What Are Secreted Extracellular Vesicles?
Secreted extracellular vesicles (EVs) are lipid bilayer-enveloped, cell-derived spheres that contain cytosol cargoes such as nucleic acid, lipids, or proteins. The cell origin of these extracellular vesicles determines their inner content. These extracellular vesicles are secreted and released by various cells into biofluids and intercellular microenvironments. A variety of secreted extracellular vesicles are present in urine, amniotic fluid, breast milk, saliva, blood, and other body fluids. They are highly conserved in both prokaryotes and eukaryotes.
The Classification of Secreted Extracellular Vesicles
Since extracellular vesicles are a heterogeneous class of vesicles with a diverse range of sizes, they are divided into exosomes, microvesicles, and apoptotic bodies according to their physical characteristics, biogenesis or release pathways, and function.
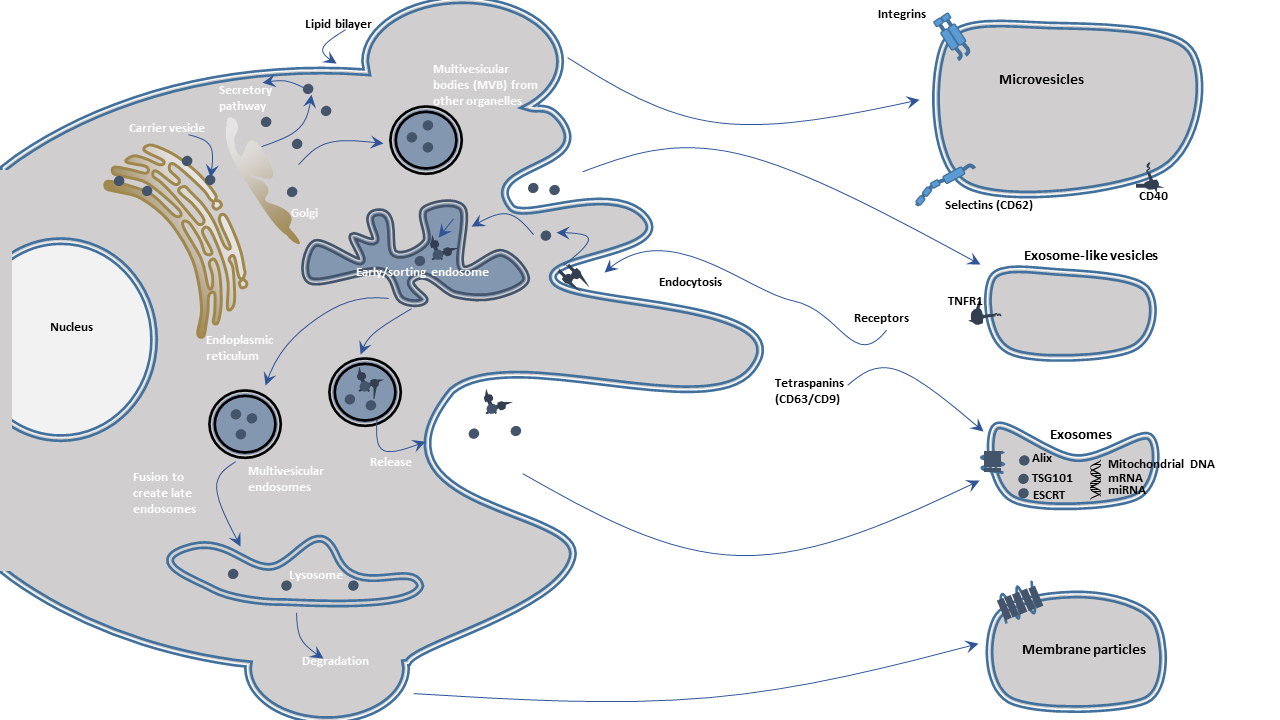
Exosomes are intraluminal vesicles (ILVs) with a diameter of 30 to 150 nm and are enriched in endosome-derived components. They are generated inside multivesicular endosomes or multivesicular bodies (MVBs) and are released upon the fusion of MVBs with the plasma membrane. When observed under an electron microscope, they show a typical cup-shaped or well-defined circular morphology.
