[1] Hegazy, Marihan, et al. "Desmosomal cadherins in health and disease." Annual Review of Pathology: Mechanisms of Disease 17 (2022): 47-72.
[2] Kosaka, K., et al. "Pemphigus vegetans with antibodies against desmoglein 1 and desmocollin 1-3: a case report and literature review." Journal of the European Academy of Dermatology and Venereology 36.12 (2022): e998-e1000.
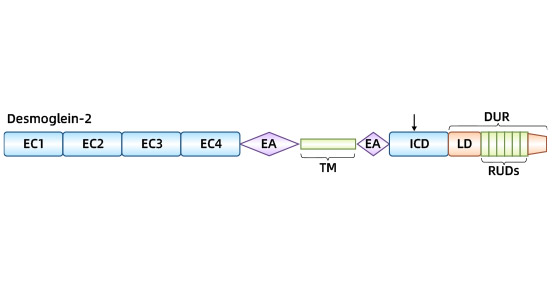
[3] Sokol, Ena. "Structure of desmosomes." Autoimmune Bullous Diseases: Text and Review. cham: Springer International Publishing, 2022. 61-64.
[4] Myo Min, Kay K., et al. "Desmoglein-2 is important for islet function and β-cell survival." Cell Death & Disease 13.10 (2022): 911.
[5] Zhao, W. L., et al. "Analysis of clinical characteristics, prognosis and antibody pathogenicity of pemphigus patients positive for anti desmoglein IgG autoantibodies in remission: a retrospective cohort study." Journal of the European Academy of Dermatology and Venereology 36.2 (2022) : 271-278. : 271-278.
[6] Yanqing, L. I., et al. "Research progress in the function of desmoglein-2 in digestive system tumors." JOURNAL OF SHANGHAI JIAOTONG UNIVERSITY (MEDICAL SCIENCE) 42.2 (2022): 247.
[7] Shiba, Mikio, et al. "Phenotypic recapitulation and correction of desmoglein-2-deficient cardiomyopathy using human-induced pluripotent stem cell -derived cardiomyocytes." Human Molecular Genetics 30.15 (2021): 1384-1397.
[8] Hawthorne, Robert N., et al. "Altered electrical, biomolecular, and immunologic phenotypes in a novel patient-derived stem cell model of desmoglein-2 mutant ARVC." Journal of Clinical Medicine 10.14 (2021): 3061.
[9] Debus, Jana Davina, et al. "In vitro analysis of arrhythmogenic cardiomyopathy associated desmoglein-2 (DSG2) mutations reveals diverse glycosylation patterns." journal of molecular and cellular cardiology 129 (2019): 303-313.
[10] Yang, Tingting, et al. "DSG2 expression is low in colon cancer and correlates with poor survival." bmc gastroenterology 21.1 (2021): 1-10.
[11] Qin, Shuhang, et al. "DSG2 expression is correlated with poor prognosis and promotes early-stage cervical cancer." Cancer Cell International 20 (2020) : 1-13.
[12] Lee, Kyungmin, et al. "Dsg2-mediated c-Met activation in anaplastic thyroid cancer motility and invasion." Endocrine-Related Cancer 27.11 (2020). 601-614.
[13] Yang, Tingting, et al. "DSG2 expression is low in colon cancer and correlates with poor survival." bmc gastroenterology 21.1 (2021): 1-10.
[14] Zhou, Bing-Xia, and Yan Li. "Significance of desmoglein-2 on cell malignant behaviors via mediating MAPK signaling in cervical cancer." The Kaohsiung journal of medical sciences 36.5 (2020): 336-343.
[15] Chen, Liang, et al. "A founder homozygous DSG2 variant in East Asia results in ARVC with full penetrance and heart failure phenotype." International Journal of Cardiology 274 (2019): 263-270.
[16] Kuo, Kelly, Rosa Speranza, and Rinat Hackmon. "Fetal dilated cardiomyopathy associated with variants of uncertain significance in MYH7 and DSG2 genes. A case report and review of the literature." Journal of Obstetrics and Gynaecology Canada 42.9 (2020): 1147-1150.
[17] Overmiller, Andrew M., et al. "c-Src/Cav1-dependent activation of the EGFR by Dsg2." Oncotarget 7.25 (2016): 37536.
[18] Chen, Ling, et al. "Up-regulation of Dsg2 conferred stem cells with malignancy through wnt/β-catenin signaling pathway. "Experimental Cell Research 422.1 (2023): 113416.
[19] Eshkind, Leonid, et al. "Loss of desmoglein 2 suggests essential functions for early embryonic development and proliferation of embryonal stem cells." European journal of cell biology 81.11 (2002): 592-598.
[20] Kamekura, R., et al. "Loss of the desmosomal cadherin desmoglein-2 suppresses colon cancer cell proliferation through EGFR signaling." oncogene 33.36 (2014): 4531-4536.
