Caspase-3 stands as the central executioner protease in apoptotic cell death, cleaving critical cellular substrates during programmed cell death. This cysteine protease exists as an inactive 32 kDa proenzyme that undergoes proteolytic processing into active p17 and p12 subunits upon apoptotic stimulation. Understanding caspase-3 activation and expression patterns provides essential insights into cancer biology, cellular stress responses, and metabolic regulation, making it a cornerstone target for researchers investigating cell fate decisions.
This recombinant monoclonal antibody, generated against a synthetic peptide derived from human pro-caspase-3, offers the reproducibility that demanding experimental workflows require. Because the antibody sequence is defined and production occurs in a controlled recombinant system, you can expect consistent performance across experiments and between lot numbers, eliminating the variability that can complicate long-term studies or multi-site collaborations.
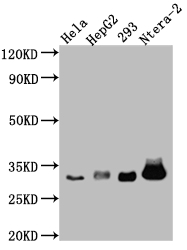
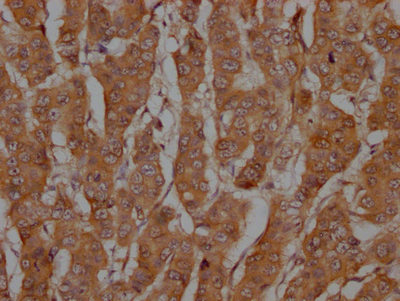
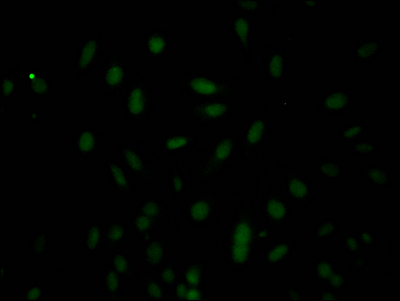
Validation data demonstrates reliable detection of the 32 kDa pro-caspase-3 form across multiple human cell lines in western blot applications, including HeLa, HepG2, HEK293, and Ntera-2 lysates at 1:1000 dilution. The observed band size matches the predicted molecular weight precisely, confirming specific target recognition. Beyond western blotting, this antibody performs effectively in immunohistochemistry, where it has been validated in paraffin-embedded human breast cancer tissue sections, and in immunofluorescence applications using HeLa cells. This application flexibility allows you to track caspase-3 expression from biochemical analysis through tissue-level and single-cell imaging studies.
Whether you are investigating apoptotic pathway activation in cancer models, examining metabolic stress responses, or characterizing cell death mechanisms, this antibody provides a dependable tool for detecting pro-caspase-3 in human samples across multiple experimental platforms.