[1] Wu, W., Chia, T., Lu, J. et al. IL-2Rα-biased agonist enhances antitumor immunity by invigorating tumor-infiltrating CD25+CD8+ T cells. Nat Cancer ( 2023). https://doi.org/10.1038/s43018-023-00612-0
[2] Dhawan, Manish, et al. "Regulatory T Cells (Tregs) and COVID-19: Unveiling the Mechanisms, and Therapeutic Potentialities with a Special Focus on Long COVID." Vaccines 11.3 (2023): 699.
[3] Sakaguchi, Shimon, et al. "Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases." Journal of immunology (Baltimore, Md.: 1950) 155.3 (1995): 1151-1164. 1151-1164.
[4] Maes, Michael. T cell activation via the CD40 ligand and transferrin receptor and deficits in T regulatory cells are associated with major depressive disorder and severity of depression. T cell activation via the CD40 ligand and transferrin receptor and deficits in T regulatory cells are associated with major depressive disorder and severity of depression.
[5] Walker, Lucy SK. "CD4+ CD25+ Treg: divide and rule?." Immunology 111.2 (2004): 129-137.
[6] Wan, Yuling, et al. "Hyperfunction of CD4 CD25 regulatory T cells in de novo acute myeloid leukemia." BMC cancer 20 (2020): 1-10.
[7] de Goër de Herve, Marie-Ghislaine, et al. "CD25 appears non essential for human peripheral Treg maintenance in vivo." PloS one 5.7 (2010): e11784.
[8] Spangler, Jamie B., et al. "Antibodies to interleukin-2 elicit selective T cell subset potentiation through distinct conformational mechanisms." Immunity 42.5 (2015): 815-825.
[9] Hsieh, Elena WY, and Joseph D. Hernandez. "Clean up by aisle 2: roles for IL-2 receptors in host defense and tolerance." Current Opinion in Immunology 72 ( 2021): 298-308.
[10] Ye, Congxiu, David Brand, and Song G. Zheng. "Targeting IL-2: an unexpected effect in treating immunological diseases." Signal transduction and targeted therapy 3.1 (2018): 2.
[11] Wang, Xinquan, Mathias Rickert, and K. Christopher Garcia. "Structure of the Quaternary Complex of Interleukin-2 with Its α, ß, and γc Receptors." Science 310.5751 (2005): 1159-1163.
[12] Amaria, Rodabe N., et al. "Update on use of aldesleukin for treatment of high-risk metastatic melanoma." ImmunoTargets and therapy (2015): 79-89.
[13] Tilley, Jefferson W., et al. "Identification of a small molecule inhibitor of the IL-2/IL-2Rα receptor interaction which binds to IL-2." Journal of the American Chemical Society 119.32 (1997): 7589-7590.
[14] Zhang, Bo, et al. "Proximity-enabled covalent binding of IL-2 to IL-2Rα selectively activates regulatory T cells and suppresses autoimmunity." Signal Transduction and Targeted Therapy 8.1 (2023): 28.
[15] Shatrova, Alla N., et al. "Time-dependent regulation of IL-2R α-chain (CD25) expression by TCR signal strength and IL-2-induced STAT5 signaling in activated human blood T lymphocytes." PLoS One 11.12 (2016): e0167215.
[16] Horwitz, David A et al. "Natural and TGF-beta-induced Foxp3(+)CD4(+) CD25(+) regulatory T cells are not mirror images of each other. " Trends in immunology vol. 29, 9 (2008): 429-35. doi:10.1016/j.it.2008.06.005
[17] Nasiri, Mahboobeh, and Zarnegar Rasti. "CTLA-4 and IL-6 gene polymorphisms: risk factors for recurrent pregnancy loss." human immunology 77.12 (2016). : 1271-1274.
[18] Ware, Michael Brandon, et al. "Dual IL-6 and CTLA-4 blockade regresses pancreatic tumors in a T cell-and CXCR3-dependent manner." JCI insight 8.8 (2023).
[19] Peng, Yujia, et al. "CD25: A potential tumor therapeutic target." International Journal of Cancer 152.7 (2023): 1290-1303.
[20] Qureshi, Farhan M., et al. "Immunotherapy With Low-Dose IL-2/CD25 Prevents β-Cell Dysfunction and Dysglycemia in Prediabetic NOD Mice." Diabetes 72.6 (2023): 769-780.
[21] Eissa, Amal H., et al. "Protective role of T regulatory (Treg) cells in systemic lupus erythematosus patients with nephritis." The Egyptian Rheumatologist 45.1 (2023): 61-65.
