Hematopoietic stem cells (HSC) are a type of adult stem cells with the characteristics of stem cells: self-renewal and differentiation potential.
Hematopoietic stem cells are a kind of pluripotent stem cells derived from bone marrow, which are the "seed" cells in the blood system. HSCs forms all the cells in the blood system in the process of hematopoiesis, including various mature cells such as white blood cells, red blood cells, platelets and so on. When properly stimulated, it also differentiates into other non-hematopoietic tissues (such as adipocytes, cardiomyocytes, endothelial cells and pancreatic cells) [1].
Hematopoietic stem cells have biological functions such as regulating body balance, immune function, anti-microorganism, anti-inflammation and so on. It plays an important role in the treatment of hematological diseases, hereditary hematological diseases and autoimmune diseases.
1. Type of Hematopoietic Stem Cell
Two types of hematopoietic stem cells have been defined:
Long-term regenerative cells (LTRC) capable of maintaining self-renewal and multilineage differentiation potential throughout life.
Short-term regenerative cells (STRC). They remain pluripotent, but they show more limited potential for self-renewal.
The interval between these two types of cells to reconstruct the myeloid and / or lymphatic system is very short, about 6 weeks.
2. Sources of Hematopoietic Stem Cells
Hematopoietic stem cells are present in adult bone marrow, especially in the pelvis, femur and sternum. They are also found in umbilical cord blood and a small amount of peripheral blood.
2.1 Bone Marrow
Hematopoietic stem cell (HSCs) is a kind of pluripotent stem cell derived from bone marrow.
Hematopoietic stem cells obtained from bone marrow were collected surgically from two posterior iliac crest. About one out of every 100000 cells in bone marrow is a long-term hematopoietic stem cell (LT-HSC).
2.2 Peripheral Blood
Most hematopoietic stem cells come from the bone marrow, and a small number of stem cells and progenitor cells circulate in the blood. In clinical transplantation of human hematopoietic stem cells, donor cells can be collected from peripheral blood. Under the action of hematopoietic growth factors such as granulocyte colony stimulating factor (G-CSF), HSC in bone marrow is mobilized to peripheral blood, and then hematopoietic stem cells are collected by isolation.
2.3 Umbilical Cord Blood (UCB)
Umbilical cord blood is a rich source of hematopoietic stem cells and hematopoietic progenitor cells, and the number of different types of hematopoietic progenitor cells is about 10 times that observed in adult blood. As a special cell source of regenerative medicine, umbilical cord blood also contains many types of stem cells.
2.4 Fetal Hematopoietic System and Embryonic Hematopoietic Stem cells
Fetal hematopoietic system is an important source of hematopoietic stem cells, but it has not been applied in clinic.
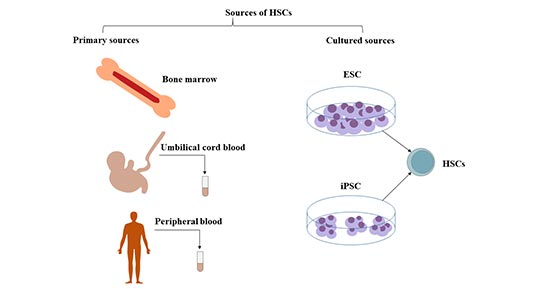
Figure 1 Sources of hematopoietic stem cells
3. Biological Characteristics of Hematopoietic Stem Cells
Hematopoietic stem cells are real stem cells because they are pluripotent and self-renewing. Other features include its heterogeneity.
Self-renewal: self-renewal of hematopoietic stem cells means that hematopoietic stem cells produce identical progeny cells. This ability enables hematopoietic stem cells to produce a complete hematopoietic system from a single cell and maintain hematopoiesis throughout an individual's life.
Pluripotency: It refers to the ability of hematopoietic stem cells to produce major hematopoietic cell types when needed. HSCs can differentiate into various blood cells from myeloid lineages (monocytes and macrophages, neutrophils, basophils, eosinophils, erythrocytes, megakaryocytes / platelets, dendritic cells) and lymphatic lineages (T, B and NK cells).
Hematopoietic stem cells have a very high proliferation potential, so that they can meet the high demand for hematopoiesis in the lifetime of normal adults.
