Lung Organoids
The lung system is composed of two parts, the airway and the alveolar space. The complexity of its organization and cells ensures the immune defense and gas exchange functions of the lung. Although traditional in vitro cell experiments and animal models have been widely used to elucidate human lung development, physiology and pathogenesis, none of these models can accurately recapitulate the interaction between the human lung environment and cells. Studies have found that lung organoids are currently the closest model to the human lung system, while in vitro lung models represented by lung organoids have also become a more accessible tool for studying lung development, function, and disease pathology.
1. Application of Lung Organoids
Lung organoids reproduce characteristics of the human airway, such as mucosal secretion, ciliary beating, and regeneration. Using this biological correlation, lung organoids can be applied to study the repair/regeneration mechanisms of lung injury as well as phenotypic changes in lung disease. They can also be used for toxicity assessment or drug testing.
Lung organoids are hollow, with cavities or cavities inside, so they are easily transmitted by light. This makes them suitable for 3D biological assays and confocal imaging for quantitative characterization of cellular contents, assessment of dead and alive, and cell sorting for specific markers.
Organoids produced from lung cells can also be used to study early-stage lung cancer and to identify and test potential treatments. The lung cancer organoid model can simulate the complex cellular structure and biological behavior of the tumor on the basis of retaining the genomic changes of the parental tumor. Therefore, lung cancer organoids are widely used in signal transduction, biomarker research, as well as lung cancer drug screening and efficacy prediction. In recent years, lung cancer organoid models have become important preclinical models in the field of tumor research.
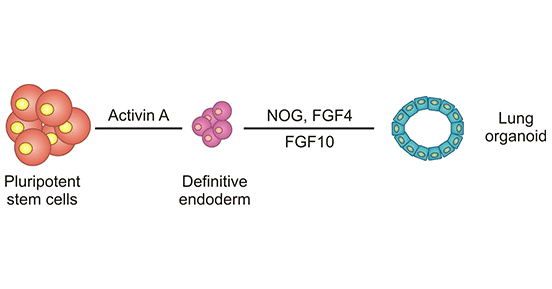
Figure 1. Establishment process of lung organoids
2. Recent Research on Lung Organoids
The first self-organizing 3D structures of adult human airway epithelium were described in collagen by Puchelle and colleagues in 1993 [1]. Rossant and colleagues first described the creation of lung organoids from human iPSCs, which included the use of CFTR-mutant iPS cells as a proof-of-concept for simulating cystic fibrosis (CF) [2]. Dye BR et al. adopted a modified trajectory to produce mature lung organoids with basal, ciliated, and club cells [3].
In 2017, Hans-Willem Snoeck and colleagues used hiPSCs to generate lung organoids, which were the first cultures to contain branching airways and alveolar structures and were similar to human lung tissues [4]. Expression analysis and structural characterization showed that branching reached mid-gestation in humans. The researchers infected the lung organoids with the respiratory syncytial virus (RSV), causing small airway obstruction and bronchiolitis, consistent with what was observed in the lungs. Tan Q et al. created airway organoids using a 3D combination of human adult primary bronchial epithelial cells, lung fibroblasts, and lung microvascular endothelial cells [5].
In 2021, Professor Honglong Ji from the University of Texas Taylor Medical Center published an online article entitled "Fibrinolytic niche is required for alveolar type 2 cell-mediated alveologenesis via a uPA-A6-CD44+-ENaC signal cascade" in the well-known journal Signal Transduction and Targeted Therapy in the field of targeted therapy. This study is the first in the world to discover a new regulation mechanism of urokinase uPA on the regeneration of lung epithelial stem cells through the A6-CD44+-ENaC signaling axis [6]. Through the in vitro culture of alveolar organoids, it is the first time to prove the regulation of the fibrinolytic system on ENaC, revealing that the fibrinolytic system may serve as a new intervention target for alveolar stem cell repair and regeneration.
COVID-19 is escalating globally, and organoids play a critical part in the investigation of SARS-CoV-2. To closely resemble the circumstances after SARS-CoV-2 infection in the patients, the researchers blended lung organoids with other organoids [7]. SARS-CoV-2 infection of the lung organoids highly recapitulates real-world patients' lung infections and shows the specialized functions different cell types play in infected lungs. Lung and other organoids, as well as the multi-tissue organ-on-a-chip platform, could be used for identifying the pathological process of the organs after SARS-CoV-2 infection, screening candidate drugs, and developing and assessing vaccines for safety and efficacy [8-10].
References:
[1] Benali R, Tournier JM, et al. (1993) Tubule formation by human surface respiratory epithelial cells cultured in a three-dimensional collagen lattice [J]. Am J Physiol 264: L183–L192.
[2] Wong AP, Bear CE, et al. (2012) Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein [J]. Nat Biotechnol 30: 876–882.
[3] Dye BR, Hill DR, et al. (2015) In vitro generation of human pluripotent stem cell derived lung organoids [J]. Elife 4: e05098.
[4] Chen, YW., Huang, S., de Carvalho, A. et al. A three-dimensional model of human lung development and disease from pluripotent stem cells [J]. Nat Cell Biol 19, 542–549 (2017).
[5] Tan Q, Choi KM, Sicard D, Tschumperlin DJ. (2017) Human airway organoid engineering as a step toward lung regeneration and disease modeling [J]. Biomaterials 113: 118–132.
[6] Gibran Ali, Mo Zhang, et al. Fibrinolytic niche is required for alveolar type 2 cell-mediated alveologenesis via a uPA-A6-CD44+-ENaC signal cascade [J]. Signal Transduction and Targeted Therapy 6, Article number: 97 (2021).
[7] Han Y, Duan X, et al. Identification of SARS-CoV-2 inhibitors using lung and colonic organoids [J]. Nature. 2020;589(7841):270–75.
[8] Zhao B, Ni C, Gao R, et al. Recapitulation of SARS-CoV-2 infection and cholangiocyte damage with human liver ductal organoids [J]. Protein Cell. 2020;11(10):771–5.
[9] Meyer-Berg H, Zhou Yang L, et al. Identification of aav serotypes for lung gene therapy in human embryonic stem cell-derived lung organoids [J]. Stem Cell Res Ther. 2020;11(1):448.
[10] Katsura H, Sontake V, et al. Human lung stem cell-based alveolospheres provide insights into SARS-CoV-2-mediated interferon responses and pneumocyte dysfunction [J]. Cell Stem Cell. 2020;27(6):890–904.