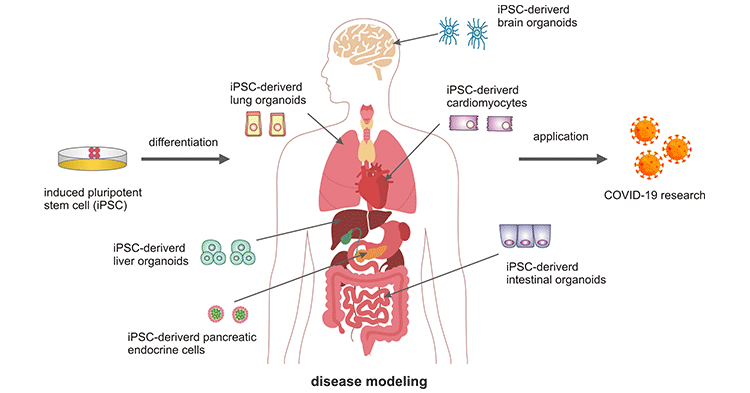
Organoids and SARS-CoV-2 Infection
Since the emergence of SARS-CoV-2 in late 2019, human beings around the world have been continuously attacked. Although experts have developed several effective vaccines, this stubborn virus still cannot be completely controlled or eliminated. And there are currently no specific drugs for SARS-CoV-2. Constant mutation and evolution of the SARS-CoV-2 make related research more difficult. The lack of humanized viral infection models that study the pathogenesis of SARS-CoV-2 and the methods to inhibit the virus is one of the key problems restricting the development of COVID-19 treatment candidates.
The application of organoids in infectious disease modeling was underestimated to a large extent before the emergence of the SARS-CoV-2 pandemic. Organoids are versatile models for viral, bacterial, and protozoan infection. They can provide quick and reliable information on pathogen biology and drug screening, and thus play a critical role in the fight against new infectious diseases.
Figure 1. Application of human iPSC-derived organoid models for COVID-19 research
The picture is cited from: https://www.frontiersin.org/articles/10.3389/fcell.2021.720099/full
1. Recent Studies of Organoid Applications in SARS-CoV-2
Considering highly similarity of organoids with corresponding organs in vivo in the histology and genetic characteristics, organoid corps came into play as a "stand-in" for studying SARS-CoV-2-host interactions [1]. This corps is composed of the lung, brain, heart, kidney, and other organoid models. These organoids are mainly used to simulate SARS-CoV-2 infection experiments and then to study the infection targets and screen drug candidates.
Paola Spitalieri et al. successfully prepared long-term culturable human lung organoids (hLORGs) in the process of simulating the development and maturation of human fetal lung organs in vitro [2]. The model is currently being used for cell tropism, host cell response, genetic variation affecting virus susceptibility, screening of new drug candidates, etc.
Yuling Han et al. have shown that lung organoid models induced by human pluripotent stem cells (hPSC-LOs) are capable of infection with SARS-CoV-2 and exhibit strong chemokine induction of SARS-CoV-2 infection [3]. It was found that alveolar type II-like cell (AT2) markers can be highly expressed in stem cells, and AT2-like cell populations in lung organoids are highly similar to human lung AT2 cells. In addition, ACE2, TMPRSS2, and FURIN were all enriched in AT2-like cells.
In 2020, Eric Song et al. observed significant infection of human brain organoids after infection with SARS-CoV-2, accompanied by metabolic changes of infected neurons and adjacent neurons [4]. Jacob et al. found that the expression of inflammatory signaling-related genes was up-regulated in the brain organoid model infected with SARS-CoV-2 [5]. Wang et al. identified the important pathogenic factor of SARS-CoV-2: ApoE4 using the brain organoid model [6].
The Clevers group observed SARS-CoV-2 infection in human AdSC-derived intestinal organoid samples, re-emphasizing the ability of organoids to rapidly and efficiently reproduce the viral infection process in vitro [7]. Mithal et al. identified several interferon-related genes in human intestinal organoids that can be infected and activated by SARS-CoV-2, including BST2, OASL, MX1, IFITM1, and IRF7 [8]. The discovery of these genes provides a strong basis and research direction for subsequent drug development.
COVID-19 research also uses other related organoids, including kidney, retina, vascular, and liver organoids. Monteil et al. demonstrated that SARS-CoV-2 can directly interact with vascular and renal organoids and that this interaction can be blocked by the clinical-grade soluble ACE2 [9]. Yang et al. have shown that human liver organoids and bile duct organoids are also susceptible to SARS-CoV-2 infection and that the induction pattern of chemokines is similar to that found in patient samples [10].
Researchers have found some SARS-CoV-2-associated proteins and conducted structure-function studies and drug design & screening on them through organoid models.
2. Related Hot Targets on SARS-CoV-2 Research:
References:
[1] Bang M. Tran,Georgia Deliyannis, et al. Organoid Models of SARS-CoV-2 Infection: What Have We Learned about COVID-19 [J]? Organoids 2022, 1(1), 2-27.
[2] Spitalieri, Paola, et al. Two Different Therapeutic Approaches for SARS-CoV-2 in hiPSCs-Derived Lung Organoids [J]. Cells, 2022, 11.7: 1235.
[3] Yuling Han, Xiaohua Duan, et al. Identification of SARS-CoV-2 Inhibitors using Lung and Colonic Organoids [J]. Nature. 2021 Jan;589(7841):270-275.
[4] Song, Eric, et al. Neuroinvasive potential of SARS-CoV-2 revealed in a human brain organoid model [J]. 2020.
[5] Jacob, Fadi, et al. Human pluripotent stem cell-derived neural cells and brain organoids reveal SARS-CoV-2 neurotropism predominates in choroid plexus epithelium [J]. Cell stem cell, 2020, 27.6: 937-950. e9.
[6] Wang, Lu, et al. A human three-dimensional neural-perivascular 'assembloid' promotes astrocytic development and enables modeling of SARS-CoV-2 neuropathology [J]. Nature medicine, 2021, 27.9: 1600-1606.
[7] Lamers, Mart M., et al. SARS-CoV-2 productively infects human gut enterocytes [J]. Science, 2020, 369.6499: 50-54.
[8] Mithal, Aditya, et al. Human pluripotent stem cell-derived intestinal organoids model SARS-CoV-2 infection revealing a common epithelial inflammatory response [J]. Stem cell reports, 2021, 16.4: 940-953.
[9] Monteil V, Kwon H, Prado P, et al. Inhibition of SARS-CoV-2 Infections in Engineered Human Tissues Using Clinical-Grade Soluble Human ACE2 [J].Cell, 2020, S0092-8674 (20) 30399-8.
[10] YANG, Liuliu, et al. A human pluripotent stem cell-based platform to study SARS-CoV-2 tropism and model virus infection in human cells and organoids [J]. Cell stem cell, 2020, 27.1: 125-136. e7.