Bill Gates, one of the world's richest men and founder of Microsoft, announced a $100 million research grant in the fall of 2017 to help with Alzheimer's research.
September 21st is World Alzheimer's Disease Day. According to the data of the International Alzheimer's Disease Association as of September 2022, about 50 million people worldwide suffer from Alzheimer's disease. As the average human life expectancy increases and the aging society intensifies, the prevalence of Alzheimer's disease is also rising. It is estimated that by 2050, the number of Alzheimer's patients will increase to more than 150 million.
1. What Is Alzheimer's Disease?
Alzheimer's disease (AD) is a neurodegenerative disorder during presenium and senectitude, characterized by cognitive dysfunction and progressive memory decline [1]. It is an irreversible brain disease that destroys memory, thinking, and the ability to carry out daily behaviors. And it is the most common type of dementia.
It initially causes a progressive loss of episodic memory and cognitive function and subsequently triggers verbal and visuospatial skills deficiencies, which are often accompanied by behavioral disorders such as apathy, aggressiveness, and depression.
Since Alzheimer's disease is a slowly progressive disease, it begins many years before symptoms appear and deteriorates with time. The rate of progression and the abilities that are impacted differ from person to person. Alzheimer's is not curable, however, medications can assist neurons in the brain temporarily interacting with one another and so ease symptoms for varied periods of time.
Alzheimer's disease affects 10% of people over the age of 65 years around the world, and the risk of developing Alzheimer's disease doubles every 5 years.
The characteristic pathological change is cerebral cortex atrophy with beta-amyloid protein deposition, neurofibrillary tangles, loss of mush memorability neurons, and senile plaques. At present, there is no powerful treatment or drugs to delay the disease progression.
2. What Causes Alzheimer's Disease?
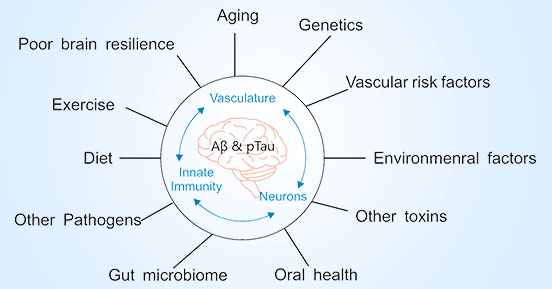
Unfortunately, the pathogenesis of Alzheimer's disease has not yet been fully deciphered due to its complexity. Although the exact cause of Alzheimer's disease is not yet fully understood, several factors are thought to increase the risk of developing the condition, including genetic, lifestyle, environmental, and epigenetic factors.
Figure 1. The pathogenic factors of Alzheimer's disease
At present, the mainstream view is that the gradual deposition of extracellular plaques of insoluble β-amyloid peptide (Aβ) and the accumulation of intracellular tau-containing neurofibrillary tangles (NFT) in the brain leads to neuronal death and cognitive impairment [2][3].
Amyloid-β (Aβ) peptide is the main component of neuritic or senile plaques outside neurons in the brain parenchyma and derives from the proteolytic cleavage of amyloid precursor protein (APP) by β-secretases and γ-secretases. Progressive accumulation of Aβ peptide interferes with neuron-to-neuron communication at synapses and possibly contribute to cell death.
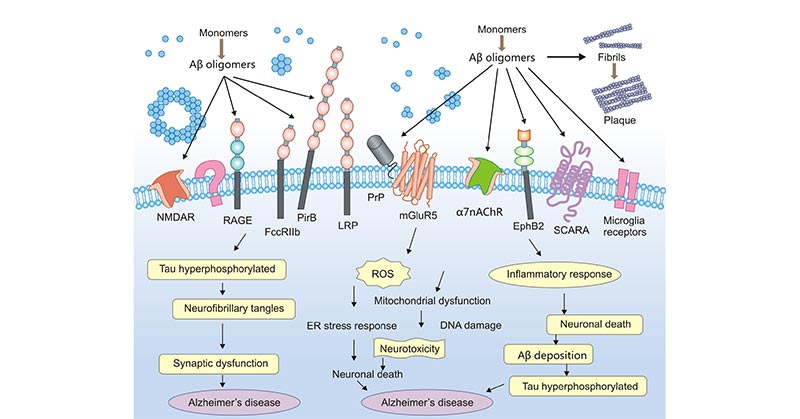
Aβ promotes the development of Alzheimer's disease and triggers a harmful cascade that includes Tau pathology and neurodegeneration [4]. The Aβ binding to multiple Aβ receptors including the P75NRT, LRP, PrPc, mGluR5, α7nAChR, NMDAR, β-AR, EphR, and PirB, activate downstream signaling pathways to generate reactive oxygen species (ROS), hyperphosphorylated Tau protein, producing and transducing neurotoxic signals into neurons, resulting in cellular defects such as mitochondrial dysfunction, the endoplasmic reticulum (ER) stress response, inflammatory responses, and neuronal death [5].
