[1] Fürstenau, Cristina Ribas, et al. "Ecto-nucleotide pyrophosphatase/phosphodiesterase as part of a multiple system for nucleotide hydrolysis by platelets from rats: kinetic characterization and biochemical properties." Platelets 17.2 (2006): 84-91.
[2] Iqbal, Jamshed. "Ectonucleotidases: potential target in drug discovery and development." Mini Reviews in Medicinal Chemistry 19.11 (2019): 866-869.
[3] Stella, Joséli, et al. "Differential ectonucleotidase expression in human bladder cancer cell lines." Urologic Oncology: Seminars and Original Investigations. vol. 28. no. 3. Elsevier, 2010.
[4] Bollen, Mathieu, et al. "Nucleotide pyrophosphatases/phosphodiesterases on the move." Critical reviews in biochemistry and molecular biology 35.6 ( 2000): 393-432.
[5] Ferrero, Enza, Angelo C. Faini, and Fabio Malavasi. "A phylogenetic view of the leukocyte ectonucleotidases." Immunology Letters 205 (2019): 51-58.
[6] Sakagami, Hideki, et al. "Biochemical and molecular characterization of a novel choline-specific glycerophosphodiester phosphodiesterase belonging to the nucleotide pyrophosphatase/phosphodiesterase family." Journal of Biological Chemistry 280.24 (2005): 23084-23093.
[7] Orriss, Isabel R., et al. "The regulation of osteoblast function and bone mineralization by extracellular nucleotides: the role of p2x receptors." Bone 51.3 (2012): 389-400.
[8] Vollmayer, Petra, et al. "Hydrolysis of diadenosine polyphosphates by nucleotide pyrophosphatases/phosphodiesterases." European journal of biochemistry 270.14 (2003): 2971-2978.
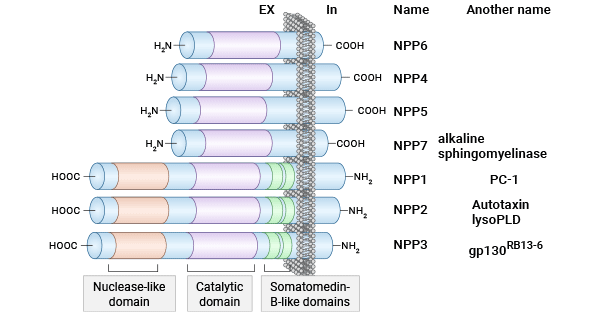
[9] Borza, Razvan, et al. "Structure and function of the ecto-nucleotide pyrophosphatase/phosphodiesterase (ENPP) family: tidying up diversity." Journal of Biological Chemistry 298.2 (2022).
[10] Goding, James W., Bert Grobben, and Herman Slegers. "Physiological and pathophysiological functions of the ecto-nucleotide pyrophosphatase/ phosphodiesterase family." Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1638.1 (2003): 1-19.
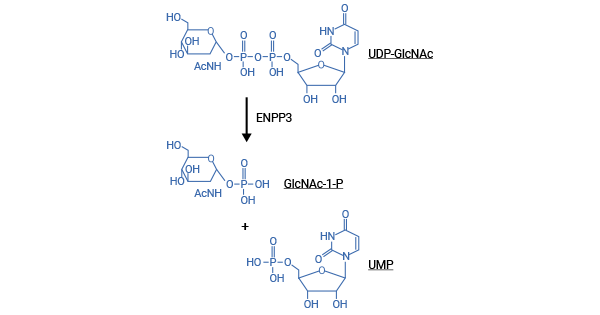
[11] Korekane, Hiroaki, et al. "Identification of ectonucleotide pyrophosphatase/phosphodiesterase 3 (ENPP3) as a regulator of N- acetylglucosaminyltransferase GnT-IX (GnT-Vb)." Journal of Biological Chemistry 288.39 (2013): 27912-27926.
