothelin |
2020-8-28 |
3. GPC3
GPC3 is a carcinoembryonic antigen involved in cell proliferation, differentiation, migration and apoptosis. It is barely expressed in normal tissues, but is expressed in 70-80% of hepatocellular carcinomas. In addition to the first solid tumor CAR-T therapy targeting Claudin18.2, there is good news about CAR-T cell therapy targeting GPC3 for advanced hepatocellular carcinoma (HCC). A clinical study published in the international oncology journal Clinical Cancer Research, makes waves. Notably, this is the first global clinical trial targeting GPC3 CAR-T cells for the treatment of HCC. This study demonstrated that CAR-GPC3 T-cell therapy in patients with advanced HCC was well tolerated, largely safe and controlled, with no patients experiencing off-target-based toxicologic effects. Overall, CAR-GPC3 T-cell therapy initially showed a good clinical benefit. Therefore, the prospect of CAR-GPC3 T-cell therapy for HCC treatment, should not be underestimated.
Based on data from Pharmacodia, there are currently eight GPC3 CAR-T clinical programs, six in clinical phase I, involving pharmaceutical and research institutes such as Shanghai CARsgen Co., Nanjing Legend Biotech Co., and Baylor College of Medicine.
| Drug name |
Clinical stage |
Company |
Indications |
Target |
Last update date |
| CT-017 |
Clinical Phase I |
Shanghai CARsgen Co.; |
Hepatocellular carcinoma; |
GPC |
2021-11-24 |
| LB-2101 |
preclinical |
Nanjing Legend Biotech Co; |
Non-small cell lung cancer ;
Hepatocellular carcinoma ; |
GPC |
2021-11-24 |
| GPC3-CAR-T cell therapy |
Clinical Phase I |
Shanghai CARsgen Co.; |
Solid tumors ;
Hepatocellular carcinoma ;
Squamous cell carcinoma; |
GPC |
2021-11-01 |
| LQ-102 |
preclinical |
Shanghai Luoqi Biomedical Technology Co; |
Hepatocellular carcinoma; |
GPC |
2021-02-04 |
| Anti-GPC3 chimeric antigen receptor T cell therapy |
Clinical Phase I |
Hunan Zhaotai Medical Group Co; |
Lung cancer ;
Liver cancer; |
GPC |
2020-12-18 |
| Anti-GPC3 CAR T-cell therapy |
Clinical Phase I |
Nanjing University; |
Hepatocellular carcinoma; |
GPC |
2020-12-18 |
| GPC3-T2 CAR-T cell therapy |
Clinical Phase I |
The Second Hospital of Guangzhou Medical University; |
Hepatocellular carcinoma ;
Squamous cell carcinoma ; |
GPC |
2020-12-18 |
| GLYCAR T cell therapy |
Clinical Phase I |
Baylor College of Medicine ;
Methodist Hospital; |
Hepatocellular carcinoma; |
GPC |
2020-08-28 |
4. HER2
Human epidermal growth factor receptor 2 (HER2) is overexpressed in many cancers and has been extensively studied in breast cancer. It has been reported that HER2 is largely expressed on glioblastoma cells, but no HER2 expression is observed in normal brain tissue. A clinical study using HER2 CAR-T cell immunotherapy for children with refractory myeloid metastatic rhabdomyosarcoma was published in the journal Nature Communications on July 15, 2020. Recently, Nicholas A. Vitanza's team from the Ben Towne Children's Cancer Research Center published an important study in journal Nature Medicine, regarding the HER2 CAR-T cell therapy for the treatment of relapsed central nervous system tumors. Thus, HER2 CAR-T is also expected to bring new insights for the solid tumors.
