[1] Nguyen-Thoi, T., et al. "A cell-based smoothed discrete shear gap method (CS-DSG3) using triangular elements for static and free vibration analyses of shell structures." International Journal of Mechanical Sciences 74 (2013): 32-45.
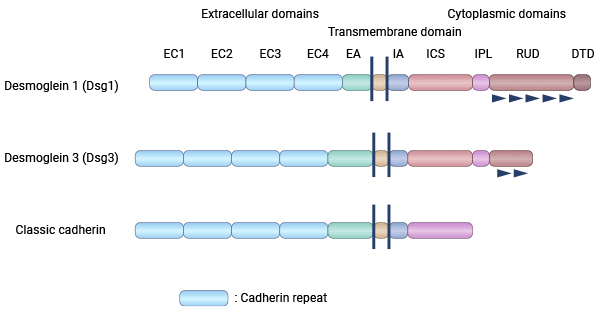
[2] Ishikawa, H., et al. "Cloning of the mouse desmoglein 3 gene (Dsg3): interspecies conservation within the cadherin superfamily." Experimental dermatology 9.4 (2000): 229-239.
[3] Amagai, Masayuki. "Desmoglein as a target in autoimmunity and infection." Journal of the American Academy of Dermatology 48.2 (2003): 244-252.
[4] Heupel, Wolfgang-Moritz, et al. "Pemphigus vulgaris IgG directly inhibit desmoglein 3-mediated transinteraction." The Journal of Immunology 181.3 (2008): 1825-1834.
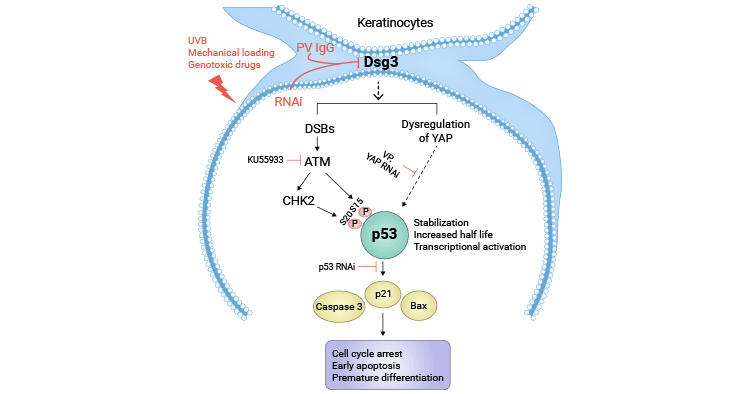
[5] Rehman, Ambreen, and Hong Wan. "A novel regulatory pathway of Desmoglein-3 in keratinocyte stress response." Journal of Cellular Signaling 1.4 (2020): 169-179.
[6] Bumiller-Bini Hoch, Valéria, et al. "Marked to Die-Cell Death Mechanisms for Keratinocyte Acantholysis in Pemphigus Diseases." Life 12.3 (2022): 329.
[7] Jolly, Puneet S., et al. "p38MAPK signaling and desmoglein-3 internalization are linked events in pemphigus acantholysis." Journal of Biological Chemistry 285.12 (2010): 8936-8941.
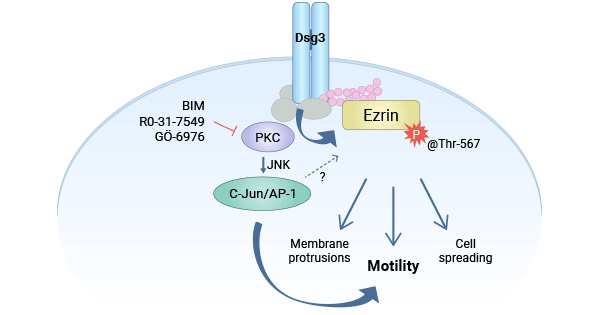
[8] Brown, L., et al. "Desmoglein 3 promotes cancer cell migration and invasion by regulating activator protein 1 and protein kinase C-dependent-Ezrin activation." Oncogene 33.18 (2014): 2363-2374.
[9] Savci-Heijink, Cemile Dilara, et al. "The role of desmoglein-3 in the diagnosis of squamous cell carcinoma of the lung." The American journal of pathology 174.5 (2009): 1629-1637.
[10] Baron, Sylvain, et al. "Unimpaired skin carcinogenesis in Desmoglein 3 knockout mice." PloS one 7.11 (2012): e50024.
[11] Amagai, Masayuki, et al. "Antibodies against desmoglein 3 (pemphigus vulgaris antigen) are present in sera from patients with paraneoplastic pemphigus and cause acantholysis in vivo in neonatal mice." The Journal of clinical investigation 102.4 (1998): 775-782.