[12] Taniguchi, Naoyuki, et al. "Identification of ectonucleotide pyrophosphatase/phosphodiesterase 3 as a new modifier of glycan biosynthesis (788.3). " The FASEB Journal 28 (2014): 788-3.
[13] Stone, Kelly D., Calman Prussin, and Dean D. Metcalfe. "IgE, mast cells, basophils, and eosinophils. "Journal of Allergy and Clinical Immunology 125.2 ( 2010): S73-S80.
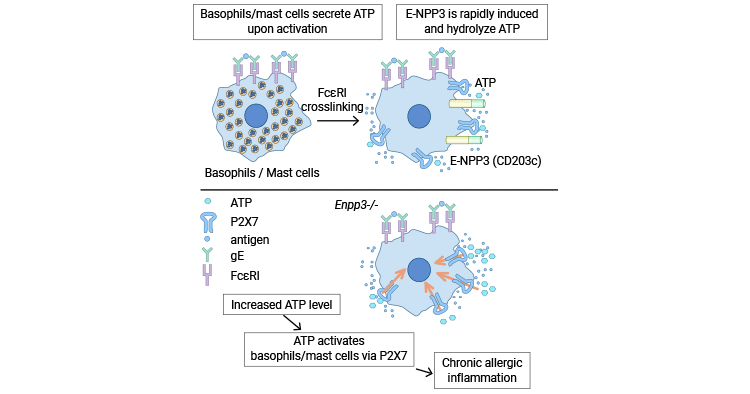
[14] Tsai, Shih Han, et al. "The ectoenzyme E-NPP3 negatively regulates ATP-dependent chronic allergic responses by basophils and mast cells." Immunity 42.2 (2015): 279-293.
[15] Lourenço, F. D., et al. "Activated status of basophils in chronic urticaria leads to interleukin-3 hyper-responsiveness and enhancement of histamine release induced by anti-IgE stimulus." British Journal of Dermatology 158.5 (2008): 979-986.
[16] Bühring, Hans-Jörg, Anna Streble, and Peter Valent. "The basophil-specific ectoenzyme E-NPP3 (CD203c) as a marker for cell activation and allergy diagnosis." International archives of allergy and immunology 133.4 (2004): 317-329.
[17] Ono, Emiko, et al. "CD203c expression on human basophils is associated with asthma exacerbation." Journal of allergy and clinical immunology 125.2 (2010): 483-489.
[18] Eberlein-König, B., et al. "Comparison of basophil activation tests using CD63 or CD203c expression in patients with insect venom allergy ." Allergy 61.9 (2006): 1084-1085.
[19] Bühring, Hans-Jörg, Anna Streble, and Peter Valent. "The basophil-specific ectoenzyme E-NPP3 (CD203c) as a marker for cell activation and allergy diagnosis." International archives of allergy and immunology 133.4 (2004): 317-329.
[20] Mikkelsen, Susan, et al. "Basophil sensitivity through CD63 or CD203c is a functional measure for specific immunotherapy." Clinical and Molecular Allergy 8 (2010): 1-9.
[21] Oda, Yoshiko, et al. "Improved FcεRI-mediated CD203c basophil responsiveness reflects rapid responses to omalizumab in chronic spontaneous urticaria." The Journal of Allergy and Clinical Immunology: In Practice 9.3 (2021): 1166-1176.
[22] Eberlein, Bernadette, et al. "Optimizing of the basophil activation test: comparison of different basophil identification markers." Cytometry Part B : Clinical Cytometry 88.3 (2015): 183-189.
[23] Reimer, J. M., et al. "Isolation of transcriptionally active umbilical cord blood-derived basophils expressing FcɛRI, HLA -DR and CD203c." Allergy 61.9 (2006): 1063-1070.
[24] Hauswirth, A. W., et al. "CD203c is overexpressed on neoplastic mast cells in systemic mastocytosis and is upregulated upon IgE receptor cross-linking. " International journal of immunopathology and pharmacology 21.4 (2008): 797-806.