[22] Pfender, Nikolai, and Roland Martin. "Daclizumab (anti-CD25) in multiple sclerosis." Experimental neurology 262 (2014): 44-51.
[23] Lawson, C. A., et al. "Early rheumatoid arthritis is associated with a deficit in the CD4+ CD25 high regulatory T cell population in peripheral blood." Rheumatology 45.10 (2006): 1210-1217.
[24] Mrowietz, Ulrich, Kejian Zhu, and Enno Christophers. "Treatment of severe psoriasis with anti-CD25 monoclonal antibodies. "Archives of Dermatology 136.5 (2000): 675-676.
[25] Abdulahad, Wayel H., et al. "Functional defect of circulating regulatory CD4+ T cells in patients with Wegener's granulomatosis in remission." Arthritis & Rheumatism 56.6 (2007): 2080-2091.
[26] Zhang, Min, et al. "Expression of immune molecules CD25 and CXCL13 correlated with clinical severity of myasthenia gravis." Journal of Molecular Neuroscience 50 (2013): 317-323.
[27] Chen, Alex, et al. "NEO-TRA1: A CD25-Targeted De Novo non-Alpha Agonist of the IL-2 Receptor Selectively Expands Regulatory T Cells." Blood 140. Supplement 1 (2022): 1652-1653.
[28] Schneider, Jessica, et al. "Healthy-like CD4+ regulatory and CD4+ conventional T-cell receptor repertoires predict protection from GVHD following donor lymphocyte infusion." International Journal of Molecular Sciences 23.18 (2022): 10914.
[29] Rech, Andrew J., and Robert H. Vonderheide. "Clinical use of anti-CD25 antibody daclizumab to enhance immune responses to tumor antigen vaccination by targeting regulatory T cells." Annals of the New York Academy of Sciences 1174.1 (2009): 99-106.
[30] Li, Hui, et al. "CD 4+ CD 25+ Regulatory T Cells Decreased the Antitumor Activity of Cytokine-Induced Killer (CIK) Cells of Lung Cancer Patients." Journal of clinical immunology 27 (2007): 317-326.
[31] Gönen, Mithat, et al. "CD25 expression status improves prognostic risk classification in AML independent of established biomarkers: ECOG phase 3 trial , E1900." Blood, The Journal of the American Society of Hematology 120.11 (2012): 2297-2306.
[32] Nakase, Kazunori, et al. "CD22 expression in acute myeloid leukemia: close correlation with interleukin-2 receptor α-chain (CD25) expression and poor prognosis." Leukemia & Lymphoma 63.9 (2022): 2251-2253.
[33] Woo, Edward Y., et al. "Regulatory CD4+ CD25+ T cells in tumors from patients with early-stage non-small cell lung cancer and late-stage ovarian cancer." Cancer research 61.12 (2001): 4766-4772.
[34] deLeeuw, Ronald J., et al. "CD25 identifies a subset of CD4+ FoxP3- TIL that are exhausted yet prognostically favorable in human ovarian cancer." Cancer immunology research 3.3 (2015): 245-253.
[35] Stuller, Kathleen A., et al. "CD25+ T cells induce Helicobacter pylori-specific CD25- T-cell anergy but are not required to maintain persistent hyporesponsiveness." European journal of immunology 38.12 (2008): 3426-3435.
[36] Miao, Y., et al. "Association analysis of the IL2RA gene with alopecia areata in a Chinese population." Dermatology 227.4 (2014): 299-304.
[37] Dimova, Tanya, et al. "Maternal Foxp3 expressing CD4+ CD25+ and CD4+ CD25- regulatory T-cell populations are enriched in human early normal pregnancy decidua: A phenotypic study of paired decidual and peripheral blood samples." American journal of reproductive immunology 66 (2011): 44-56.
[38] Li, Ting-ting, et al. "Effect and Mechanism of Sodium Butyrate on Neuronal Recovery and Prognosis in Diabetic Stroke." Journal of Neuroimmune Pharmacology (2023): 1-17.