[12] Brandão, Maria Luiza Figueiredo Braga, et al. "Refractory pemphigus vulgaris associated with herpes infection: case report and review." Revista do Instituto de Medicina Tropical de São Paulo 53 (2011): 113-117.
[13] Pitoia, Fabián, et al. "Prevalence of thyroid autoimmunity in patients with pemphigus vulgaris." MEDICINA-BUENOS AIRES- 65.4 (2005): 307.
[14] Scully, Crispian, and Stephen J. Challacombe. "Pemphigus vulgaris: update on etiopathogenesis, oral manifestations, and management." Critical Reviews in Oral Biology & Medicine 13.5 (2002): 397-408.
[15] Baker, John, Kristina Seiffert-Sinha, and Animesh A. Sinha. "Case report: Documentation of cutaneous only pemphigus vulgaris without history of mucosal lesions in North America." Frontiers in Immunology 13 (2022): 969279.
[16] Arbache, Samia Trigo, et al. "Immunofluorescence testing in the diagnosis of autoimmune blistering diseases: overview of 10-year experience." Anais brasileiros de dermatologia 89 (2014): 885-889.
[17] Candiz, María Emilia, et al. "Diagnóstico serológico de patologías ampollares autoinmunitarias." Dermatología Argentina 24.4 (2018): 177-184.
[18] Lim, Yen Loo, et al. "Autoimmune Pemphigus: latest advances and emerging therapies." Frontiers in Molecular Biosciences 8 (2022): 808536.
[19] Calle Isaza, Juliana, Isabel Cristina Ávila Gómez, and Ana María Abreu Vélez. "Autoimmune blistering diseases of the pemphigus group." Iatreia 27.3 (2014): 309-319.
[20] Aoyama, Yurni, and Yasuo Kitajima. "Pemphigus vulgaris-IgG causes a rapid depletion of desmoglein 3 (Dsg3) from the Triton X-100 soluble pools, leading to the formation of Dsg3-depleted desmosomes in a human squamous carcinoma cell line, DJM-1 cells." Journal of Investigative Dermatology 112.1 (1999): 67-71.
[21] Tsuchisaka, A., et al. "Presence of autoimmune regulator and absence of desmoglein 1 in a thymoma in a patient with pemphigus foliaceus." British Journal of Dermatology 173.1 (2015): 268-271.
[22] Huang, Yu-Mei, et al. "Integrated analysis of bulk and single-cell RNA sequencing reveals the interaction of PKP1 and tumor-infiltrating B cells and their therapeutic potential for nasopharyngeal carcinoma." Frontiers in Genetics 13 (2022): 935749.
[23] Dong, Boming, et al. "Computed tomographic images reflect the biologic behavior of small lung adenocarcinoma: they correlate with cell proliferation, microvascularization, cell adhesion, degradation of extracellular matrix, and K-ras mutation." The Journal of Thoracic and Cardiovascular Surgery 130.3 (2005): 733-739.
[24] Khoor, A., et al. "DSG3/TTF-1: A single chromogen antibody cocktail to distinguish between Squamous Cell Carcinoma (SCC) and adenocarcinoma of the lung." Virchows Arch 467.1 (2015): S1-S279.
[25] ZHANG, Xindong, et al. "Expression and clinical significance of CK5/6, DSG3, P40, TTF-1, CK7, NapsinA in small biopsy specimens of non-small cell lung cancer." Chinese Journal of Primary Medicine and Pharmacy (2020): 218-221.
[26] Pancewicz, J., and W. Niklinska. "A brief overview of clinical implications of desmoglein 3 in lung cancer." Progress in Health Sciences 11.2 (2021): 141-144.
[27] Collaborators at the Keio and Osaka Universities Present Key Non-Clinical Data with Stable/Functional-Induced Regulatory T Cells (S/F-iTregs) in Autoimmune Disease Models.

