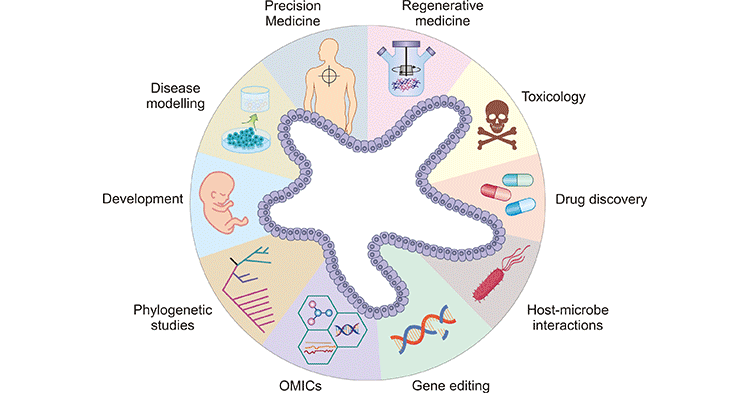
Multiple Applications of Organoids
Compared with the traditional 2D cell culture model, 3D cultured organoids contain a variety of cell types, which can form functional "micro-organs" and can be better used to simulate the occurrence process and physiological and pathological states of organs and tissues. Organoids compensate for certain limitations in existing model systems by providing a stable system easily scalable for culture and manipulation while being more in vivo physically representive of the body.
So far, organoid cultures have been used in a variety of tissues, including the gut, heart, liver, pancreas, kidney, prostate, lung, retina, and brain. Organoids can better simulate the in vivo environment, and undoubtedly provide a better solution for tumor research, disease modeling, drug screening, regenerative medicine, precision medicine, and other fields between animal and cell levels.
Figure 1. Diverse applications of organoid technology
This picture is cited from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7468890/
Organoids provide a platform for modeling pathologies for both embryonic development and adult tissue. Organoids are similar in function and structure to in vivo organs and can also be used to model pathogenic processes, including models of infectious, genetic, and degenerative diseases. For example, if gastric organoids are infected with Helicobacter pylori, the infection mechanism of Helicobacter pylori can be explored. Cells containing the pathologies can be generated using gene-editing techniques or extracted from patients. Pathological conditions in kidney organoids have been generated using a CRISPR/Cas9 knockout system. Cortical organoids, generated from human iPSCs, have been developed to model Miller-Dieker Syndrome.
In disease modeling, organoids generated from human cells can overcome several limitations of animal models. Next to the ethical considerations, some animal models cannot fully recapitulate the human condition, such as with lung tissue.
Organoids can often be obtained from normal human epithelial cells and can be used to model mutations in all stages of malignancies in vitro, which can be used to reveal the complexity of the cancer genome and the important role of genetic mutations in tumorigenesis and development. Discovery of drug targets for precision cancer therapy. A significantly useful development of organoid culturing for cancer research is co-culturing organoids with immune cells. Such methods are important steps toward approaching the physiological tumor microenvironment (TME) and provide a platform for immune-based therapies.
The tumor is a complex disease, no matter the pathogenesis, metastasis mechanism, or the patient's response to treatment and drug resistance mechanism, there are many problems that need to be explored urgently. Future research needs to continuously improve the simulation of TME to create tumor organoid models that are closer to the original tumor properties, so as to gain a more comprehensive understanding of malignant tumors.
1. Drug Discovery and Screening
In general, the creation of a new drug requires a series of processes such as selecting drug targets, identifying lead compounds, selecting candidate drugs, conducting safety and efficacy evaluations in animals, and conducting clinical trials in humans. The current drug research and development is mainly based on molecules, cells, and animals. It has to go through the process of animal to human, and cannot be tested directly on humans, because it will involve drug toxicity and effectiveness. However, animal models are still far from human beings. Therefore, there is a great need for a disease model that can be approached by the human body, so as to reduce the cost and development time of new drug development. The application of organoids can greatly improve the efficiency of drug research and development.
Organoids are attractive for drug discovery since they can be produced on a relatively large scale for an array of subtypes capturing disease heterogeneity and stored long-term, creating biobanks. Organoid biobanks provide opportunities to test drug safety and efficacy. Toxicology studies can be used to assess safety. For efficacy testing, several high-throughput methods have been developed that can generate results for drug response within a week.