[21] Zhou, Bing-Xia, and Yan Li. "Significance of desmoglein-2 on cell malignant behaviors via mediating MAPK signaling in cervical cancer." The Kaohsiung journal of medical sciences 36.5 (2020): 336-343.
[22] Lin, Yen-Nien, et al. "Extracellular vesicles from immortalized cardiosphere-derived cells attenuate arrhythmogenic cardiomyopathy in desmoglein -2 mutant mice." European Heart Journal 42.35 (2021): 3558-3571.
[23] Brennan, Donna, and Mỹ G. Mahoney. "Increased expression of Dsg2 in malignant skin carcinomas: A tissue-microarray based study." Cell adhesion & migration 3.2 (2009): 148-154.
[24] Barber, Alison G., et al. "PI 3K/AKT pathway regulates E-cadherin and Desmoglein 2 in aggressive prostate cancer." cancer medicine 4.8 ( 2015): 1258-1271.
[25] Kim, Jiho, et al. "Desmoglein-2 as a prognostic and biomarker in ovarian cancer." Cancer Biology & Therapy 21.12 (2020): 1154-1162.
[26] Gonzalez-Pastor, Rebeca, Peter S. Goedegebuure, and David T. Curiel. "Understanding and addressing barriers to successful adenovirus-based virotherapy for ovarian cancer." Cancer gene therapy 28.5 (2021): 375-389.
[27] Qin, Shuhang, et al. "DSG2 expression is correlated with poor prognosis and promotes early-stage cervical cancer." Cancer Cell International 20 (2020) : 1-13.
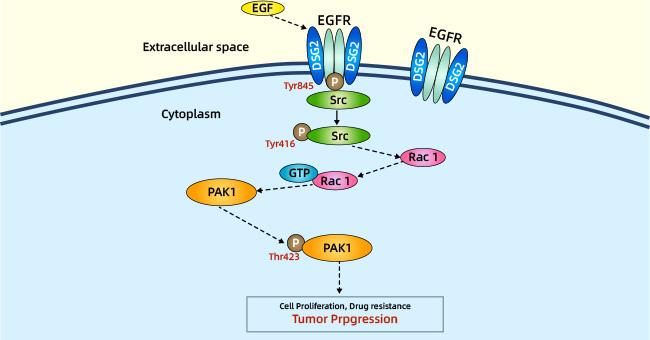
[28] Jin, Runsen, et al. "Desmoglein-2 modulates tumor progression and osimertinib drug resistance through the EGFR/Src/PAK1 pathway in lung adenocarcinoma." Cancer Letters 483 (2020): 46-58.
[29] Chen, Liang, et al. "A founder homozygous DSG2 variant in East Asia results in ARVC with full penetrance and heart failure phenotype." International Journal of Cardiology 274 (2019): 263-270.
[30] Schlegel, Nicolas, Kevin Boerner, and Jens Waschke. "Targeting desmosomal adhesion and signalling for intestinal barrier stabilization in inflammatory bowel diseases-Lessons from experimental models and patients." Acta physiologica 231.1 (2021): e13492.
[31] Wang, Hongjie, et al. "Structural and functional studies on the interaction of adenovirus fiber knobs and desmoglein 2." journal of virology 87.21 (2013 ): 11346-11362.
[32] Wang, Hongjie, et al. "Intracellular signaling and desmoglein 2 shedding triggered by human adenoviruses Ad3, Ad14, and Ad14P1." journal of virology 89.21 (2015): 10841-10859.
[33] Zhang, Jing, et al. "Desmoglein 2 (DSG2) is a receptor of human adenovirus type 55 causing adult severe community-acquired pneumonia." Virologica Sinica 36 (2021): 1400-1410.
[34] Ramani, Vishnu C., Leah Hennings, and Randy S. Haun. "Desmoglein 2 is a substrate of kallikrein 7 in pancreatic cancer." BMC cancer 8.1 (2008): 1-10.
[35] Klessner, Jodi L., et al. "EGFR and ADAMs cooperate to regulate shedding and endocytic trafficking of the desmosomal cadherin desmoglein 2." Molecular biology of the cell 20.1 (2009): 328-337.
[36] Liu, Yin-Qiao, et al. "Serum DSG2 as a potential biomarker for diagnosis of esophageal squamous cell carcinoma and esophagogastric junction adenocarcinoma." Bioscience Reports 42.5 (2022): BSR20212612.
[37] Tan, Lih Yin, et al. "Desmoglein 2 promotes vasculogenic mimicry in melanoma and is associated with poor clinical outcome." Oncotarget 7.29 (2016). 46492.
[38] Overmiller, Andrew M., et al. "c-Src/Cav1-dependent activation of the EGFR by Dsg2." Oncotarget 7.25 (2016): 37536.
[39] Overmiller, Andrew M., et al. "Desmoglein 2 modulates extracellular vesicle release from squamous cell carcinoma keratinocytes." The FASEB Journal 31.8 (2017): 3412.




Comments
Leave a Comment