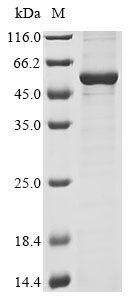
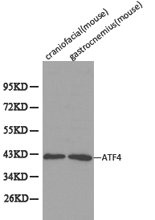
ATF4, activating transcription factor 4, alias CREB-2.ATF4 is a basic region leucine zipper transcription factor, a member of the bZIP family, encoded by the ATF4 gene in the human genome. It plays a key role in cellular regulation of multiple stress responses, including amino acid deficiency, oxidative stress, and endoplasmic reticulum stress, etc. The mechanism of action of ATF4 is mainly to regulate the transcription of downstream genes by binding to cAMP response elements (CREs) on the promoter regions of target genes or to the associated DNA sequences.
The biological significance of ATF4 is mainly reflected in its regulation of cellular metabolism, survival and stress response regulation. It is involved in the regulation of amino acid metabolism, antioxidant response, protein folding and damage repair. During endoplasmic reticulum stress, ATF4 can activate the unfolded protein response (UPR) to help cells restore protein homeostasis. In addition, ATF4 is involved in the regulation of the cell cycle, apoptosis, and autophagy, affecting cell fate.Abnormal function of ATF4 has been associated with a variety of diseases, including metabolic disorders, neurodegenerative disorders, and certain types of cancers, making it a potential target in drug development.