What Is Inflammatory Pain?
Inflammatory pain refers to increased sensitivity that results from inflammatory responses linked to tissue damage. It is the perception and affective response to detrimental stimuli that take place during an inflammatory or immune response. Pain caused by inflamed or injured tissues may arise spontaneously without an external trigger, that is, normally harmless stimuli could also cause inflammatory pain.
Inflammatory pain is mainly characterized by hypersensitivity both at the inflammatory center and in the adjacent tissue and the decline in the threshold to nociceptor activation even under innocuous stimuli.
Symptoms of Inflammatory Pain
Inflammatory pain is characterized by five classic symptoms: redness, heat, swelling, pain or hypersensitivity, and loss of function.
The Mechanisms of Inflammatory Pain
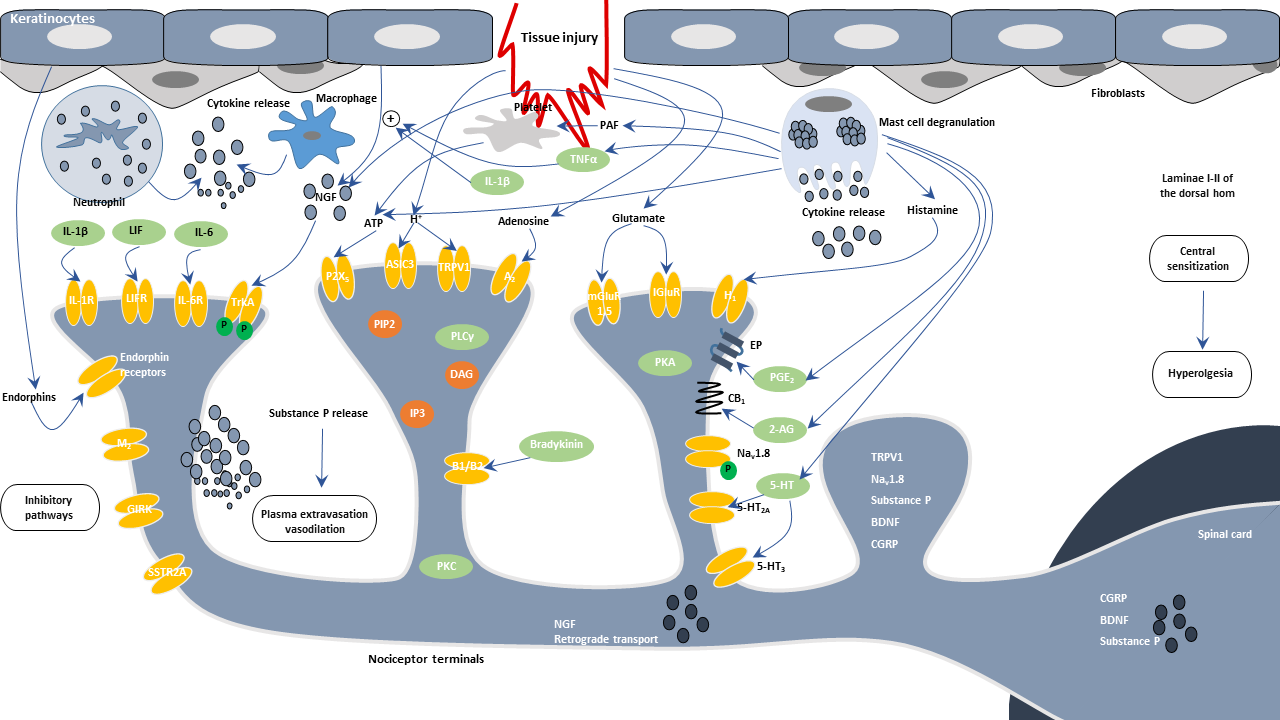
Inflammatory pain is a kind of nociceptor pain caused by inflammatory factors-mediated activation and sensitization of nociceptors.
Inflammatory pain is achieved through complex mechanisms in which receptors, ion channels, and neurotransmitters or regulatory proteins are regulated by mechanical, thermal, and chemical transduction. Nociceptors are excitatory neurons with free nerve endings. They branch out from the axons and innervate the dermis and epidermis. They are responsible for "converting" harmful signals into action potentials. The inflammatory mediators released from the nociceptive terminal increase vascular permeability, leading to local edema and escape of injury by-products, such as prostaglandins, bradykinin, growth factors, and cytokines. Inflammatory pain is largely due to prostaglandins, which are generated in response to inflammation and tissue insults primarily by inducible cyclooxygenase-2 (COX-2). These substances can sensitize nociceptors and excite them, resulting in declined firing thresholds and ectopic discharges. However, these analgesics decrease the activation threshold and can alter the characteristics of main sensory neurons around the wounded area, leading to pain sensation near the actual injured area.
At the cellular level, neuropeptides such as substance P released from the injured site stimulate resident immune cells such as mast cells to secrete cytokines, leading to vasodilation and plasma extravasation. A large number of these secreted molecules also directly or indirectly act on nociceptor ion channels and receptors, leading to hypersensitivity.
The Biological Significance of Inflammatory Pain
Inflammatory pain, part of nociception, is a critical component of our body's defense system because it makes us extremely uncomfortable and unpleasant. Inflammatory pain is not only an alarm that alerts the body to potential harm but also more serves as a reminder of a recent injury, discouraging activities that risk re-injury so that recovery can proceed quickly. Inflammatory pain is associated with tissue damage and the resulting inflammatory process. It is adaptive in that it triggers physiologic responses that promote healing.
Treatment and Control of Inflammatory Pain
Effective control of inflammatory pain may be the result of the interaction of opioid peptides derived from leukocytes with receptors on peripheral sensory neurons. Classical non-specific cyclooxygenase inhibitors such as aspirin and related drugs or recently developed COX-2 specific inhibitors suppress prostaglandin formation, thus achieving profound analgesia.