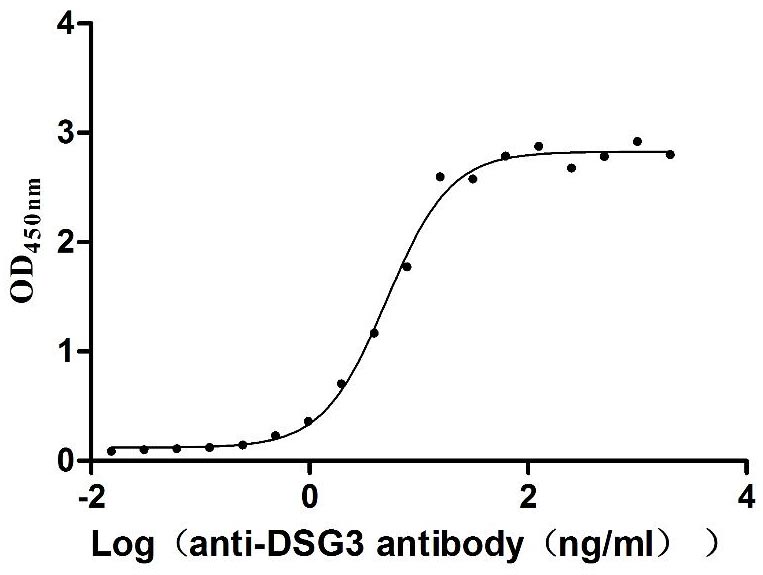

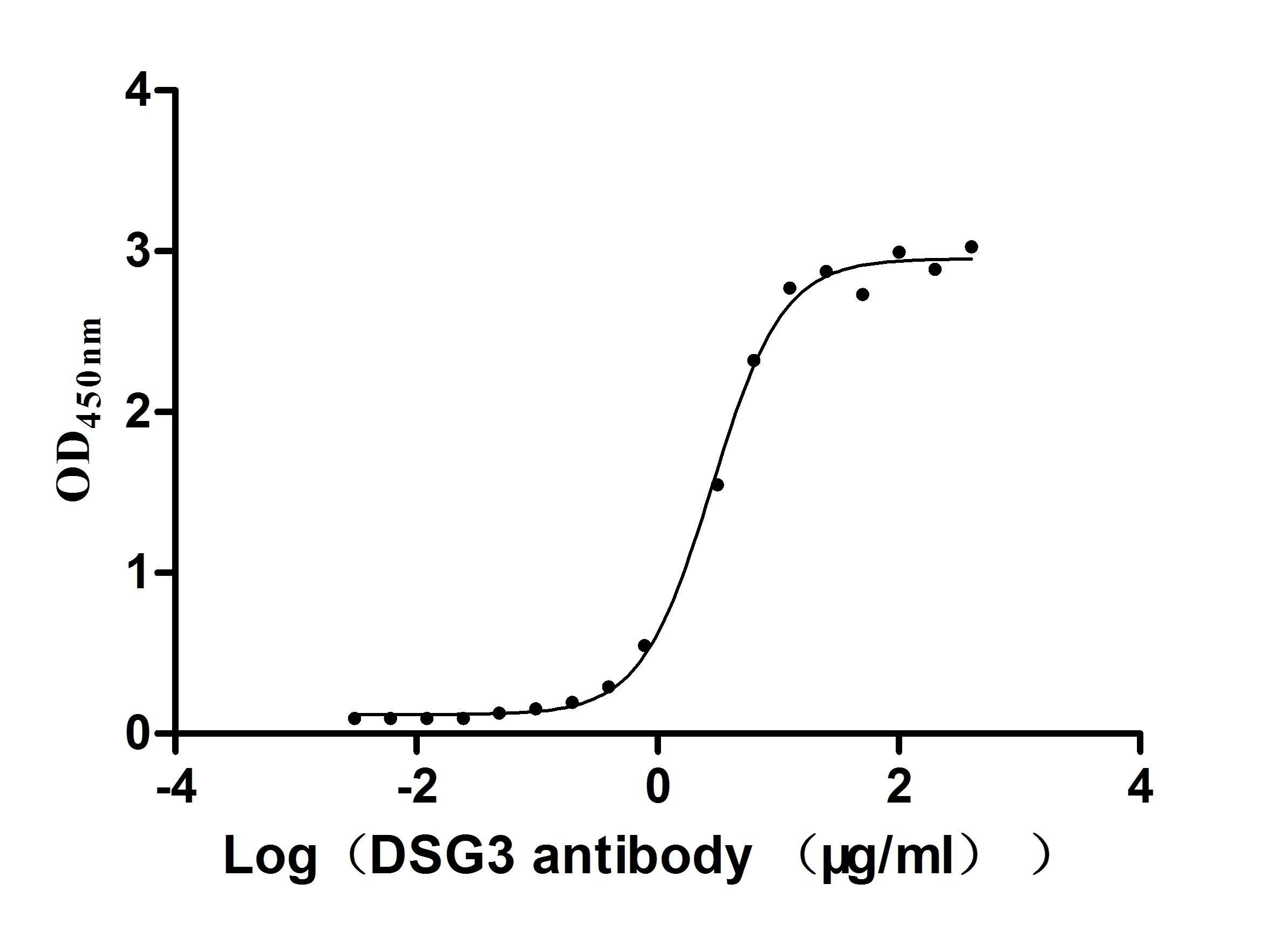


Comments
Leave a Comment