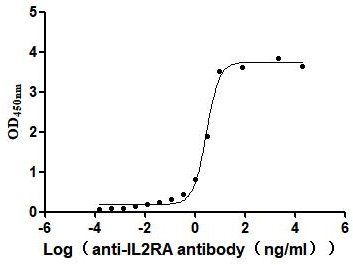
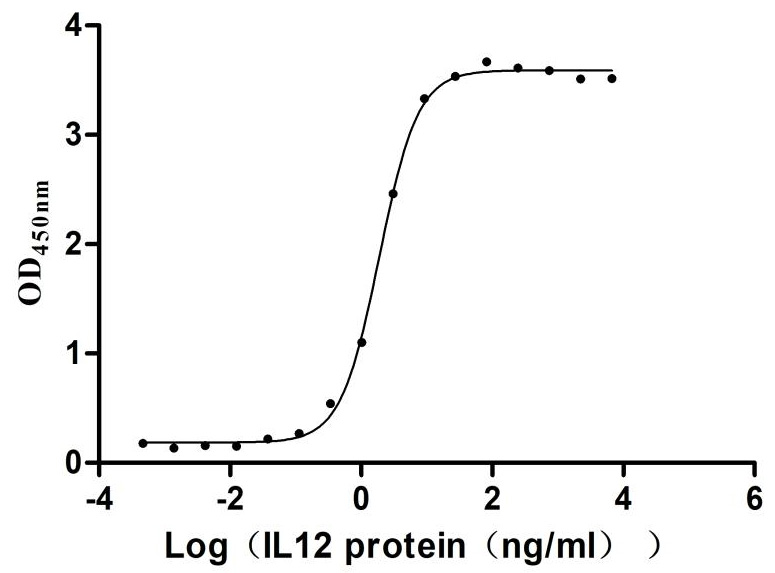
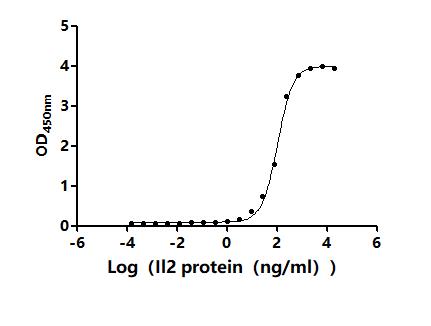
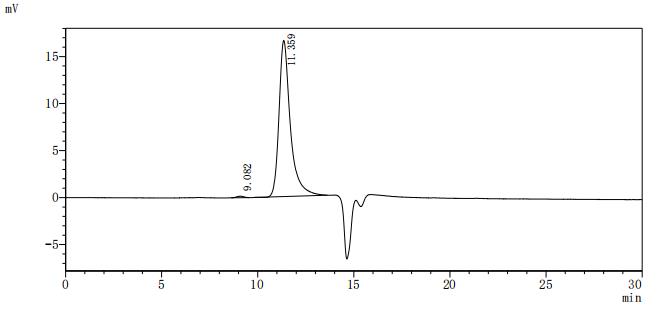
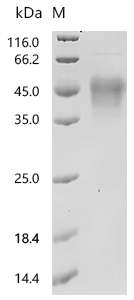
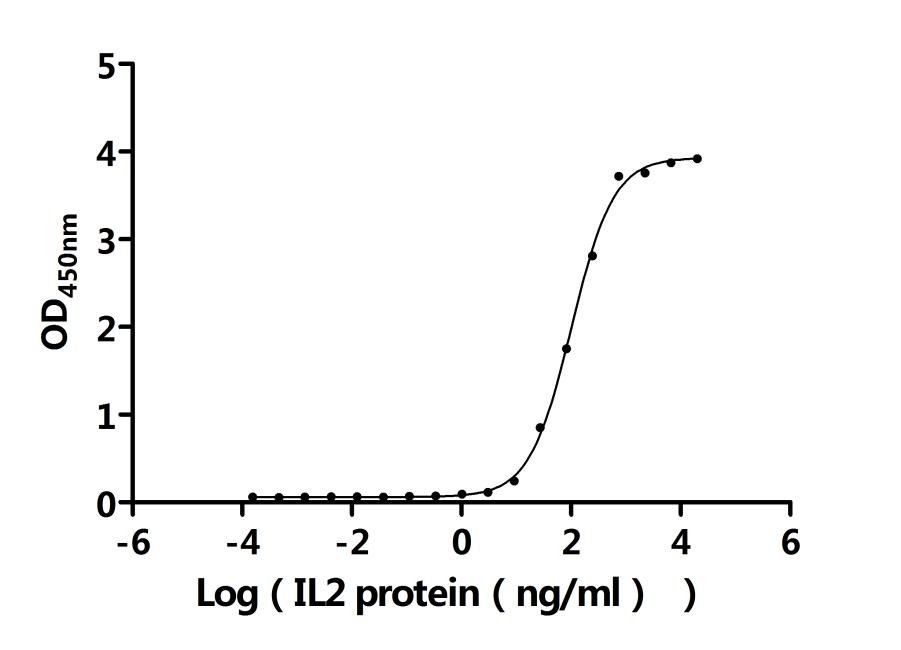



Comments
Leave a Comment