[25] Kambe, Naotomo, et al. "Development of both human connective tissue-type and mucosal-type mast cells in mice from hematopoietic stem cells with identical distribution pattern to human body." Blood 103.3 (2004): 860-867.
[26] Krauth M T, Mirkina I, Herrmann H, et al. Midostaurin (PKC412) inhibits immunoglobulin E-dependent activation and mediator release in human blood basophils and mast cells[J]. Clinical & Experimental Allergy, 2009, 39(11): 1711-1720.
[27] Takagi, Shinsuke, et al. "Membrane-bound human SCF/KL promotes in vivo human hematopoietic engraftment and myeloid differentiation." Blood, The Journal of the American Society of Hematology 119.12 (2012): 2768-2777.
[28] Masini E, Vannacci A, Failli P, et al. A carbon monoxide-releasing molecule (CORM-3) abrogates polymorphonuclear granulocyte-induced activation of endothelial cells and mast cells[J]. The FASEB Journal, 2008, 22(9): 3380-3388.
[29] Vannacci A, Giannini L, Fabrizi F, et al. Effects of the carbon monoxide releasing molecule CORM-3 in a coincubation model of rat mast cells with human neutrophils[J]. Inflammation Research, 2007, 56: S13-S14.
[30] Doñate F, Raitano A, Morrison K, et al. AGS16F is a novel antibody drug conjugate directed against ENPP3 for the treatment of renal cell carcinoma[J]. Clinical Cancer Research, 2016, 22(8): 1989-1999.
[31] Trapero C, Jover L, Fernández-Montolí M E, et al. Analysis of the ectoenzymes ADA, ALP, ENPP1, and ENPP3, in the contents of ovarian endometriomas as candidate biomarkers of endometriosis[J]. American Journal of Reproductive Immunology, 2018, 79(2): e12794.
[32] Chen L, Pan X, Hu X H, et al. G ene expression differences among different msi statuses in colorectal cancer[J]. International journal of cancer, 2018, 143(7): 1731-1740.
[33] Li J, Mo Y, Wei Q, et al. High Infiltration of CD203c+ Mast Cells Reflects Immunosuppression and Hinders Prognostic Benefit in Stage II-III Colorectal Cancer[J]. Journal of Inflammation Research, 2023: 723-735.
[34] Ullah S, El-Gamal M I, Zaib S, et al. Synthesis, biological evaluation, and docking studies of new pyrazole-based thiourea and sulfonamide derivatives as inhibitors of nucleotide pyrophosphatase/phosphodiesterase[J]. Bioorganic Chemistry, 2020, 99: 103783.
[35] Ullah S, El-Gamal M I, El-Gamal R, et al. Synthesis, biological evaluation, and docking studies of novel pyrrolo [2, 3-b] pyridine derivatives as both ectonucleotide pyrophosphatase/phosphodiesterase inhibitors and antiproliferative agents[J]. European Journal of Medicinal Chemistry, 2021, 217: 113339.
[36] Goltzman D. Inferences from genetically modified mouse models on the skeletal actions of vitamin D[J]. The Journal of Steroid Biochemistry and Molecular Biology, 2015, 148: 219-224.
[37] Zhou M, Qi L, Gu Y. GRIA2/ENPP3 regulates the proliferation and migration of vascular smooth muscle cells in the restenosis process Post-PTA in lower extremity arteries[J]. Frontiers in Physiology, 2021, 12: 712400.
[38] Ullah S, Pelletier J, Sévigny J, et al. Synthesis and Biological Evaluation of Arylamide Sulphonate Derivatives as Ectonucleotide Pyrophosphatase/ Phosphodiesterase-1 and-3 Inhibitors[J]. ACS omega, 2022, 7(30): 26905-26918.

