Kidney Organoids
Most renal organoids are 3D structures formed by pluripotent stem cells (PSCs), which simulate the development process of the human kidney in vitro and undergo induced differentiation. Although it is not a real kidney, renal organoids can form various structures similar to human nephrons, such as glomerular pedal cells, proximal tubules, distal tubules, collecting tubules, etc. They can be used for cell repair and treatment of kidney diseases, as well as for simulating kidney development and disease occurrence and screening drugs to improve renal function.
With approximately 10% of adults worldwide suffering from chronic kidney disease, the increasing incidence of kidney disease is a huge challenge for both clinical and public health efforts. The kidney organoid model not only takes into account the advantages of the animal models and 2D-cell culture models in the establishment of the renal disease models, but also can screen therapeutic drugs and detect drug nephrotoxicity. In the future, renal organoids are also expected to be the source of donor's kidneys for kidney transplantation.
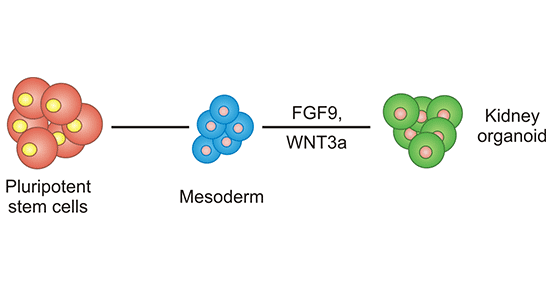
Figure 1. Establishment process of kidney organoids
1. Recent Development of Kidney Organoids
In 2015, scientists successfully used stem cells to grow a kidney organoid in the lab, which contains the various cells and microstructures of the kidney [1]. This kidney organoid contains only two different types of mature cells, which are not yet enough to replace human organs. But the breakthrough could have other applications, such as replacing laboratory animals in drug toxicology tests. During the same period, Ryuji Morizane et al. developed a method for generating kidney organoids from human pluripotent stem cells in vitro [2]. These organoids contain a large number of well-organized nephrons but lack a network of blood vessels to infuse blood. This defect is overcome by making organoids flow fluid over bioengineered devices. They first demonstrated more advanced renal structure and function in human nephroid organs, which is important for creating tissue fragments for drug testing and disease modeling, and ultimately in vivo therapy.
In 2018, the first artificial kidney capable of producing urine emerged [3]. The researchers bathed human embryonic stem cells in a culture medium containing molecules that promote kidney development. The cells were combined with a gelatinous substance that mimics natural connective tissue and injected under the skin of mice to form a tiny clump. After three months, the tissue has developed into nephrons, the basic units of kidney structure and function. These renal organoids consist of proximal tubules, distal tubules, and Bowman's capsule. The team used the fluorescent protein Dextran to bind to a urinal-like substrate to measure the "urine" produced by the nephron filtering blood, which confirmed the kidney organoid's ability to filter blood and produce urine.
In 2019, scientists successfully cultured a vascularized biological artificial kidney by exposing the renal organoids to the shear stress of cellular fluid [4]. This maturation of vascularization is a work that lays the foundation for the eventual success of the biological artificial kidney. They are not yet sure that blood from kidneys can be infused at any time, but it is a significant improvement over transplanting into animals.
In 2021, Zhongwei Li et al. report for the first time a novel 3D culture method that can be used in vitro to culture, expand, and induce the differentiation of human and mouse primary collecting duct progenitor cells (GSCS) or human pluripotent stem cells (hPSC)-induced collecting duct progenitor cells into mature collecting ducts as organoids [5]. This system provides an important element in collecting tube progenitor cells for the synthesis of artificial kidneys from stem cells. Combined with gene-editing technology, this set of extracorporeal inducible collecting duct organoid models can be widely applied to study kidney development and disease mechanism.
Acute kidney injury is a very common clinical condition, usually caused by different medications. When human kidney organoids were exposed to the chemotherapy drug cisplatin, Navin Gupta and colleagues found that many genes, including FANCD2 and Rad51, were activated during repair, but their expression declined as kidney damage becomes irreversible [6]. They finally identified a compound called SCR7 through drug screening tests that helps maintain the activity of FANCD2 and RAD51 to salvage normal tissue repair and prevent the progression of chronic kidney disease (CKD) in the researchers' model of cisplatin-induced organoid injury.
2. Research Targets for Kidney Diseases:
References:
[1] Minoru Takasato, Pei X. Er, et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis [J]. Nature 526, 564–568 (2015).
[2] Ryuji Morizane, Joseph Bonventre, et al. Nephron organoids derived from human pluripotent stem cells model kidney development and injury [J]. Nat Biotechnol. 2015 Nov; 33(11): 1193–1200.
[3] Ioannis Bantounas, Parisa Ranjzad, et al. Generation of Functioning Nephrons by Implanting Human Pluripotent Stem Cell-Derived Kidney Progenitors [J]. Stem Cell Reports. 2018 Mar 13;10(3):766-779.
[4] Homan KA, Gupta N, Kroll KT, et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro [J]. Nat Methods. 2019 Feb 11.
[5] Zeng, Z., Huang, B., Parvez, R.K. et al. Generation of patterned kidney organoids that recapitulate the adult kidney collecting duct system from expandable ureteric bud progenitors [J]. Nat Commun 12, 3641 (2021).
[6] Navin Gupta, Takuya Matsumoto, et al. Modeling injury and repair in kidney organoids reveals that homologous recombination governs tubular intrinsic repair [J]. Science Translational Medicine, 2022; 14 (634).