Intestinal Organoids
After the Clevers research group 2007 proved that Lgr5+ epithelial cells are true intestinal epithelial stem cells in mouse models [1], the field of intestinal stem cell research has made great progress in the following years. One of the most influential breakthroughs in this field is the establishment of an in vitro 3D culture system for intestinal stem cells. Clevers and his colleagues generated gut organoids from adult intestinal stem cells upon 3D culture in Matrigel in 2009 [2]. This was the first description of Gut organoid culture was first described using mouse small intestinal segments.
3D intestinal organoids consist of a closed circulating hollow cavity lined with a layer of intestinal epithelial cell lines. Differentiated cell lineages of the intestinal epithelium, including intestinal epithelial cells, enteroendocrine cells, and goblet (Paneth) cells, line the villous zone. Intestinal organoids can be derived from both organ-restricted adult stem cells (ASCs) and pluripotent stem cells (PSCs). Organoids generated from these two stem cells contain all of the intestinal epithelial cell types found in vivo, in similar proportions and arrangements. They can simulate intestinal tissue to a greater extent and therefore are called "mini-guts". Colorectal tumor cells can also be cultured with this 3D system to obtain tumor-like organs. In general, intestinal organoids are valuable models for studying intestinal nutrient transport, drug absorption, gastrointestinal cancer, and intestinal glucagon hormone secretion.
Intestinal organoids are now widely used to study human diseases. Compared with previous intestinal disease models including cell line culture or in vivo animal experiments, organoid culture has obvious advantages, such as high culture success rate, short culture period, small tissue difference, and ease to establish a low malignant tumor model. Compared with traditional experimental models (2D cell line culture and animal model), intestinal organoids have many advantages. The intestinal organoid model constructed in vitro contains all types of intestinal epithelial cells and has physiological functions such as water and iron absorption and transport, which are incomparable to single-cell lines. In addition, there is a large species difference between the animal model and human model, which cannot completely simulate the real physiological reaction of the human intestine, while intestinal organoids derived from human tissues can more accurately reflect the physiological and pathological changes of the human body.
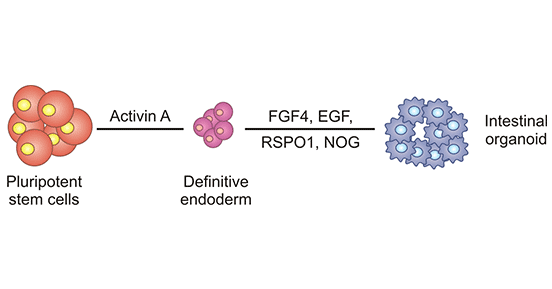
Figure 1. Establishment process of intestinal organoids
1. Recent Research on Gut Organoids
Y. Ui et al. [3] reported in 2012 that mouse experiments confirmed that intestinal organoids cultured in vitro could repair Dextransulfatesodium (DSS)-induced colitis by anal enema and that these cultured organoids could accurately reach the injured epithelial site and repair damaged intestinal epithelium.
Laween Meran et al. used the intact nanostructured decellularized human intestinal matrix as a bioscaffold [4]. Proteomic and Raman spectroscopy analyses revealed highly similar biochemical signatures of the human small intestine and colon scaffolds, suggesting that they can be used interchangeably as platforms for intestinal engineering. Then, the researchers implanted the ex vivo-expanded jejunal organoids on any type of scaffold to effectively reconstruct a hollow graft with physiological functions such as digestion and absorption. The graft was transplanted into the mouse kidney capsule or subcutaneously and can survive and form luminal structures. This study provided proof-of-concept data for the use of engineered patient-specific jejunum transplantation in children with intestinal failure and advanced the clinical trial process for the treatment of children with intestinal failure.
On July 3, 2020, organoid research pioneer Hans Clevers and his team published an article titled "SARS-CoV-2 productively infects human gut enterocytes" in the journal Science, which found that in small intestinal organoids SARS-CoV-2 easily infects intestinal epithelial cells, and the viral response genes of the infected intestinal epithelial cells are significantly up-regulated [5]. This study provided evidence that intestinal epithelial cells support SARS-CoV-2 replication, suggesting that human intestinal organoids can serve as experimental models for viral infection and biological studies.
Ana C Silva et al. used a human multi-lineage iPSC-derived organoid that recapitulates the coordinated development and maturation of the heart and gut in vivo, demonstrating that the presence of endodermal tissue (gut/intestine) in the organoids contributed to the development and maturation of the cardiac tissue [6]. This study showed that cooperative tissues from many germ lineages may be produced and matured inside a single organoid model, a breakthrough that will aid in the investigation of multi-tissue interactions during development, physiological maturation, and disease.
Satoshi Watanabe et al. used a flexible catheter to infuse about 1,000 organoids into the colon where the most epithelial damage occurred [7]. Epithelial cells of the cultured organoids are attached to the damaged surface and bound to the host's epithelial tissue (the layer of cells inside the colon), eventually forming an intact epithelial tissue in which part of the damaged epithelial layer has been replaced by organoid epithelial cells. This study lays the groundwork for a human clinical trial using colon organoid transplantation to treat severe ulcerative colitis. Today, the protocol developed in this study has been translated into clinical practice, and both the scientific and clinical communities are excited about future clinical applications.
2. Research Targets for Intestinal Diseases
References:
[1] Barker N, van Es J H, Kuipers J, et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature, 2007, 449: 1003–1007.
[2] Sato, T., Vries, R. G., et al. (2009). Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche [J]. Nature 459, 262–265.
[3] Yui S, Nakamura T, Sato T, et al. Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5+ stem cell. Nat Med, 2012, 18: 618–623.
[4] Laween Meran, Isobel Massie, et al. Engineering transplantable jejunal mucosal grafts using patient-derived organoids from children with intestinal failure [J]. Nature Medicine volume 26, pages1593–1601 (2020).
[5] Mart M. Lamers, Joep Beumer, et al. SARS-CoV-2 productively infects human gut enterocytes [J]. Science. 2020 May 1 : eabc1669.
[6] Ana C Silva, Oriane B Matthys, et al. Co-emergence of cardiac and gut tissues promotes cardiomyocyte maturation within human iPSC-derived organoids [J]. Cell Stem Cell. 2021 Dec 2;28(12):2137-2152.e6.
[7] Satoshi Watanabe, Sakurako Kobayashi, et al. Transplantation of intestinal organoids into a mouse model of colitis [J]. Nat Protoc. 2022 Mar;17(3):649-671.