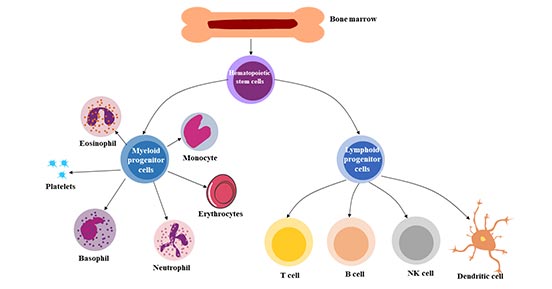
Figure 2 The pluripotency of hematopoietic stem cells
Plasticity: It refers to the ability of hematopoietic stem cells to differentiate into multiple non-hematopoietic tissues such as cardiomyocytes [2].
Hematopoietic stem cell transport: From embryonic origin, HSPC moves from one niche to another. Hematopoietic stem cell niche is the spatial location and physiological microenvironment on which hematopoietic stem cells rely for survival and self-renewal. The transport of hematopoietic stem cells can be divided into homing (describing the trend of cells arriving in a particular environment), retention (the ability of cells to remain in that environment after arrival) and implantation (the ability of hematopoietic stem cells to divide in the environment and form functional offspring).
Heterogeneity: Heterogeneity within hematopoietic stem cells means that hematopoietic stem cells have different physiological characteristics, such as cell cycle state [3] and self-renewal ability [4], have different responses to different external signals, and can form different lineages [5] [6] [7] [8] after transplantation.
The Muller-Sieberg group was one of the first groups to determine HSC heterogeneity. The heterogeneity of HSC can spread stably. How is the heterogeneity within hematopoietic stem cells produced?
The following are possible reasons for HSC heterogeneity:
-
Different sources of hematopoietic stem cells: HSC may come from mesoderm cells, endothelial cells or from anterior hematopoietic stem cells of dorsal aorta.
-
Differences in regulatory factors: the type of HSC may be regulated by different induced developmental tissues (such as AGM, placenta, yolk sac, head) and may change with their migration in circulation, or by developmental niche (possibly blood vessels, liver, nerves or bones), or by different developmental mechanisms (such as EHT).
-
In addition, epigenetic modifications may also explain the existence of different HSC types.
4. HSC and Immune System
HSC niche is the spatial location and physiological microenvironment on which hematopoietic stem cells rely for survival and self-renewal. It maintains the dynamic balance between self-renewal and differentiation of stem cells.
Studies have shown that in vivo, the niche of hematopoietic stem cells provides immune privileged sites for hematopoietic stem cells. Regulatory T cells (tregs) and hematopoietic stem cells are located in the endosteal region of the bone marrow to protect the hematopoietic stem cells from immune attack.
In addition, hematopoietic stem cells themselves have a certain immune immunity potential by regulating the expression of surface immune molecules. When HSCs was activated by strong inflammatory signals and mobilized into circulation, the level of CD47 on the surface of HSCs was significantly up-regulated. CD47 binds to signal regulatory proteins on macrophages to inhibit phagocytosis. The increased expression of CD47 on the surface of mobilized hematopoietic stem cells can protect these cells from phagocytosis.
5. Hierarchy of Human Hematopoietic System
The origin of all blood cells in the hematopoietic system is thought to come from self-renewing hematopoietic stem cells.
Hematopoietic stem cell and progenitor cell (HSPCs) bank can be divided into three types of cells: long-term hematopoietic stem cell (LT-HSC), short-term hematopoietic stem cell (ST-HSPC) and multipotent progenitor cells (MPP).
In the hematopoietic system, long-term hematopoietic stem cells (LT-HSC) are located at the top of all mature blood cells, maintaining a primitive multipotential pool throughout life through their self-renewal and asymmetric cell division potential. In a stable state, progeny derived from hematopoietic stem cells have the ability to rapidly proliferate in peripheral stimulation, replacing the loss of activated, consumed or aged blood cells [9] [10].
St-HSPC and its progenitor cells have the ability to maintain normal hematopoiesis for 6-8 weeks. Loss of normal LT-HSC and ST-HSPC functions is a marker of natural stem cell aging and several hematopoietic diseases, especially related to the development and progression of hematological malignancies [11].
