What Are Pluripotent Stem Cells?
Pluripotent stem cells are a class of pluripotent cells with the ability to self-renew indefinitely in vitro and differentiate into various cell types in the body. They are important tools for studying gene function, building disease models and advancing the field of regenerative medicine.
What Are Signaling Pathways Regulating Pluripotency of Stem Cells?
The pluripotency of stem cells is regulated and maintained by transcriptional networks that include a series of pluripotent transcription factors. These pluripotent genes stimulate or inhibit the expression of downstream genes, inducing the occurrence of some signaling pathways and regulating the pluripotency of stem cells.
The Function of Signaling Pathways Regulating Pluripotency of Stem Cells
Stem cells retain the potential for self-renewal and multidirectional differentiation in the action of signaling pathways regulating the pluripotency of stem cells. Having a better understanding of the mechanism of these pathways could help scientists study gene function & disease pathogenesis and promote the development of regenerative medicine.
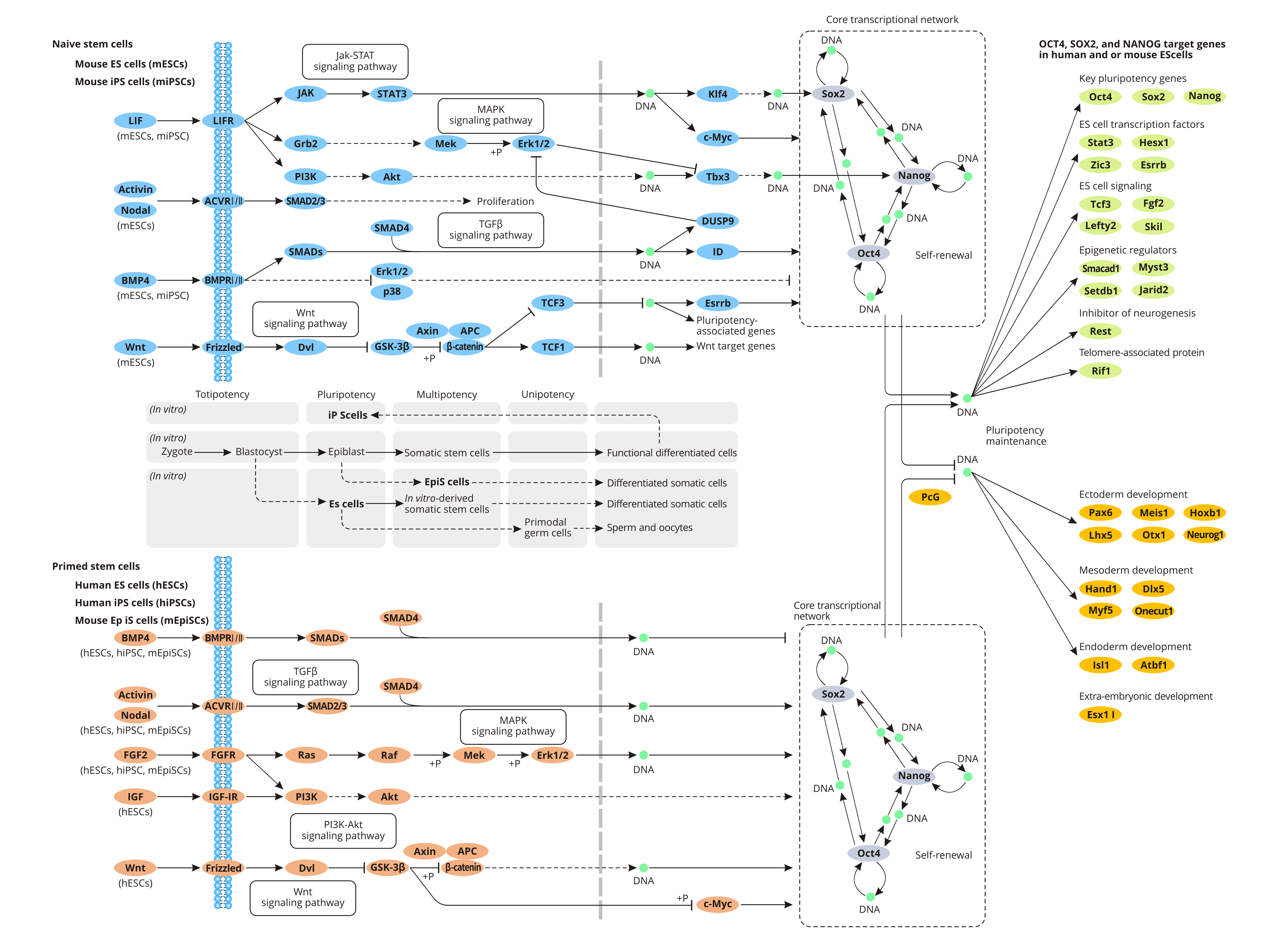
Signaling Pathways Regulating Pluripotency of Stem Cells
Human pluripotent stem cells (hPSCs) are originally established on the feeder layer, murine embryo fibroblast (MEF) irradiated or treated with mitomycin. The feeder layer not only provides nutrition for cell growth but also secrets cytokines (Activin A, IGF-2) that synergize with bFGF (basic fibroblast growth factor) in the culture solution to maintain the pluripotency of human stem cells.
Low concentrations of bFGF can maintain the pluripotency of hPSC in the presence of feeder cells. Because the insulin-like growth factor (IGF) induced by bFGF activates the PI3K-AKT signaling pathway, which inhibits the expression of GSK3, inhibiting the degradation of β-Catenin and maintaining the pluripotency of hPSCs.
Besides, Activin/Nodal/TGF-β respectively binds to their receptors on the cell membrane in the presence of bFGF, activating intracellular cytoplasmic SMAD2/SMAD3. Active SMAD2/3 and SMAD4 form heterodimers into the nucleus, regulating the expression of related genes that maintain the pluripotency of hPSCs. SMAD2/3 can be directly conjugated to the proximal promoter region of Nanog to upregulate the expression of this gene. When the expression of Nanog is increased, the expression of SMAD2/3 is inhibited by negative feedback regulation, and Activin/Nodal/TGF-β signaling pathway in hPSC is controlled to ensure that hPSCs cannot be induced to differentiate into mesendoderm.
BMP4 (bone morphognic protein 4) binds to BMPR, phosphorylating cellular SMAD1, SMAD5, and SMAD8 rather than SMAD2/3. Phosphorylated SMAD1/5/8 further binds to SMAD4 and enters into the nucleus to modulate transcription. When BMP4 in the culture solution is not enough to maintain hPSC self-renewing, the BMP4-SMAD1/5/8 signaling upregulates with the differentiation of hPSCs.



