Recombinant proteins are proteins produced in an appropriate host system based on exogenous DNA rather than the host's native DNA. The exogenous DNA, also called the gene of interest (GOI), is introduced into the host cells through a suitable expression vector and then expressed into proteins using the host genetic machinery.
Currently, recombinant protein production is one of the most powerful techniques used in life sciences. Due to their advantages such as high purity, specificity, efficiency, safety, customization, scalability, and consistency, recombinant proteins have been widely applied in various fields, including medicine, biological research, and biotechnology, etc.
Table of Contents
1. Medicine
Among protein-based biopharmaceuticals, recombinant proteins form the largest category, encompassing enzymes, hormones, cytokines, growth factors, blood clotting factors, monoclonal antibodies (mAbs), and antibody-related products (e.g., Fc-fusion proteins and antibody fragments). They are used for therapeutics, diagnostics, and drug discovery and development, as well as vaccine development and production.
1.1 Therapeutic Recombinant Proteins
The most profound impact of recombinant technology is in the clinic, where it has created entirely new classes of drugs. These biologics offer targeted mechanisms of action that were unimaginable for traditional small-molecule pharmaceuticals, leading to more effective and specific treatments for chronic and severe diseases.
● Recombinant Enzymes
In many genetic disorders, individuals may lack functional enzymes due to genetic mutations. Recombinant enzymes can be designed to mimic the missing or defective enzymes. Recombinant glucocerebrosidase and recombinant alpha-galactosidase A are well-established enzyme replacement therapies (ERT) for Gaucher's disease and Fabry's disease, respectively.
● Recombinant Cytokines and Growth Factors (Replacement Therapies)
Some genetic disorders involve deficiencies in cytokines (including interleukins, interferons, and colony-stimulating factors), signaling proteins that regulate immune responses and cell growth. Recombinant cytokines can be administered to compensate for these deficiencies. Recombinant erythropoietin (EPO) can stimulate red blood cell production and is used in anemia.
Some recombinant cytokines, such as immune checkpoint proteins, also play a role in immunotherapy by amplifying the immune response against certain types of cancer. They function to stimulate the immune system, elicit immune cell responses, and enhance anti-tumor responses.
● Recombinant Hormones
Genetic conditions may lead to hormonal imbalances. Recombinant proteins can be engineered to mimic hormones and restore normal physiological functions. Recombinant insulin is used to control and treat diabetes. Recombinant human growth hormone (rhGH) is used to stimulate growth in children with growth disorders.
● Recombinant Clotting Factors
Hemophilia is a genetic disorder characterized by deficiencies in blood-clotting factors. Recombinant clotting factors (factor VIII and factor IX) have dramatically improved the quality of life and life expectancy for individuals with hemophilia.
Recombinant antibodies, especially monoclonal antibodies (mAbs), have revolutionized the treatment of various cancers, autoimmune diseases, and infectious diseases. Rituximab is a CD20 monoclonal antibody widely used in lymphoma. Trastuzumab (Herceptin) targets the HER2 receptor on certain breast cancer cells, inhibiting their growth and signaling the immune system to attack them. Adalimumab (Humira) binds to and neutralizes TNF-α, providing relief in conditions like rheumatoid arthritis and Crohn's disease.
1.2 Diagnostic Agents
Beyond therapy, recombinant proteins are the backbone of many diagnostic platforms. They serve as capture reagents, detection elements, calibrators, and sometimes as reference standards.
In immunoassays such as ELISA, chemiluminescent assays (CLIA), and lateral flow tests, recombinant proteins are used to:
- Present antigens to capture antibodies in patient samples (e.g., viral spike or nucleocapsid proteins in infection serology).
- Act as detection antibodies or labeled binding partners that recognize specific analytes.
- Serve as calibrators with known concentration to generate standard curves for quantitative readouts.
Compared with native or crude antigens, recombinant versions offer better batch‑to‑batch consistency, easier engineering of tags or domains, and quicker response when a new pathogen or variant emerges.
1.3 Precision Medicine
Recombinant proteins enable the development of targeted therapies tailored to specific molecular targets or pathways, improving treatment outcomes while minimizing side effects. Recombinant technology allows for the customization of therapeutic proteins to match the specific genetic variations present in individual patients.
1.4 Vaccines
Protein‑based subunit vaccines rely on recombinant antigens rather than whole pathogens. Recombinant proteins are engineered to express specific antigens derived from pathogens such as viruses or bacteria, which are highly immunogenic and can induce a protective immune response. These recombinant antigens, such as surface proteins, subunits, or epitopes, are key components in various vaccine platforms, including protein subunit vaccines, virus-like particle (VLP) vaccines, and conjugate vaccines.