Pluripotent progenitor cell (MPPs) is a pluripotent progenitor cell that has lost its self-renewal potential but can still differentiate into all multiple lines produced by hematopoietic stem cells. MPPs further produce oligopotent progenitors cells, the common lymphoid progenitor (CLPs) and common myeloid progenitor (CMPs).
All these low-potential progenitor cells can differentiate into committed lineages:
CMPs can differentiate into megakaryocyte / erythrocyte progenitor cell (MEPs), granulocyte / macrophage progenitor cell (GMPs) and dendritic cell (DC) progenitor cell.
CLPs can differentiate into T cell progenitor cells, B cell progenitor cells, NK progenitor cells and DC progenitor cells [12] [13].
DC progenitor cells (CD8+DC, CD8-DC and plasma cell-like DC) can be derived from CMPs and CLPs.
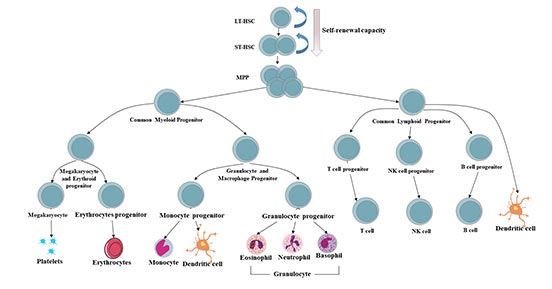
Figure 3 Hierarchy of human hematopoietic system
6. Cell surface Markers of Hematopoietic Stem Cells
Although there are many studies on hematopoietic stem cells, no single molecular marker has been found to be expressed only by hematopoietic stem cells.
Hematopoietic stem cells are called Lin- because of the lack of expression of mature blood cell markers. The identification or isolation of hematopoietic stem cells is generally based on the combination of several different cell surface markers to separate rare hematopoietic stem cells from the surrounding blood cells.
There are many differences between human hematopoietic stem cell and mouse hematopoietic stem cell markers.
Mouse HSC: EMCN+, CD34lo/-, SCA-1+, Thy1.1+/lo, CD38+, C-kit+, lin-
Human HSC: EMCN+, CD34+, CD59+, Thy1/CD90+, CD38lo/-, C-kit/CD117+, lin-
The markers that distinguish mouse hematopoietic stem cells (LT-HSC) from short-term (ST-HSC) and multipotent progenitor cells (early MPP and late MPP) are as follows.
LT-HSC: CD34-, CD38-, SCA-1+, Thy1.1+/lo, C-kit+, lin-, CD135-, Slamf1/CD150+
ST-HSC: CD34+, CD38+, SCA-1+, Thy1.1+/lo, C-kit+, lin-, CD135-, Slamf1/CD150+, Mac-1 (CD11b)lo
Early MPP: CD34+, SCA-1+, Thy1.1-, C-kit+, lin-, CD135+, Slamf1/CD150-, Mac-1 (CD11b)lo, CD4lo
Late MPP: CD34+, SCA-1+, Thy1.1-, C-kit+, lin-, CD135high, Slamf1/CD150-, Mac-1 (CD11b)lo, CD4lo
6.1 Introduction of Major Markers
6.1.1 CD34
CD34 is one of the most important markers, and is the first widely studied molecule in the isolation and identification of hematopoietic stem cells and their progenitor cells. The expression of CD34 in human umbilical cord blood, bone marrow and peripheral blood was about 0.1-4.9% [14]. CD34 is expressed on 0.55% of human bone marrow cells, and it is also expressed on early progenitor cells, but not on mature bone marrow cells. Other surface markers have been used in conjunction with CD34 to distinguish primordial cell populations.
6.1.2 CD38
CD38, also known as cyclic ADP ribosomal hydrolase, is a glycoprotein present on the surface of many immune cells (leukocytes), including CD4+, CD8+, B lymphocytes and natural killer cells. CD38 markers are used to distinguish hematopoietic stem cell pluripotent progenitor cells (CD38 -) from committed progenitor cells (CD38+).
6.1.3 CD90 (Thy1)
CD90, also known as thy-1, is a 28-30 kDa GPI-linked membrane glycoprotein. CD90 is expressed in hematopoietic stem cells, neurons, thymus cells, peripheral T cells, fibroblasts and stromal cells.