Figure 2. Biological functions of Aβ in Alzheimer's disease
This picture is cited from: https://www.nature.com/articles/aps201728
Aβ peptides also build up as vascular amyloid around the blood vessels of the brain which is viewed as cerebral amyloid angiopathy (CAA) [6]. Vascular amyloid impairs the brain's ability to absorb essential nutrients from the blood and leads to vascular dysfunction in the brain.
Abnormal deposits of the microtubule-associated protein tau in the form of neurofibrillary tangles inside neurons, hinder the transport of cargo inside neurons. This is a major driver of neuronal dysfunction and cell death and is closely linked to clinical disease progression. Tau also interacts with Fyn kinase to mediate Aβ toxicity [7].
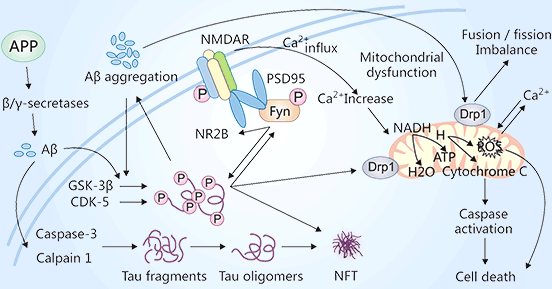
Aβ stimulates GSK-3β and CDK5 to phosphorylate tau protein and caspase-3 and calpain-1 to hydrolyze tau protein, creating tau oligomers that ultimately form NFTs. Aβ-activated Fyn speeds up tau phosphorylation and binds to tau. The action of phosphorylated Fyn on NR2B forms the NMDAR-PSD95-Fyn complex. Ca2+ levels rise as a result of NMDAR activation, which impacts the function of mitochondria. By acting on Drp1, Aβ and phosphorylated tau cause the fusion and fission of mitochondria. This disruption of mitochondrial dynamics causes an increase in reactive oxygen species (ROS) and apoptosis [8].
Figure 3. Reciprocal toxicity between Aβ and tau
This picture is cited from: https://www.ijbs.com/v17p2181.htm
Eventually, both extracellular amyloid-β deposits and intracellular tau tangles cause irreversible lesions in the brain, leading to atrophy of the brain and a loss of cognitive function.
Increasing evidence has demonstrated that inhibition of the interaction may be more effective than solely focusing on the neurotoxicity of Aβ or tau alone.
3. What Is the Best Treatment for Alzheimer's Disease?
Although it is a common neurodegenerative disease, Alzheimer's disease is insidious and difficult to diagnose early, which has caused a heavy social and economic burden to the global public health system. There is currently no cure for Alzheimer's disease, but medicines are available that can help relieve some of the symptoms.
Before Aduhelm's launch, only six Alzheimer's drugs had been approved by the FDA, including tacrine (1993), donepezil (1996), rivastigmine (2000), galantamine (2001), memantine hydrochloride (2003), and memantine/donepezil combination (2014). And these drugs can only improve the symptoms of patients, do not reduce the pathological changes of AD, nor can they reverse or slow down the disease process. Donepezil, rivastigmine, and galantamine are acetylcholinesterase (AChE) inhibitors, while memantine is an N-methyl-D-aspartate (NMDA) receptor antagonist.
Aduhelm (Aducanumab), was approved by the FDA in June 2021 and is the first FDA-approved drug to address the underlying biology of Alzheimer's disease rather than the symptoms. Aducanumab is a high-affinity, amyloid-β–directed, full-humanized IgG1 monoclonal antibody. It selectively binds to amyloid deposits in the brains of AD patients and then clears them out by activating the immune system.
Although no breakthroughs have been made so far, researchers have never stopped searching for clues and therapies for Alzheimer's disease.
