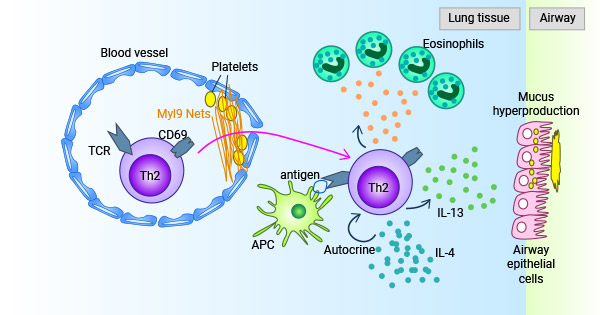
[1] Iwamura, Chiaki, et al. "Elevated Myl9 reflects the Myl9-containing microthrombi in SARS-CoV-2-induced lung exudative vasculitis and predicts COVID-19 severity." Proceedings of the National Academy of Sciences 119.33 (2022): e2203437119.
[2] Kimura, Motoko Y., et al. "A new therapeutic target: the CD69-Myl9 system in immune responses." Seminars in immunopathology. vol. 41. Springer Berlin Heidelberg, 2019.
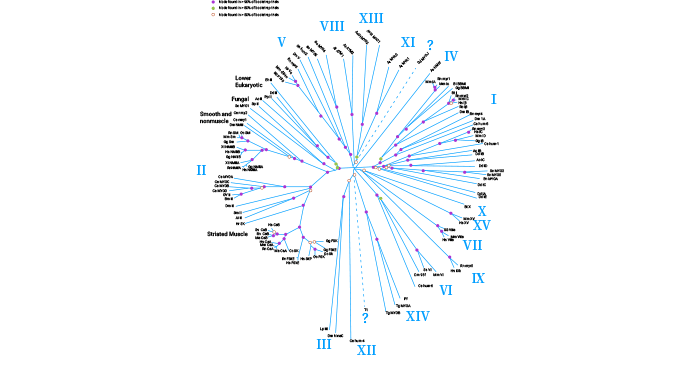
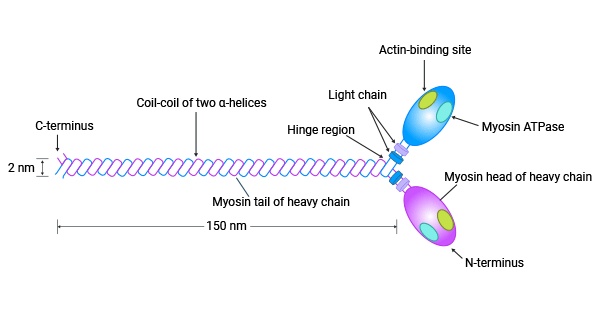
[3] Sellers, James R. "Myosins: a diverse superfamily." Biochimica et Biophysica Acta (BBA)-Molecular Cell Research 1496.1 (2000): 3-22.
[4] Fitz, Gillian N., et al. "Protrusion growth driven by myosin-generated force." Developmental Cell 58.1 (2023): 18-33.
[5] Hartman, M. Amanda, and James A. Spudich. "The myosin superfamily at a glance. "Journal of cell science 125.7 (2012): 1627-1632.
[6] Lewis, Christopher TA, and Julien Ochala. "Myosin heavy chain as a novel key modulator of striated muscle resting state. "Physiology 38.1 (2023): 3-9.
[7] Bhanu P. Jena. "Myosin: Cellular Molecular Motor" Cellular Nanomachines (2020): 79-89.
[8] Kruthika, Banavathy S., et al. "Expression pattern and prognostic significance of myosin light chain 9 (MYL9): a novel biomarker in glioblastoma." Journal of Clinical Pathology 72.10 (2019): 677-681.
[9] Shehadeh, Lina A., et al. "Dynamic regulation of vascular myosin light chain (MYL9) with injury and aging." PLoS One 6.10 (2011): e25855.
[10] Zhu, Kongxi, et al. "Long non-coding RNA MBNL1-AS1 regulates proliferation, migration, and invasion of cancer stem cells in colon cancer by interacting with MYL9 via sponging microRNA-412-3p." Clinics and research in hepatology and gastroenterology 44.1 (2020): 101-114.