The co-expression of CD90 on CD34 + CD38- cells defines hematopoietic stem cells, while CD34 + CD38-CD90 - defines multipotent progenitor cells.
6.1.4 CD117 (C-Kit)
CD117 is a 145kDa protein tyrosine kinase, also known as c-Kit. CD117 is expressed on pluripotent hematopoietic progenitor cells (about 1.4% of bone marrow cells), mast cells and acute myeloid leukemia cell (AML).
By binding to its ligands, it can induce the phosphorylation of CD117 and stimulate the proliferation and survival of primitive hematopoietic stem cells, red blood cells and monocytes. It plays an important role in gamete formation, melanin formation and hematopoiesis.
6.1.5 CD135 (Flk-2)
CD135, also known as FLK-2, FLT3 and Ly-72, is a type III tyrosine kinase receptor. CD135 is expressed not only in normal CD34+ hematopoietic stem cells, but also in malignant hematopoietic cells, including AML, ALL and CML BC.
The combination of CD135 and FLT3 ligand can regulate the growth of hematopoietic stem cells and promote the survival of primitive hematopoietic progenitor cells with myeloid and B lymphatic potential.
6.1.6 CD150 (SLAM)
CD150, also known as SLAM, type I transmembrane glycoprotein signal transduction lymphocyte activation molecule, is a typical member of SLAM subgroup of CD2 protein family.
SLAM is expressed in thymocytes, T cell subsets, B cells, dendritic cells, macrophages and hematopoietic stem cells.
6.1.7 CD184 (CXCR4)
CD184, is also called Fusin or CXCR4. It is widely expressed in blood and tissue cells, including B cells and T cells, monocytes, macrophages, dendritic cells, granulocytes, megakaryocytes / platelets, lymphoid cells, myeloid precursor cells, endothelial cells, epithelial cells, astrocytes and neurons.
In the bone marrow niche, hematopoietic stem cells express CXCR4. CXCR4 is the receptor of chemokine CXCL12 (SDF-1), which mediates blood cell migration and participates in B lymphocyte and bone marrow production, cardiogenesis, angiogenesis and cerebellar development.
The interaction between CXCR4 and SDF-1 is responsible for the homing and retention of HSC in the niche. The interaction of CXCR4/CXCL12 (SDF-1) in HSC niche is considered to be the mechanism of HSC mobilization [15].
6.1.8 Ly-6A/E (Sca-1)
Ly-6a /E, also known as sca-1, is a member of the ly-6 polygene family, a protein linked to hematopoietic stem cells by glycosylphosphatidylinositol (GPI).
Its expression on pluripotent hematopoietic stem cell (HSC) has been used as a marker of HSC in two kinds of Ly 6 haploid mice. Sca-1 positive hematopoietic stem cells were found in bone marrow, fetal liver, peripheral blood and spleen of adult animals. Ly-6A/E is thought to be involved in the regulation of T and B cell responses.
7. Clinical Application of HSC
7.1 Hematopoietic Cell Transplantation (HCT)
According to the source of stem cells, hematopoietic cell transplantation can be divided into bone marrow transplantation, peripheral blood stem cell transplantation and umbilical cord blood transplantation.
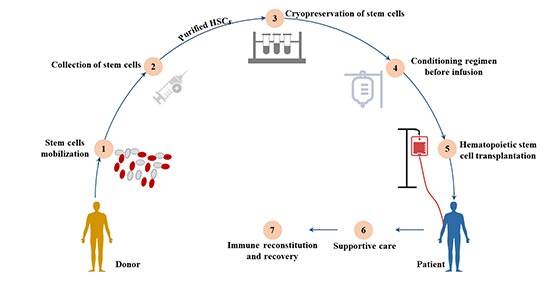
Figure 4 Hematopoietic cell transplantation (HCT)
7.1.1 Bone Marrow Transplantation
Bone marrow transplantation is a common type of hematopoietic stem cell transplantation. Hematopoietic stem cells play an important role in the re-proliferation of patients' blood cells in bone marrow transplantation.
According to the relationship between donors and recipients, it can be divided into autologous hematopoietic stem cell transplantation and allogeneic hematopoietic stem cell transplantation.
In addition to solid tumors, bone marrow transplantation is also used to treat hematological diseases such as leukemia and immune system disorders [16].