Data from Pharmacodia show that there are currently 12 items for HER2 CAR-T clinical programs, two are in clinical phase II, from Aurora Biopharma.
| Drug name |
Clinical stage |
Company |
Indications |
Target |
Last update date |
| Anti-HER2 CAR T-cell therapy |
Clinical Phase I |
Baylor College of Medicine; |
Sarcoma; |
HER2 |
2021-07-14 |
| TT-16 |
preclinical |
Tessa Therapeutics; |
Head and neck cancer; |
HER2 |
2020-09-29 |
| HER2.taNK |
Clinical Phase I |
Nantkwest; |
Glioblastoma; |
HER2 |
2020-09-27 |
| MB-103 |
Clinical Phase I |
City of Hope National Medical Center ;
Mustang Bio; |
Glioblastoma;
Brain metastases ; |
HER2 |
2020-09-07 |
| AU-105 |
Clinical Phase II |
Aurora Biopharma; |
Glioblastoma; |
HER2 |
2020-08-28 |
| AU-101 |
Clinical Phase II |
Aurora Biopharma; |
Osteosarcoma;
Glioblastoma;
Breast cancer; |
HER2 |
2020-08-28 |
| TGFBeta-resistant-HER2-EBV-CTLs |
Clinical Phase I |
Baylor College of Medicine ;
Methodist Hospital; |
Tumors ; |
HER2 |
2020-08-28 |
| HER.CAR-CMV-specific-CTLs |
Clinical Phase I |
Baylor College of Medicine ;
Methodist Hospital ;
Texas Children's Hospital; |
Glioblastoma; |
HER2 |
2020-08-28 |
| Anti-HER2 CAR T-cell therapy (Seattle Children's Hospital) |
Clinical Phase I |
Seattle Institute for Pediatrics ; |
Tumors of the central nervous system; |
HER2 |
2020-08-28 |
| Autologous-HER2-specific-T-cells |
Clinical Phase I |
Baylor College of Medicine ;
Methodist Hospital ;
Texas Children's Hospital; |
Sarcoma; |
HER2 |
2020-08-28 |
| CIDeCAR |
Clinical Phase I |
Bellicum;
National Cancer Institute; |
Osteosarcoma; |
HER2 |
2020-08-28 |
| HER2(EQ)BBzeta/CD19 T cells (City of Hope) |
Clinical Phase I |
City of Hope National Medical Center ;
Mustang Bio; |
Glioma; |
HER2 |
2020-08-28 |
5. EGFR
The epidermal growth factor receptor (EGFR) is a tyrosine kinase receptor with gene amplification and/or mutations in approximately 50% of adult primary glioblastoma, making it an attractive candidate for targeted therapy. Domestically, CAR-T using EGFR as a target has been reported for the treatment of advanced refractory non-small cell lung cancer patients with strong positive EGFR expression (EGFR expression over 50%). The results of the study showed evaluable efficacy in 11 of these patients: two patients had significant tumor shrinkage and five had stable disease. In 2020, in a study published in journal Clinical & Translational Immunology, which demonstrated for the first time that EGFR is an immunotherapeutic target for triple-negative breast cancer (TNBC). The research team successfully designed novel CAR-T cells targeting EGFR (EGFR CAR-T) and evaluated the efficacy of EGFR CAR-T against TNBC in vitro and in vivo. The experimental results showed that EGFR CAR-T cells had potent and specific inhibition of TNBC cell growth in vitro, and this result was further demonstrated in vivo in a xenograft mouse model that with minimal extratumoral cytotoxicity. Thus, EGFR CAR-T for solid tumors are likely to provide a new direction for the treatment of TNBC.
Data from Pharmacodia show that there are currently four EGFR CAR-T clinical programs. The research involved Shenzhen Second People's Hospital, Guangzhou Bio-Gene Technology Co., and Seattle Children's Hospital.
