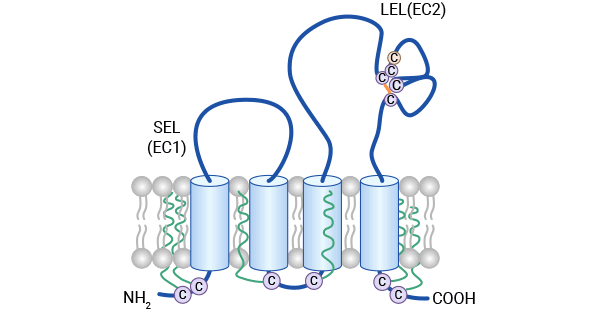
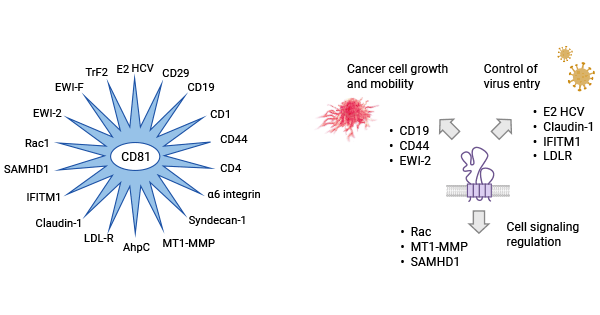
[1] Bailly, Christian, and Xavier Thuru. "Targeting of Tetraspanin CD81 with Monoclonal Antibodies and Small Molecules to Combat Cancers and Viral Diseases." Cancers 15.7 (2023): 2186.
[2] Fénéant, Lucie, Shoshana Levy, and Laurence Cocquerel. "CD81 and hepatitis C virus (HCV) infection." Viruses 6.2 (2014): 535-572.
[3] Koutsoudakis, George, et al. "The level of CD81 cell surface expression is a key determinant for productive entry of hepatitis C virus into host cells." Journal of virology 81.2 (2007): 588-598.
[4] Akazawa, Daisuke, et al. "CD81 expression is important for the permissiveness of Huh7 cell clones for heterogeneous hepatitis C virus infection." Journal of virology 81.10 (2007): 5036-5045.
[5] Inoue, G. A. K. U., N. O. R. I. O. Horiike, and M. O. R. I. K. A. Z. U. Onji. "The CD81 expression in liver in hepatocellular carcinoma." International journal of molecular medicine 7.1 (2001): 67-138.
[6] Bailly, Christian, and Xavier Thuru. "Targeting of Tetraspanin CD81 with Monoclonal Antibodies and Small Molecules to Combat Cancers and Viral Diseases." Cancers 15.7 (2023): 2186.
[7] Wünschmann, Sabina, et al. "Characterization of hepatitis C virus (HCV) and HCV E2 interactions with CD81 and the low-density lipoprotein receptor." Journal of virology 74.21 (2000): 10055-10062.
[8] Todros-Dawda, Izabela, et al. "The tetraspanin CD53 modulates responses from activating NK cell receptors, promoting LFA-1 activation and dampening NK cell effector functions." PLoS One 9.5 (2014): e97844.
[9] CHAN12, BOSCO MC, et al. "Integrin α2βΐ on rat myeloma cells modulates interaction of α4β1 integrin with vascular cell adhesion molecule-1 but not hbronectin." Cells, Proteins and Materials: Festschrift in Honor of the 65th Birthday of Dr. John L. Brash (2003): 119.
[10] Tippett, Emma, et al. "Characterization of tetraspanins CD9, CD53, CD63, and CD81 in monocytes and macrophages in HIV-1 infection." Journal of Leukocyte Biology 93.6 (2013): 913-920.
[11] Rubio, Karla, et al. "Extracellular vesicles induce aggressive lung cancer via non-canonical integrin-EGFR-KRAS signaling." bioRxiv (2022): 2022-08.
[12] Rubio, Karla, et al. "Non-canonical integrin signaling activates EGFR and RAS-MAPK-ERK signaling in small cell lung cancer." Theranostics 13.8 (2023): 2384.