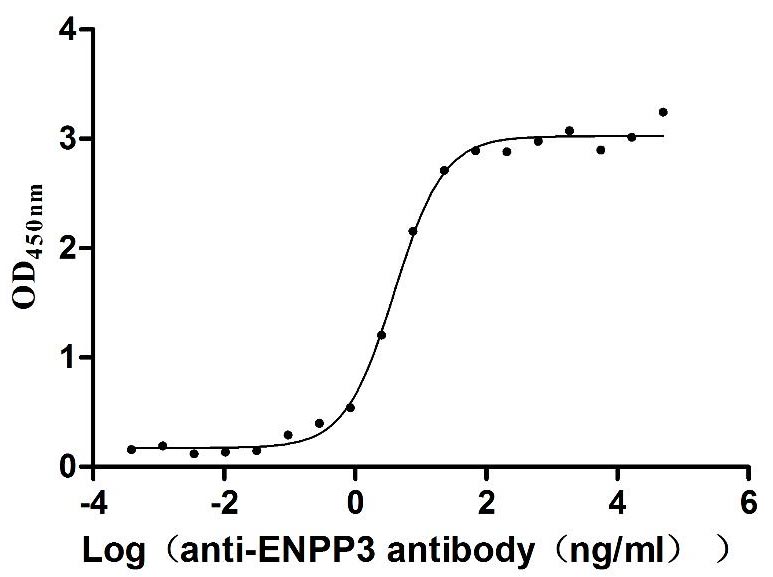
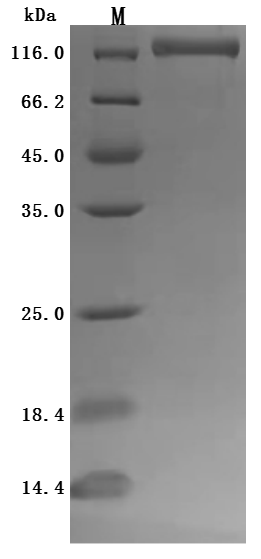
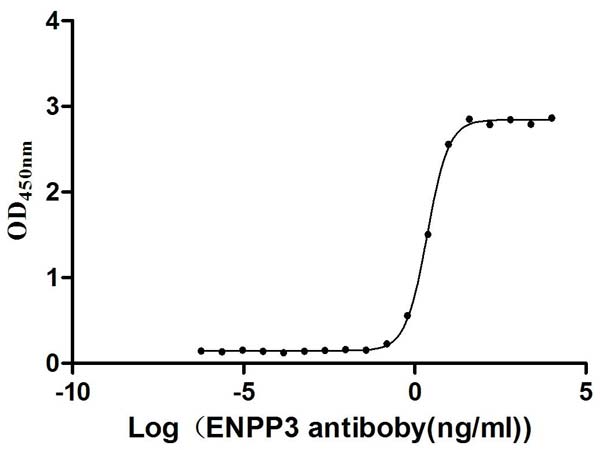
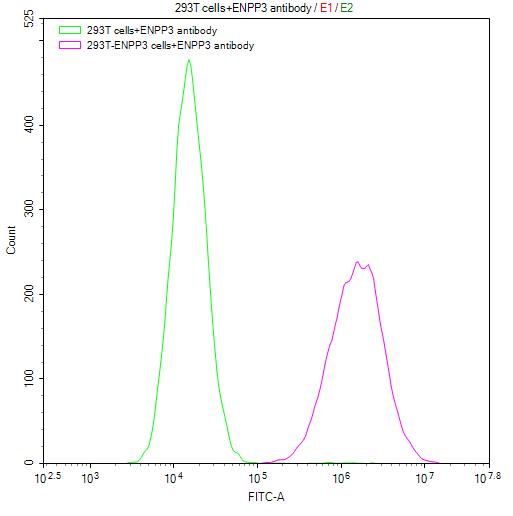


Comments
Leave a Comment