Another major advantage of organoid technology in drug development is that normal organoids can be generated and utilized to screen for drugs that target only tumor cells without harming healthy cells. In clinical trials, intolerable adverse reactions are the main cause of drug failure, including liver toxicity, cardiotoxicity, and renal toxicity. Liver organoids can be used to test the hepatotoxicity of drugs. Drug-related hepatotoxicity is mainly mediated by cytochrome P450 enzymes, and the effects of cytochrome P450 enzymes can be observed in liver organoids at near-physiological levels. Cardiotoxic responses such as arrhythmias and reduced cardiac function can also be tested in organoids. In addition, kidney organoids have also been used in drug toxicology studies. Applying organoid technology to preclinical drug toxicology research can help reduce the failure rate of drug clinical trials and reduce the occurrence of adverse events in drug clinical trials. The organoid drug testing model can not completely replace the animal model, but it can play an important complementary function.
So far, organoids have been used to screen drugs for diseases such as colon cancer and cystic fibrosis.
2. Regenerative Medicine and Organ Repair
Organoids have also been utilized in transplantation studies to evaluate regenerative capacity in addition to in vitro applications. First, organoid functionality is established if transplanting into illness mouse models results in a higher survival rate. Second, successful transplantations may pave the way for the use of organoids as tissue-engineered grafts in regenerative medicine applications. Autologous organ transplantation is less vulnerable to magnetic fields or hacking than implantable medical devices and lowers the chance of rejection compared to allogeneic organ transplantation.
The organoid strategy can also enable gene correction in the event of contemporary inherited genetic abnormalities by replacing damaged organs with restored tissue.
3. Personalized Medicine
Predicting patient response to therapy is another potential application of organoid technology. Organoids provide a unique insight into the individual patient's disease and show potential for personalized therapies. By performing some therapeutic manipulations on patient-derived organoids in vitro, the response of such organoids to treatment can be used to predict the response of the patient providing the biopsy to the treatment, so as to select the most appropriate treatment plan for the patient. Personalized medicine aims to develop effective treatment strategies for each patient by better characterizing the disease at the molecular and pharmacogenomic levels. Organoids hold great potential for personalized treatment regimens for patients. For example, measuring the function of cystic fibrosis transmembrane conductance modulators in rectal organoids from cystic fibrosis patients can determine who would benefit from cystic fibrosis transmembrane conductance modulator corrective therapy. Genetic analysis of 1,977 cancer-related genes in organoids from 14 patients with metastatic colorectal cancer showed that 90% of somatic mutations were identical in organoids and biopsy specimens from the same patients, and the organoids The correlation coefficient with the corresponding DNA copy number profile of the original tumor was 0.89. This experiment shows that organoids can well capture the genetic properties of the original tumor, providing strong evidence for the application of organoids in personalized medicine.
4. Immune Therapy
In vitro co-culture of organoids and immune cells can expand immune cells, enhance immune response, and provide a strong guarantee for subsequent immunotherapy. In addition, the study of the tumor microenvironment is also inseparable from the co-culture technology of organoids with immune cells and other cells.
In summary, organoids derived from different human tissues can be used for drug research, which is conducive to drug screening and new drug development. They can also be gene-edited to study the mechanism of tumor occurrence and development and be used in vitro to predict the patient's response to clinical treatment thus aiding to develop a personalized medical plan for the patient.
While it is clear that organoids have many potential uses, it is important to keep in mind their current limitations. In particular, all organoid systems established to date still need to be improved in terms of reproducibility of in vivo development. For example, while retinal organoids showed typical laminar organization well, outer segments failed to form. For example, photoreceptors cannot fully mature to become sensitive to light. Likewise, cerebral organoids recapitulate fairly early events in brain development, but later features such as cortical lamellae are not fully formed. And more culture methods are needed to develop to improve the generation efficiency, shorten the culture period of the organoid model, and reduce the culture cost.
The problem of maturation appears to be a common obstacle in organoid technology, and it remains to be seen whether it will significantly affect its therapeutic and research potential. Human intestinal organoids have been shown to have characteristics of the mature intestine and produce Lgr5+ adult stem cells. Other organoids may be induced to fully mature once heterotopic transplantation may be available for therapeutic purposes. These studies may become a major focus of future organoid research.