| Drug name |
Clinical stage |
Company |
Indications |
Target |
Last update date |
| Anti-EGFR CAR-T cell therapy |
Clinical Phase II |
Shenzhen Second People's Hospital; |
Colorectal cancer; |
EGFR |
2021-03-02 |
| Anti-EGFR CAR-T cell therapy |
Clinical Phase II |
Cellular Biomedicine Group (Shanghai) Co;
General Hospital of the Chinese People's Liberation Army ; |
Tumors ; |
EGFR |
2020-12-18 |
| Autologous EGFR-CAR T cells |
Clinical Phase I |
Guangzhou Bio-Gene Technology Co;
Zhongshan University; |
Non-small cell lung cancer ; |
EGFR |
2020-12-18 |
| Anti-EGFR CAR T-cell therapy |
Clinical Phase I |
Seattle Children's Hospital; |
Tumors of the central nervous system; |
EGFR |
2020-08-28 |
To assist pharmaceutical companies in the research of CAR-T therapies in solid tumors, CUSABIO developed a series of active protein products with high purity, superb specificity, excellent bioactivity, including Claudin 18.2, GPC3, MSLN, HER2, EGFR, etc. You are welcome to contact us.
● Recombinant Human CLDN18.2-VLPs (Active) (Code: CSB-MP005498HU(A5))
Specificity Validated by Western blot (WB)
-WB.jpg)
CSB-MP005498HU(A5) is detected by Mouse anti-6*His monoclonal antibody. The three bands respectively correspond to monomer, Homodimer, Homotrimer
Activity Validated by Functional ELISA
-AC1.jpg)
Immobilized human CLDN18.2 (CSB-MP005498HU(A5)) at 5 μg/ml can bind anti-CLDN18.2 recombinant Monoclonal Antibody (CSB-RA005498A1HU),the EC50 is 5.225-9.256 ng/ml.
Specificity Validated by Western blot (WB)
-WB2.jpg)
CSB-MP005498HU(A5) is detected by Anti-Claudin 18 Antibody. The two bands respectively correspond to monomer, Homodimer
Correct Structure Validated by TEM
-TEM1.jpg)
The VLP-like structures of CLDN18.2 has been confirmed by TEM.
● Recombinant Human Mesothelin(MSLN),partial (Active) (Code: CSB-MP015044HUc9)
Purity Validated by SDS-PAGE
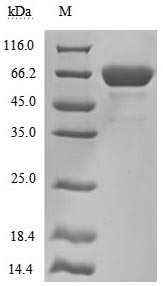
Purity is greater than 92% as determined by SDS-PAGE. (Tris-Glycine gel) Discontinuous SDS-PAGE (reduced) with 5% enrichment gel and 15%
Activity Validated by Functional ELISA
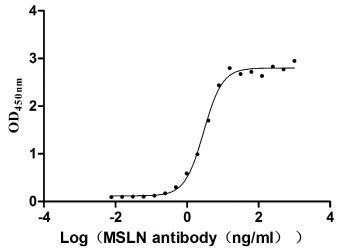
Immobilized MSLN at 2 μg/ml can bind Anti-MSLN rabbit monoclonal antibody, the EC50 of the MSLN protein is 2.657-3.177 ng/ml.
● Recombinant Human Epidermal growth factor receptor(EGFR),partial (Active) (Code: CSB-MP007479HU)
Purity Validated by SDS-PAGE
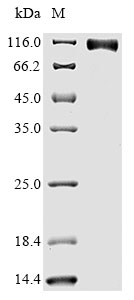
Purity is greater than 90% as determined by SDS-PAGE. (Tris-Glycine gel) Discontinuous SDS-PAGE (reduced) with 5% enrichment gel and 15%
Activity Validated by Functional ELISA
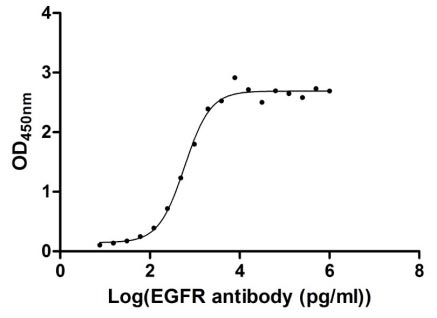
Immobilized EGFR at 1 μg/ml can bind Anti-EGFR recombinant antibody, the EC50 of human EGFR protein is 2.867-3.571 ng/ml.