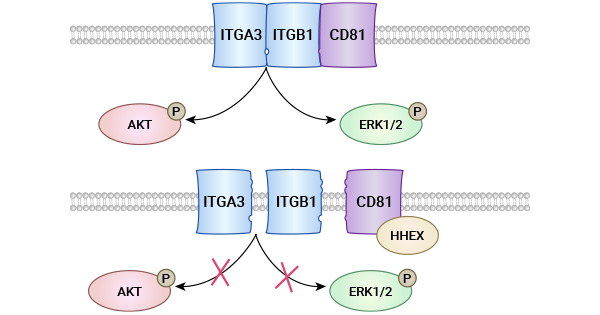
[13] Guo Wenjie. Molecular mechanism of HHEX regulating integrin signaling pathway in lung cancer cells through CD81 [D]. Shandong University, 2021.
[14] Meertens, Laurent, et al. "The tight junction proteins claudin-1,-6, and-9 are entry cofactors for hepatitis C virus." Journal of virology 82.7 (2008): 3555-3560.
[15] Farquhar, Michelle J., et al. "Hepatitis C virus induces CD81 and claudin-1 endocytosis." Journal of virology 86.8 (2012): 4305-4316.
[16] Krieger, Sophie E., et al. "Inhibition of hepatitis C virus infection by anti‐claudin‐1 antibodies is mediated by neutralization of E2–CD81–Claudin‐1 associations." Hepatology 51.4 (2010): 1144-1157.
[17] Harris, Helen J., et al. "CD81 and claudin 1 coreceptor association: role in hepatitis C virus entry." Journal of virology 82.10 (2008): 5007-5020.
[18] Fofana, Isabel, et al. "Functional analysis of claudin-6 and claudin-9 as entry factors for hepatitis C virus infection of human hepatocytes by using monoclonal antibodies." Journal of virology 87.18 (2013): 10405-10410.
[19] Vences-Catalán, Felipe, et al. "CD81 as a tumor target." Biochemical Society Transactions 45.2 (2017): 531-535.
[20] Belov, Larissa, et al. "Extensive surface protein profiles of extracellular vesicles from cancer cells may provide diagnostic signatures from blood samples." Journal of extracellular vesicles 5.1 (2016): 25355.
[21] Li, Yandong, et al. "KLF4-mediated upregulation of CD9 and CD81 suppresses hepatocellular carcinoma development via JNK signaling." Cell death & disease 11.4 (2020): 299.
[22] Yoo, Tae-Hyoung, et al. "CD81 is a candidate tumor suppressor gene in human gastric cancer." Cellular Oncology 36 (2013): 141-153.
[23] Hong, In-Kee, et al. "The tetraspanin CD81 protein increases melanoma cell motility by up-regulating metalloproteinase MT1-MMP expression through the pro-oncogenic Akt-dependent Sp1 activation signaling pathways." Journal of Biological Chemistry 289.22 (2014): 15691-15704.
[24] Hong, In-Kee, et al. "Tetraspanin CD81/TAPA-1 up-regulates MT1-MMP involved in melanoma cell motility through Akt-dependent Sp1 activation signaling pathways." Cancer Research 70.8_Supplement (2010): 526-526.
[25] Vences-Catalán, Felipe, et al. "Tetraspanin CD81 promotes tumor growth and metastasis by modulating the functions of T regulatory and myeloid-derived suppressor cells." Cancer research 75.21 (2015): 4517-4526.
[26] Fan, Weijian, et al. "Human umbilical cord mesenchymal stem cell-derived exosomes promote microcirculation in aged diabetic mice by TGF-β1 signaling pathway." Diabetology & Metabolic Syndrome 15.1 (2023): 234.
[27] Xie, Xin-hui, et al. "Hyper-inflammation of astrocytes in patients of major depressive disorder: Evidence from serum astrocyte-derived extracellular vesicles." Brain, Behavior, and Immunity 109 (2023): 51-62.
[28] Ahmad, Sarfaraz, et al. "Chymase in Plasma and Urine Extracellular Vesicles: Novel Biomarkers for Primary Hypertension." medRxiv (2023): 2023-11.
[29] Benke, Márton, et al. "MiR-200b categorizes patients into pancreas cystic lesion subgroups with different malignant potential." Scientific Reports 13.1 (2023): 19820.
[30] Chen, Zhilong, et al. "Exosome-delivered circRNA circSYT15 contributes to cisplatin resistance in cervical cancer cells through the miR-503-5p/RSF1 axis." Cell Cycle (2023): 1-18.



Comments
Leave a Comment