[11] Jalagadugula, Gauthami, et al. "Regulation of platelet myosin light chain (MYL9) by RUNX1: implications for thrombocytopenia and platelet dysfunction in RUNX1 haplodeficiency." Blood, The Journal of the American Society of Hematology 116.26 (2010): 6037-6045.
[12] Moreno, Carolina Araujo, et al. "Homozygous deletion in MYL9 expands the molecular basis of megacystis-microcolon- intestinal hypoperistalsis syndrome." European Journal of Human Genetics 26.5 (2018): 669-675.
[13] Licht, Alexander H., et al. "Junb regulates arterial contraction capacity, cellular contractility, and motility via its target Myl9 in mice." The Journal of clinical investigation 120.7 (2010): 2307-2318.
[14] Matsushita, Katsunori, et al. "Clinicopathological significance of MYL9 expression in pancreatic ductal adenocarcinoma." Cancer Reports 5.10 (2022 ): e1582.
[15] Lv, Minghe, Lumeng Luo, and Xue Chen. "The landscape of prognostic and immunological role of myosin light chain 9 (MYL9) in human tumors." Immunity. Inflammation and Disease 10.2 (2022): 241-254.
[16] Gilles, Laure, et al. "MAL/SRF complex is involved in platelet formation and megakaryocyte migration by regulating MYL9 (MLC2) and MMP9." Blood, The Journal of the American Society of Hematology 114.19 (2009): 4221-4232.
[17] Tan, Xiang, and Mingwu Chen. "MYLK and MYL9 expression in non-small cell lung cancer identified by bioinformatics analysis of public expression data." Tumor Biology 35 (2014): 12189-12200.
[18] Luo, Xue-Gang, et al. "Histone methyltransferase SMYD3 promotes MRTF-A-mediated transactivation of MYL9 and migration of MCF-7 breast cancer cells." Cancer letters 344.1 (2014): 129-137.
[19] Hirasawa, Yuichi, et al. "Methylation status of genes upregulated by demethylating agent 5-aza-2′-deoxycytidine in hepatocellular carcinoma." Oncology 71.1-2 (2007): 77-85.
[20] Deng, Yuao, et al. "High expression of MYL9 indicates poor clinical prognosis of epithelial ovarian cancer." Recent Patents on Anti-Cancer Drug Discovery 16.4 (2021): 533-539.
[21] Wang, Jian-Hua, et al. "Expression and prognostic significance of MYL9 in esophageal squamous cell carcinoma." ploS one 12.4 (2017): e0175280.
[22] Zhao, S., W. Xiong, and K. Xu. "MiR-663a, regulated by lncRNA GAS5, contributes to osteosarcoma development through targeting MYL9. "Human & Experimental Toxicology 39.12 (2020): 1607-1618.
[23] Huang, Ya-qiang, et al. "Decreased expression of myosin light chain MYL9 in stroma predicts malignant progression and poor biochemical recurrence- free survival in prostate cancer." medical oncology 31 (2014): 1-9.
[24] Huang, Chu-Han, et al. "MYL9 deficiency is neonatal lethal in mice due to abnormalities in the lung and the muscularis propria of the bladder and intestine ." Plos one 17.7 (2022): e0270820.
[25] Dong, Ningxin, et al. "Identification and validation of critical genes with prognostic value in gastric cancer." Frontiers in Cell and Developmental Biology 10 (2022): 1072062.
[26] Qiu, Xiao, et al. "Weighted gene co-expression network analysis identified MYL9 and CNN1 are associated with recurrence in colorectal cancer." journal of Cancer 11.8 (2020): 2348.
[27] Zhou, Rui, et al. "PRPF19 facilitates colorectal cancer liver metastasis through activation of the Src-YAP1 pathway via K63-linked ubiquitination of MYL9." Cell Death & Disease 14.4 (2023): 258.
[28] Feng, Min, et al. "Myosin light chain 9 promotes the proliferation, invasion, migration and angiogenesis of colorectal cancer cells by binding to Yes- associated protein 1 and regulating Hippo signaling." Bioengineered 13.1 (2022): 96-106.