Affinity Validated by LSPR Assay
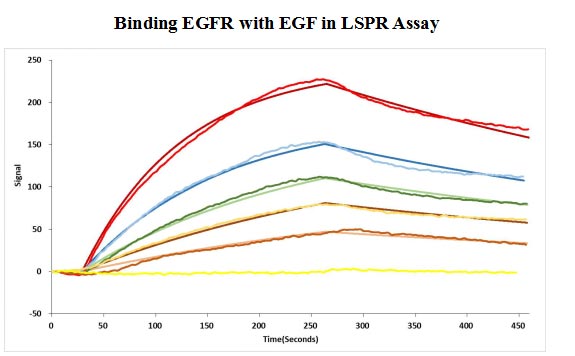
Human EGF protein captured on COOH chip can bind Human EGFR protein, his and Myc tag (CSB-MP007479HU) with an affinity constant of 11.9nM as detected by LSPR Assay.
Activity Validated by Functional ELISA
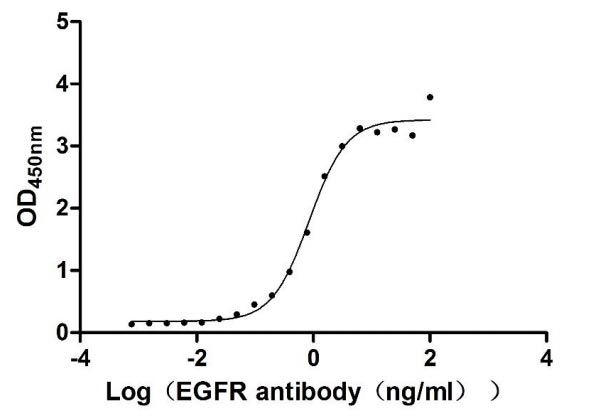
Immobilized EGFR at 1 μg/ml can bind Cetuximab, the EC50 of human EGFR protein is 0.6919-1.047 ng/ml.
References
[1] Upadhaya, Samik, et al. "The clinical pipeline for cancer cell therapies." Nature reviews. drug Discovery (2021).
[2] Berger, Trisha R., and Marcela V. Maus. "Mechanisms of response and resistance to CAR T cell therapies. "Current Opinion in Immunology 69 (2021): 56-64.
[3] Mikkilineni, Lekha, and James N. Kochenderfer. "CAR T cell therapies for patients with multiple myeloma. "Nature Reviews Clinical Oncology 18.2 (2021) : 71-84.
[4] Kyuno, Daisuke, et al. "Claudin-18.2 as a therapeutic target in cancers: cumulative findings from basic research and clinical trials." Tissue Barriers (2021): 1967080.
[5] McGuire, Christina, et al. "Increasing immune cell infiltration in hepatocellular carcinoma tumors using a novel GPC3-targeting aptamer conjugate." (2021): 1707-1707.
[6] Klampatsa, Astero, Vivian Dimou, and Steven M. Albelda. "Mesothelin-targeted CAR-T cell therapy for solid tumors. "Expert opinion on biological therapy 21.4 (2021): 473-486.
[7] Vitanza, Nicholas A., et al. "Locoregional infusion of HER2-specific CAR T cells in children and young adults with recurrent or refractory CNS tumors: an interim analysis." nature medicine 27.9 (2021): 1544-1552.
[8] Zhang, Yajun, et al. "Phase I clinical trial of EGFR-specific CAR-T cells generated by the piggyBac transposon system in advanced relapsed/refractory non-small cell lung cancer patients." journal of Cancer Research and Clinical Oncology (2021): 1-10.
CUSABIO team. 2021 Insights on the CAR-T Cell Therapy: the Most Popular Targets for Solid Tumor!. https://www.cusabio.com/c-21053.html

-WB.jpg)
-AC1.jpg)
-WB2.jpg)
-TEM1.jpg)
-SDS.jpg)
-AC1.jpg)









Comments
Leave a Comment