What are Gap Junctions?
Gap junctions (GJs) are a type of cell junction in which adjacent cells are connected through connection channels. These channels act as bridges between the cytoplasm of neighboring cells and allow direct cell-cell transfer of ions, small molecules, and electrical impulses. They are found in almost all animal cells except for some particular cells such as sperm cells and red blood cells.
The Formation of Gap Junction
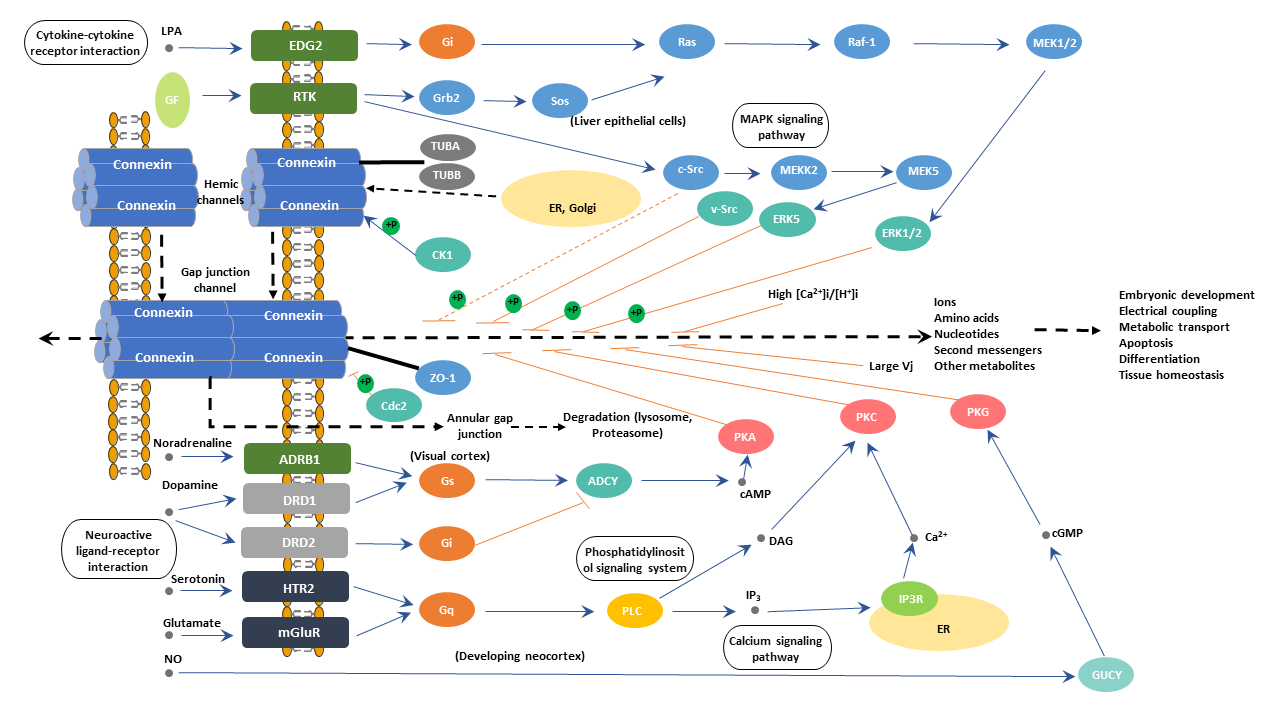
Connexins (Cxs) are the basic elements for the formation of the gap junction. And connexins have four transmembrane, two extracellular, and two intracellular domains. The extracellular regions are responsible for the interaction between connexons on adjacent cells. The intracellular domains contain both the carboxy and amino-terminal, which are targets of post-translational modifications. Post-translational modification is important in the regulation of channel activity. There are 21 Cx isoforms in humans, of which the Cx43 protein is the major cell gap junction protein. The prerequisite for the formation of functional gap junction is the assembly of six connexin proteins into hemichannels (HCs) and their insertion into the membrane. The hemichannel is also called connexon. Hemichannels from one cell need to dock to hemichannels located on a neighboring cell to form an intercellular gap junction channel that allows for the intercellular communication.
The Function of Gap Junction
In addition to establishing intercellular communication between adjacent cells, gap junctions also play an important role in multiple cellular processes such as cell metabolism regulation, growth regulation & differentiation, ontogenesis, and the transmission of electroexcitatory activities in tissues. And studies have found that gap junctions may also function inhibitors in somatic cell mutations because a cell' neighbors could compensate for its loss of a key metabolic enzyme or ion channel.
The Regulation of Gap Junction
The transfer of ions and small molecules through a gap junction is mediated by passive diffusion. Gap junctions allow for the passage of organic ions and molecules with a relative mass weight of less than 1.2 kDa. The charge of these passing molecules or ions regulates transfer selectivity.
And studies have revealed that the type of Cx that makes up the gap junctional channels determines the selectivity and permeability of the gap junctions. Furthermore, connexons composed of two different Cxs showed different conductances than the respective homomeric channels in vivo experiments. Gap junctions composed of different Cxs displayed different selectivity and permeability to second messengers in heterologous systems obtained after cell transfection. Various organs' different or specific function may be associated with differences in the selectivity for second messengers or conductance of their respective gap junctions. Besides, these findings may also explain the diversity and differential tissue distribution of Cx.
Gating mechanisms can also modulate ions and molecules to pass through gap junctions. And the gating of gap junction channels is controlled by voltage differences, decrease in the intracellular pH, and increases in intracellular calcium levels. Cx phosphorylation changes the gating activity of connection channels.
Diseases and Gap Junctions
Studies have confirmed that there are more gap junctions between sites prone to epilepsy such as the hippocampus and cerebral cortical neurons. And changes in gap junctions may lead to the development of epilepsy in individuals with epilepsy susceptibility.
Gap junctions participate in the vasculogenesis and control cell migration and proliferation. Experiments observed that after rat suffered mechanical damage in carotid vascular smooth muscle cells, the expression of Cx43 protein in the rat' carotid intima increased. In cultured endothelial cells, endothelial cell Cx43 protein expression also increased in the injured area. When the cells were transfected with a negative connexin inhibitor, Cx43 protein expression down-regulated and damage repair significantly slowed down.
Moreover, studies have found that tumor cells generally have gap junction communication defects, and the recovery of cell gap junction communication is related to tumor growth control.