[29] Sheng, Meiling, Zhaohui Dong, and Yanping Xie. "Identification of tumor-educated platelet biomarkers of non-small-cell lung cancer. "OncoTargets and therapy (2018): 8143-8151.
[30] Tan, Xiang, and Mingwu Chen. "MYLK and MYL9 expression in non-small cell lung cancer identified by bioinformatics analysis of public expression data." Tumor Biology 35 (2014): 12189-12200.
[31] Zhang, Chunling, et al. "Myocardin-related transcription factor A is up-regulated by 17β-estradiol and promotes migration of MCF-7 breast cancer cells via transactivation of MYL9 and CYR61." Acta Biochim Biophys Sin 45.11 (2013): 921-927.
[32] Kandler, Justin L., et al. "Compound heterozygous loss of function variants in MYL9 in a child with megacystis-microcolon intestinal hypoperistalsis syndrome." Molecular Genetics & Genomic Medicine 8.11 (2020): e1516.
[33] Kobayashi, Hironobu, et al. "Increased Myosin light chain 9 expression during Kawasaki disease vasculitis." Frontiers in Immunology 13 (2023). 1036672.
[34] Xiong, Yao, et al. "Targeting MRTF/SRF in CAP2-dependent dilated cardiomyopathy delays disease onset." jci insight 4.6 (2019).
[35] Sun, L., et al. "Decreased platelet expression of myosin regulatory light chain polypeptide (MYL9) and other genes with platelet dysfunction and CBFA2/ RUNX1 mutation: insights from platelet expression profiling." Journal of Thrombosis and Haemostasis 5.1 (2007): 146-154.
[36] Wen, Dingke, et al. "Single-cell RNA sequencing reveals the pathogenic relevance of intracranial atherosclerosis in blood blister-like aneurysms." Frontiers in Immunology 13 (2022): 927125.
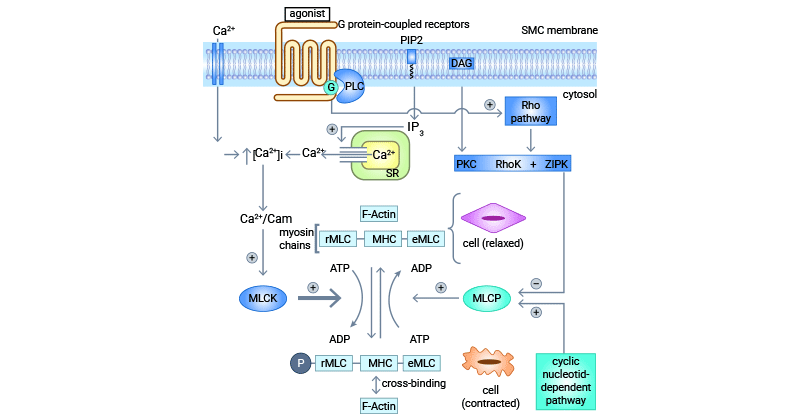
[37] Hwang, Ki-Chul. "The Role of MicroRNAs in Vascular Diseases; Smooth Muscle Cell Differentiation and De-Differentiation. "Korean Circulation Journal 44.4 (2014): 218-219.
[38] Hayashizaki, Koji, et al. "Myosin light chains 9 and 12 are functional ligands for CD69 that regulate airway inflammation." science immunology 1.3 (2016 ): eaaf9154-eaaf9154.
[39] Nakayama, Toshinori, et al. "CD4+ T cells in inflammatory diseases: pathogenic T-helper cells and the CD69-Myl9 system." International Immunology 33.12 (2021): 699-704.
[40] Yokoyama, Masaya, et al. "Myosin light chain 9/12 regulates the pathogenesis of inflammatory bowel disease." Frontiers in Immunology 11 (2021): 594297.
[41] Onodera, Atsushi, Kota Kokubo, and Toshinori Nakayama. "Epigenetic and transcriptional regulation in the induction, maintenance, heterogeneity and recall-response of effector and memory Th2 cells." Frontiers in Immunology 9 (2018): 2929.



Comments
Leave a Comment