Microvesicles are particles with a diameter of 100-1000 nm and are derived from the outward budding of the plasma membrane. Their shape is irregular.
Apoptotic bodies (ApoBDs) are the third form of extracellular vesicles. These vesicles range from 500 nm to 2 μm in size and are released through blebbing by cells undergoing apoptosis. They are often amorphously shaped and sometimes contain significant amounts of DNA.
The Function of Secreted Extracellular Vesicles
Extracellular vesicles play an important role in intercellular communication, immune responses, angiogenesis, signal transduction in health and diseases. Exosomes mediate cell-to-cell communication by delivering parent cells-derived cargoes to the recipient cells and modulate and influence their physiological condition. They mediate multiple signaling pathways through the exchange of genetic material between cells. Exosomes are also involved in tissue repair, central nervous system (CNS) communication, and pathological processes in neurodegeneration, inflammation, cancer, and cardiovascular diseases. They are also pathogenic factors or diagnostic biomarkers in some diseases because their contents reflect the cellular origin of different organ systems and the pathology. Besides, exosomes are designed as therapeutic cargo carriers targeted the diseased cells or tissues. Apoptotic bodies are involved in removing cellular contents.
Studies have reported that fungal species-derived extracellular vesicles are proposed to play a part in numerous host-pathogen interactions in both plants and animals. In fungi, extracellular vesicles may participate in the transport of macromolecules across the fungal cell wall.
The Formation Process of Secreted Extracellular Vesicles
Endocytic events such as phagocytosis, pinocytosis, receptor-mediated endocytosis pull membrane and extracellular proteins into the early endosomes. Alterations in endosomes including internal lumenal pH, phospholipids, and RAB GTPases during the endosomal trafficking confer the character of the endosomes. As the endosomes mature, they may interact with vesicles of the trans-Golgi network and progress toward multivesicular bodies. During the process, the endosomal membrane invaginates or reversely buds possibly due to the alterations in pH, membrane lipids, and functions of the endosomal sorting complexes required for transport. Cytosolic components such as proteins, nucleic acids are packaged into the invaginations at the endosomal limiting membrane surfaces, which fuse with the membrane into the lumenal interior, forming small vesicles termed "intraluminal vesicles" (ILVs). The late endosomes/MVBs proceed to degrade by fusing with lysosomes. Alternatively, MVBs fuse with the plasma membrane to release the ILVs into the extracellular space, these extracellular vesicles are exosomes. Exosomal formation and MVB transportation are modulated by the endosomal sorting complexes required for transport (ESCRT) such as Alix and TSG101. These proteins and exosomal marker proteins such as CD9, CD63, CD81, HSC70, and HSP90β, can be found in exosomes regardless of the cell type of cell from which they originate.
Microvesicles (MVs) arise from membrane blebbing or outward protrusion of the plasma membrane. As with the generation of MVBs, cytosolic components may enter into the membrane extensions. These blebs pinch-off, releasing membrane vesicles extracellularly. The process disrupts the actin cytoskeleton and membrane remodeling.
Apoptotic bodies are formed during apoptosis, involving protease activation, DNA fragmentation, and externalization of phosphatidylserine. Organelles degenerate, and various cytoplasmic blebs may arise, changing the shape of the cell and resulting in the release of apoptotic bodies. The formation of apoptotic bodies is considered a hallmark of apoptosis.
The Pathway of Secreted Extracellular Vesicles
All extracellular vesicles contain surface molecules that allow them to be targeted to recipient cells. Once attached to a target cell, extracellular vesicles can trigger signaling by direct receptor-ligand interaction or by fusing with the target cell's plasma membrane to deliver their content into its cytosol. Extracellular vesicles can also be internalized by endocytosis and/or phagocytosis. The eventual results of these processes are modification and regulation of the physiological state of the recipient cell.
As science and medicine rapidly advance, the role of exosomes continues to be discovered. The importance of exosomes has been gradually realized in scientific research and clinical diagnosis & treatment. So exosome-associated research has become a hotspot now. Correspondingly, the extraction of exosomes is particularly important. CUSABIO offers several exosome isolation kits for those who are worried about exosome extraction. These exosome isolation kits are characterized with high purity, high yield, and easy operation.