7.1.2 Peripheral Hematopoietic Stem Cell Transplantation
The concentration of circulating HSC in peripheral blood increased by 100 times after administration of hematopoietic cytokines such as GM-CSF, IL-3 or SCF. Using a cell separator, you can usually obtain the required number of HSCs from peripheral blood.
7.1.3 Umbilical Cord Blood Transplantation
Umbilical cord blood is easily available and does not pose a risk to donors, so it is an attractive source of transplantable hematopoietic stem cells.
Due to the immature immune system, lymphocytes in umbilical cord blood of newborns have been shown to be more tolerant to HLA, so there are fewer acute and chronic graft-versus-host disease (GVHD) in allogeneic transplantation environment.
7.2 Other
Hematopoietic stem cells are also widely used in graft anti-tumor therapy, tolerance induction, gene therapy [17], regenerative medicine and so on.
References
[1] Chotinantakul K, Leeanansaksiri W. Hematopoietic stem cell development, niches, and signaling pathways [J]. Bone marrow research, 2012, 2012.
[2] Chatterjee T, Sarkar R S, Dhot P S, et al. Adult stem cell plasticity: Dream or reality? [J]. Medical journal, Armed Forces India, 2010, 66(1): 56.
[3] Wilson A, Laurenti E, Oser G, et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair [J]. Cell, 2008, 135(6): 1118-1129.
[4] Ema H, Morita Y, Suda T. Heterogeneity and hierarchy of hematopoietic stem cells [J]. Experimental hematology, 2014, 42(2): 74-82. e2.
[5] Benz C, Copley M R, Kent D G, et al. Hematopoietic stem cell subtypes expand differentially during development and display distinct lymphopoietic programs [J]. Cell stem cell, 2012, 10(3): 273-283.
[6] Dykstra B, Kent D, Bowie M, et al. Long-term propagation of distinct hematopoietic differentiation programs in vivo [J]. Cell stem cell, 2007, 1(2): 218-229.
[7] Sieburg H B, Cho R H, Dykstra B, et al. The hematopoietic stem compartment consists of a limited number of discrete stem cell subsets [J]. Blood, 2006, 107(6): 2311-2316.
[8] Verovskaya E, Broekhuis M J C, Zwart E, et al. Heterogeneity of young and aged murine hematopoietic stem cells revealed by quantitative clonal analysis using cellular barcoding [J]. Blood, 2013, 122(4): 523-532.
[9] Busch K, Klapproth K, Barile M, et al. Fundamental properties of unperturbed haematopoiesis from stem cells in vivo [J]. Nature, 2015, 518(7540): 542.
[10] Sawai C M, Babovic S, Upadhaya S, et al. Hematopoietic stem cells are the major source of multilineage hematopoiesis in adult animals [J]. Immunity, 2016, 45(3): 597-609.
[1] Sun J, Ramos A, Chapman B, et al. Clonal dynamics of native haematopoiesis [J]. Nature, 2014, 514(7522): 322.
[11] Yao J C, Link D C. Concise review: the malignant hematopoietic stem cell niche [J]. Stem Cells, 2017, 35(1): 3-8.
[12] Adams G B, Scadden D T. The hematopoietic stem cell in its place [J]. Nature immunology, 2006, 7(4): 333.
[13] Mosaad Youssef Mohamed,Hematopoietic stem cells: an overview. [J] .Transfus. Apher. Sci., 2014, 51: 68-82.
[14] Pranke P, Hendrikx J, Debnath G, et al. Immunophenotype of hematopoietic stem cells from placental/umbilical cord blood after culture [J]. Brazilian journal of medical and biological research, 2005, 38(12): 1775-1789.
[15] Herbert K E, Levesque J P, Mills A K, et al. How we mobilize haemopoietic stem cells [J]. Internal medicine journal, 2011, 41(8): 588-594.
[16] Rogers I, Casper R F. Umbilical cord blood stem cells [J]. Best Practice & Research Clinical Obstetrics & Gynaecology, 2004, 18(6): 893-908.
[17] Hirschi K K. Hemogenic endothelium during development and beyond [J]. Blood, 2012, 119(21): 4823-4827.
CUSABIO team. Hematopoietic Stem Cell Surface Marker. https://www.cusabio.com/c-20980.html







Comments
Leave a Comment