Recombinant protein vaccines not only show a high safety profile but also are more stable in comparison to mRNA vaccines. Recombinant hepatitis B surface antigen (HBsAg) is used in hepatitis B vaccines. More recently, recombinant spike protein technology has been used in COVID-19 vaccine platforms.
Recombinant proteins can also be displayed on nanoparticles or virus‑like particles, enhancing immunogenicity and allowing precise control over antigen density and spacing. Human papillomavirus (HPV) vaccines include recombinant virus-like particles.
1.5 Drug Discovery and Development
Recombinant proteins are used in high-throughput screening (HTS) assays to identify potential drug candidates. Researchers can efficiently test large compound libraries against specific targets, expediting the drug discovery process.
Recombinant proteins are also used to design biological assays for studying the effects of potential drug candidates. In preclinical studies, these recombinant proteins help researchers understand the new drug's mechanism of action and assess its efficacy.
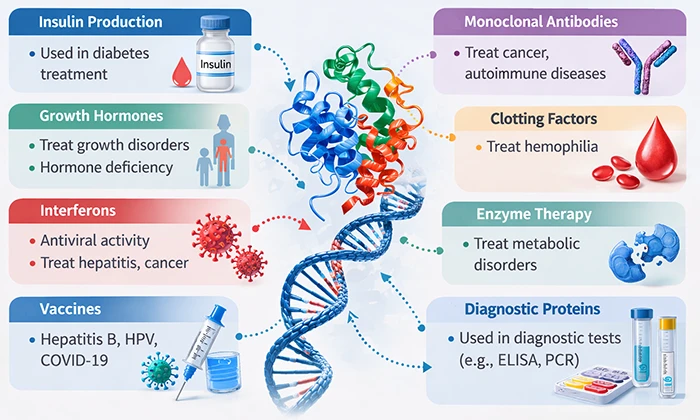
Figure 1. Applications of recombinant proteins in medicine
2. Biological Research
Recombinant proteins are essential tools in biological research, changing the way we study and understand cellular processes such as cell signaling, metabolism, growth, replication and death, transcription, translation, and protein modification.
2.1 Molecular and Cellular Tool Proteins
Recombinant proteins are central to many routine experiments:
- Cytokines and growth factors – Recombinant cytokines (such as interleukins) and growth factors (such as VEGF or EGF) are used to stimulate signaling pathways, drive differentiation, or maintain specific cell states in culture.
- Ligands, receptors, and enzymes – Purified recombinant receptors, kinases, proteases, and other enzymes are used to dissect pathways, measure activity, and validate targets in vitro.
- Tagged proteins – Affinity tags (e.g., His‑tag, FLAG‑tag, Fc‑fusion) and fluorescent fusions make it easier to purify, detect, or track proteins in pulldown assays, imaging, and interaction studies.
Because sequence and production are controlled, recombinant reagents can be standardized, which improves reproducibility across labs and over time.
2.2 Structural Biology and Biophysics
High‑quality recombinant proteins are the starting point for most structural and biophysical analyses. Crystallography, cryo‑electron microscopy, and NMR all depend on homogeneous, well‑folded samples at sufficient concentration.
Engineered constructs—such as truncated domains, stabilized mutants, or fusion partners that improve solubility—are often necessary to obtain good crystals or interpretable cryo‑EM maps. Once structures are available, they guide structure‑based drug design and rational protein engineering.
In parallel, biophysical assays such as surface plasmon resonance (SPR), isothermal titration calorimetry (ITC), and microscale thermophoresis (MST) use recombinant proteins to quantify binding affinities, kinetics, and thermodynamics. These parameters feed directly into medicinal chemistry and biological optimization.
2.3 Assay Development and High‑throughput Screening
Most biochemical and many cell‑based screening assays are built around recombinant proteins:
- Enzymatic assays – Recombinant enzymes are used to monitor activity in the presence of small molecules or biologics.
- Binding and competition assays – Recombinant targets and ligands provide defined systems to detect inhibitors, agonists, or blocking antibodies.
- High‑throughput screens (HTS) – Robust, stable recombinant proteins are essential to withstand the conditions of large‑scale screening campaigns.
Without reliable recombinant targets, it is hard to interpret screening data or reproduce hits across platforms.

Figure 2. Applications of recombinant proteins in biological research
3. Biotechnology
Recombinant proteins are also used in industry, food production, agriculture, and biomaterials and bioengineering.