The success of any clinical research is closely related to the contribution of basic scientific research. CUSABIO offers multiple products for scientific research for Alzheimer's disease, including ELISA kits, recombinant proteins, and antibodies.
|
Molecular Target
|
Product Name
|
|
Amyloid beta
|
Human amyloid beta peptide 1-40, Aβ1-40 ELISA Kit
|
|
Mouse amyloid beta peptide 1-40, Aβ1-40 ELISA Kit
|
|
Rat amyloid beta peptide 1-40, Aβ1-40 ELISA Kit
|
|
Human amyloid beta peptide 1-42, Aβ1-42 ELISA Kit
|
|
Monkey amyloid beta peptide 1-42, Aβ1-42 ELISA Kit
|
|
Mouse amyloid beta peptide 1-42, Aβ1-42 ELISA Kit
|
|
Rat amyloid beta peptide 1-42, Aβ1-42 ELISA Kit
|
|
Recombinant Human Amyloid-beta precursor protein (APP)
|
|
tau
|
Human phosphorylated microtubule-associated protein tau (pMAPT /pTAU) ELISA kit
|
|
Human cleaved microtubule-associated protein tau (C-MAPT/C-TAU) ELISA kit
|
|
Human Tau proteins ELISA kit
|
|
Rat Tau Proteins ELISA kit
|
|
MAPT Antibody
|
|
MAPT Monoclonal Antibody
|
|
Phospho-MAPT (S324) Antibody
|
|
Phospho-MAPT (S396) Antibody
|
|
Phospho-MAPT (T231) Antibody
|
|
MAPT Antibody, HRP conjugated
|
|
MAPT Antibody, FITC conjugated
|
|
MAPT Antibody, Biotin conjugated
|
|
Phospho-MAPT (Ser356) Antibody
|
|
Phospho-MAPT (Thr181) Antibody
|
|
Phospho-MAPT (Thr212) Antibody
|
|
Phospho-MAPT (Ser404) Antibody
|
|
Recombinant Mouse Microtubule-associated protein tau (Mapt)
|
|
Recombinant Rat Microtubule-associated protein tau (Mapt)
|
|
Recombinant Macaca mulatta Microtubule-associated protein tau (MAPT)
|
|
Recombinant Bovine Microtubule-associated protein tau (MAPT)
|
|
Recombinant Goat Microtubule-associated protein tau (MAPT)
|
|
Recombinant Hylobates lar Microtubule-associated protein tau (MAPT)
|
|
Recombinant Pan troglodytes Microtubule-associated protein tau (MAPT)
|
|
Recombinant Gorilla gorilla gorilla Microtubule-associated protein tau (MAPT)
|
|
Recombinant Pongo pygmaeus Microtubule-associated protein tau (MAPT)
|
|
Recombinant Spermophilus citellus Microtubule-associated protein tau (MAPT)
|
|
Recombinant Papio hamadryas Microtubule-associated protein tau (MAPT)
|
References
[1] Selkoe D. Alzheimer's disease: genes, proteins, and therapy [J]. Physiological reviews. 2001;81:741-66.
[2] Kang S, Lee YH, Lee JE. Metabolism-centric overview of the pathogenesis of Alzheimer's disease [J]. Yonsei Med J. 2017;58(3):479–88.
[3] Bloom GS . Amyloid-beta and tau: the trigger and bullet in Alzheimer disease pathogenesis [J]. JAMA Neurol 2014; 71: 505–8.
[4] Busche M, Hyman B. Synergy between amyloid-β and tau in Alzheimer's disease [J]. Nature neuroscience. 2020;23:1183-93.
[5] Canevari L, Abramov AY, Duchen MR. Toxicity of amyloid beta peptide: tales of calcium, mitochondria, and oxidative stress [J]. Neurochem Res 2004; 29: 637–50.
[6] Viswanathan A, Greenberg SM. Cerebral amyloid angiopathy in the elderly [J]. Ann Neurol 2011; 70: 871–80.
[7] Roberson E, Scearce-Levie K, et al. Reducing endogenous tau ameliorates amyloid beta-induced deficits in an Alzheimer's disease mouse model [J]. Science (New York, NY). 2007;316:750-4.
[8] Huiqin Zhang, Wei Wei, et al. Interaction between Aβ and Tau in the Pathogenesis of Alzheimer's Disease [J]. Int J Biol Sci 2021; 17(9):2181-2192.
CUSABIO team. How Does Alzheimer's Disease Happen?. https://www.cusabio.com/c-20423.html



Comments
Leave a Comment