3.1 Industry
In the breeding industry, enzymes can be added to animal feed to increase the nutritional value of feed ingredients, reduce feed and waste management costs, support animal gut health, enhance animal performance, and improve the environment. Amylases are used in the food industry to increase flavor, sweetness, and texture in various food items.
Cellulases are crucial enzymes in biofuel production, particularly in the conversion of lignocellulosic biomass into biofuels such as bioethanol. Proteases play a crucial role in detergent manufacturing, contributing to the effectiveness of laundry detergents and other cleaning products.
3.2 Food Production
Besides, lactic acid bacteria (LAB) have been used for a long time for the production of fermented foods. LAB has recently been engineered for the expression of recombinant proteins, which would have wide applications, such as improving human/animal digestion and nutrition.
3.3 Agriculture
Recombinant proteins have found various applications in agriculture; for example, crops can be genetically engineered to express proteins that confer resistance to pests, diseases (caused by viruses, bacteria, or fungi), or herbicides, contributing to improvements in crop yield and quality, and overall agricultural sustainability.
3.4 Biomaterials and Bioengineering
Recombinant proteins are used to design and produce functional biomaterials with tailored properties such as bioactivity, biocompatibility, and structural integrity. By incorporating specific protein sequences, these materials can mimic natural extracellular matrices, promoting cell adhesion, proliferation, and tissue regeneration.
Recombinant proteins contribute to the field of tissue engineering by providing bioactive components that support cell growth and tissue development. They can be engineered to mimic the structural and signaling features of native tissues, facilitating the development of organ transplantation.
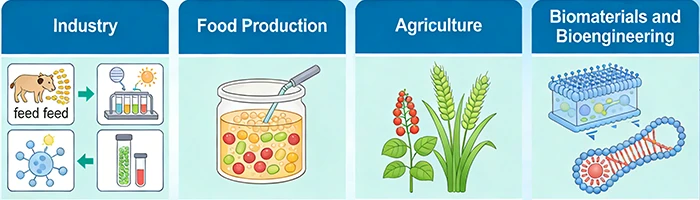
Figure 3. Applications of recombinant proteins in biotechnology
However, recombinant proteins also have limitations.
- In some cases, the production of recombinant proteins is complex, expensive, and time-consuming.
- The recombinant proteins produced in cells may not be the same as the natural forms. This difference may reduce the effectiveness of therapeutic recombinant proteins and even cause side effects. Additionally, this difference may affect the results of experiments.
- A major concern for all recombinant drugs is immunogenicity. All biotechnologically produced therapeutics may exhibit some form of immunogenicity. It is difficult to predict the safety of novel therapeutic proteins.
4. Latest Research on Recombinant Protein Applications
The versatility of recombinant protein technology is exemplified by its expanding applications across medicine and industry, from precise therapeutic targeting to industrial innovation. The following sections highlight key advances where recombinant proteins are driving progress, from new biologic drugs and vaccines to novel tools in food science and wound healing, illustrating the broad impact of this transformative platform.
4.1 Human medicine: rhBMP‑2 in spinal fusion and multidomain GLP‑1‑based fusion for improved type 2 diabetes therapy
Recombinant human BMP2 (rhBMP‑2) is a recombinant growth factor used to promote bone formation in spinal fusion surgery, where it can achieve fusion rates comparable to autologous iliac crest bone grafts. A 2024 narrative review concludes that rhBMP‑2 remains a potent osteoinductive agent for degenerative spine disease [1]. Still, it emphasizes the need for optimized dosing and safer delivery systems to reduce adverse events, such as ectopic bone formation and radiculitis.
4.2 Human vaccines: recombinant fusion PreS–RBD COVID‑19 vaccine and recombinant RBD XBB.1.5‑HR COVID‑19 vaccine
A 2024 preclinical study engineered a chimeric recombinant fusion protein (W‑PreS‑O) that combines the hepatitis B virus PreS domain with wild‑type and Omicron‑derived SARS‑CoV‑2 RBDs [2]. In mice, this recombinant W‑PreS‑O protein, formulated as a subunit vaccine, elicited very high Omicron‑specific neutralizing antibody titers, outperforming earlier PreS‑RBD constructs and a bivalent mRNA‑like comparator, indicating its promise as a booster targeting Omicron variants.
A 2025 study describes a self‑assembled trimeric recombinant protein vaccine composed of the SARS‑CoV‑2 XBB.1.5 receptor‑binding domain fused to heptad‑repeat regions (RBD XBB.1.5‑HR), produced in an insect‑cell expression system [3]. This recombinant RBD XBB.1.5‑HR vaccine induced strong neutralizing antibodies and T‑cell responses, protected animals against live Omicron EG.5.1 challenge, and boosted cross‑neutralizing titers against multiple XBB‑lineage and JN.1 subvariants in previously vaccinated humans.
4.3 Veterinary medicine: recombinant rHsp60 for Salmonella vaccines
In a 2024 systematic review of vaccines against Salmonella in animal hosts, a recombinant heat‑shock protein, rHsp60, is highlighted as one of the most efficacious subunit antigens [4]. Recombinant rHsp60 with incomplete Freund's adjuvant in mice conferred high levels of protection against Salmonella challenge, illustrating how a single bacterial recombinant protein can serve as a strong antigen for veterinary vaccine development.
4.4 Food industry: recombinant microbial amylases and lipases
A 2025 food‑enzyme review notes that recombinant DNA technology is widely used to produce thermophilic amylases and lipases for baking, starch processing, and oil modification [5]. For example, thermophilic amylase from Bacillus stearothermophilus (amyA) and lipase from Thermomyces lanuginosus (lipA) are expressed recombinantly in Escherichia coli, enabling high‑yield production of heat‑stable enzymes used to improve bread volume, texture, and shelf life, and to tailor lipid profiles in food processing.
4.5 Industrial biotechnology: recombinant leghemoglobin for alternative meats
A 2025 review on recombinant proteins in the food industry reports that soy leghemoglobin is produced recombinantly in microbial systems and used as a flavor‑active ingredient in plant‑based meat analogs [6]. Recombinant leghemoglobin confers meat‑like color and “bloody” flavor notes by binding and releasing heme, thereby improving consumer acceptance of plant‑based burgers while reducing reliance on animal‑derived components.
4.6 Wound healing/biotherapy: recombinant human IL‑33 variant
A 2025 article on new frontiers in functional recombinant protein production describes recombinant humanized mature IL‑33 (rhmatIL‑33) as a therapeutic candidate for tissue repair [7]. In preclinical models, recombinant rhmatIL‑33 accelerated wound closure and modulated local immune responses, suggesting potential as a protein‑based biologic to enhance skin regeneration in both acute and chronic wounds.
Conclusion
Overall, advancements in the field of biotechnology have increased and facilitated the production of recombinant proteins for various applications. Although recombinant proteins still have some drawbacks, their roles in medicine, research, and biotechnology are irreplaceable. We also look forward to seeing more progress in the treatment of various diseases with recombinant proteins.
References
[1] Von Benecke, J. P., Tarsitano, E., Zimmermann, L. M. A., Shakesheff, K. M., Walsh, W. R., & Bae, H. W. (2024). A Narrative Review on Recombinant Human Bone Morphogenetic Protein 2: Where Are We Now [J]? Cureus, 16(8), e67785.
[2] Gattinger, P., Kratzer, B., et al. (2024). Vaccine Based on Recombinant Fusion Protein Combining Hepatitis B Virus PreS with SARS-CoV-2 Wild-Type- and Omicron-Derived Receptor Binding Domain Strongly Induces Omicron-Neutralizing Antibodies in a Murine Model [J]. Vaccines, 12(3), 229.
[3] Yang, J., Hong, W., et al. (2025). A recombinant protein vaccine induces protective immunity against SARS-CoV-2 JN.1 and XBB-lineage subvariants [J]. Signal Transduction and Targeted Therapy, 10(1), 58.
[4] Siddique, A., Wang, Z., et al. (2024). The Evolution of Vaccines Development across Salmonella Serovars among Animal Hosts: A Systematic Review [J]. Vaccines, 12(9), 1067.
[5] Siddikey, F., Jahan, M. I., et al. (2025). Enzyme Technology in the Food Industry: Molecular Mechanisms, Applications, and Sustainable Innovations [J]. Food Science & Nutrition, 13(9), e70927.
[6] Liu M, Xiao R, Li X, Zhao Y, Huang J. A comprehensive review of recombinant technology in the food industry: Exploring expression systems, application, and future challenges [J]. Compr Rev Food Sci Food Saf. 2025 Mar;24(2):e70078.
[7] Luo, X., Ma, Y., & Xiang, Q. (2025). New Frontiers in the Production of Functional Recombinant Proteins [J]. Bioengineering, 12(4), 351.
CUSABIO team. Applications of recombinant proteins. https://www.cusabio.com/c-20272.html






Comments
Leave a Comment